Korean Circ J.
2021 Dec;51(12):1001-1014. 10.4070/kcj.2021.0161.
A First-in-Man Clinical Evaluation of Sirolimus and Ascorbic Acid-Eluting Stent Systems: a Multicenter, Subject-Blinded, Randomized Study
- Affiliations
-
- 1Division of Cardiology, Department of Internal Medicine, Hanyang University College of Medicine, Seoul, Korea
- 2CG Bio Co. Ltd., Seoul, Korea
- 3Division of Cardiology, Cardiovascular Center, Korea University Anam Hospital, Seoul, Korea
- 4Department of Cardiology, Yonsei University Wonju Severance Christian Hospital, Wonju, Korea
- 5Department of Internal Medicine and Cardiovascular Center, Seoul National University College of Medicine, Seoul, Korea
- KMID: 2523152
- DOI: http://doi.org/10.4070/kcj.2021.0161
Abstract
- Background and Objectives
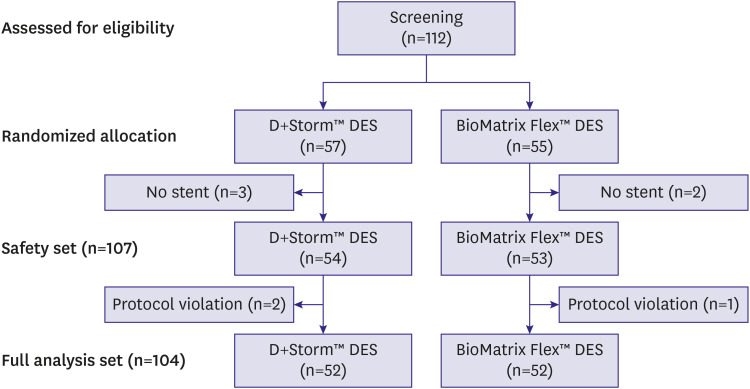
This clinical trial was conducted to evaluate the safety and efficacy of D+Storm™ drug-eluting stent (DES) and BioMatrix Flex™ DES.
Methods
This study was a multicenter, subject-single-blind, randomized, and confirmed comparative clinical trial. According to the inclusion criteria, those diagnosed with stable angina, unstable angina, silent ischemia, or non-ST-segment myocardial infarction were selected among patients with coronary artery stenosis as subjects. Among the subjects with 50% stenosis on coronary angiography, the experiment was performed on those who had a lesion with reference vessel 2.5–4.0 mm in diameter and ≤40 mm in length. The primary endpoint was an in-segment late loss and the secondary endpoints were in-stent late lumen loss, stent malapposition, the incidence of mortality, myocardial infarction, reoperation, and stent thrombosis at 36 weeks.
Results
57 patients in the D+Storm™ DES group and 55 patients in the BioMatrix Flex™ DES group were enrolled in the study. Fifty-seven patients in the D+Storm™ DES group and Fifty-five patients in the BioMatrix Flex™ DES group were enrolled in the study. An average of in-segment late lumen loss was 0.08±0.13 mm in the D+Storm™ DES group and 0.14±0.32 mm in the BioMatrix Flex™ DES group with no significant difference between the 2 groups (p=0.879). In addition, there was no significant difference in adverse events between D+Storm™ DES and BioMatrix Flex™ DES.
Conclusions
This study demonstrated the clinical effectiveness and safety of D+Storm™ DES implantation in patients with coronary artery disease over a 36-week follow-up period.
Figure
Cited by 1 articles
-
Reduction of In-Stent Restenosis and Inflammation with One Stent: New Concept of Sirolimus and Ascorbic Acid-Eluting Coronary Stent
JiWung Ryu, Jung-Sun Kim
Korean Circ J. 2021;51(12):1015-1016. doi: 10.4070/kcj.2021.0336.
Reference
-
1. Shin ES. Current status of coronary stent. Korean J Med. 2015; 89:282–290.
Article2. Choi C, Nah JW, Park JK. Development trends of the stent for coronary artery. Korean Ind Chem News. 2015; 18:10–24.3. Moses JW, Leon MB, Popma JJ, et al. Sirolimus-eluting stents versus standard stents in patients with stenosis in a native coronary artery. N Engl J Med. 2003; 349:1315–1323. PMID: 14523139.
Article4. Fioranelli M, Bottaccioli AG, Bottaccioli F, Bianchi M, Rovesti M, Roccia MG. Stress and inflammation in coronary artery disease: a review psychoneuroendocrineimmunology-based. Front Immunol. 2018; 9:2031. PMID: 30237802.
Article5. Bedair TM, Kang SN, Joung YK, Han DK. A promising approach for improving the coating stability and in vivo performance of biodegradable polymer-coated sirolimus-eluting stent. J Biomed Nanotechnol. 2016; 12:2015–2028. PMID: 29364616.6. El-Hayek G, Bangalore S, Casso Dominguez A, et al. Meta-analysis of randomized clinical trials comparing biodegradable polymer drug-eluting stent to second-generation durable polymer drug-eluting stents. JACC Cardiovasc Interv. 2017; 10:462–473. PMID: 28279314.
Article7. Lee JH, Park JH. Can Genoss DES™ stand out in the crowd of stents? Korean Circ J. 2020; 50:328–329. PMID: 32157832.
Article8. Lee DH, de la Torre Hernandez JM. The newest generation of drug-eluting stents and beyond. Eur Cardiol. 2018; 13:54–59. PMID: 30310472.
Article9. Reejhsinghani R, Lotfi AS. Prevention of stent thrombosis: challenges and solutions. Vasc Health Risk Manag. 2015; 11:93–106. PMID: 25657588.10. Wu KK, Thiagarajan P. Role of endothelium in thrombosis and hemostasis. Annu Rev Med. 1996; 47:315–331. PMID: 8712785.11. Llau JV, Ferrandis R, Sierra P, Gómez-Luque A. Prevention of the renarrowing of coronary arteries using drug-eluting stents in the perioperative period: an update. Vasc Health Risk Manag. 2010; 6:855–867. PMID: 20957131.
Article12. Saeed RW, Peng T, Metz CN. Ascorbic acid blocks the growth inhibitory effect of tumor necrosis factor-alpha on endothelial cells. Exp Biol Med (Maywood). 2003; 228:855–865. PMID: 12876306.13. Heller R, Unbehaun A, Schellenberg B, Mayer B, Werner-Felmayer G, Werner ER. L-ascorbic acid potentiates endothelial nitric oxide synthesis via a chemical stabilization of tetrahydrobiopterin. J Biol Chem. 2001; 276:40–47. PMID: 11022034.
Article14. Buccheri D, Piraino D, Andolina G, Cortese B. Understanding and managing in-stent restenosis: a review of clinical data, from pathogenesis to treatment. J Thorac Dis. 2016; 8:E1150–E1162. PMID: 27867580.
Article15. Lim KS, Park JK, Jeong MH, et al. Long-term preclinical evaluation of bioabsorbable polymer-coated drug-eluting stent in a porcine model. Macromol Res. 2017; 25:730–738.
Article16. Hoffmann R, Mintz GS, Dussaillant GR, et al. Patterns and mechanisms of in-stent restenosis. A serial intravascular ultrasound study. Circulation. 1996; 94:1247–1254. PMID: 8822976.17. Gordon PC, Gibson CM, Cohen DJ, Carrozza JP, Kuntz RE, Baim DS. Mechanisms of restenosis and redilation within coronary stents--quantitative angiographic assessment. J Am Coll Cardiol. 1993; 21:1166–1174. PMID: 8459072.
Article18. Morice MC, Serruys PW, Sousa JE, et al. A randomized comparison of a sirolimus-eluting stent with a standard stent for coronary revascularization. N Engl J Med. 2002; 346:1773–1780. PMID: 12050336.
Article19. Wenaweser P, Daemen J, Zwahlen M, et al. Incidence and correlates of drug-eluting stent thrombosis in routine clinical practice. 4-year results from a large 2-institutional cohort study. J Am Coll Cardiol. 2008; 52:1134–1140. PMID: 18804739.20. Roukoz H, Bavry AA, Sarkees ML, et al. Comprehensive meta-analysis on drug-eluting stents versus bare-metal stents during extended follow-up. Am J Med. 2009; 122:581.e1–581.e10.
Article21. Joner M, Nakazawa G, Finn AV, et al. Endothelial cell recovery between comparator polymer-based drug-eluting stents. J Am Coll Cardiol. 2008; 52:333–342. PMID: 18652940.
Article22. Kim JS, Jang IK, Kim JS, et al. Optical coherence tomography evaluation of zotarolimus-eluting stents at 9-month follow-up: comparison with sirolimus-eluting stents. Heart. 2009; 95:1907–1912. PMID: 19535352.
Article23. Aguirre R, May JM. Inflammation in the vascular bed: importance of vitamin C. Pharmacol Ther. 2008; 119:96–103. PMID: 18582947.
Article24. Grube E, Buellesfeld L. BioMatrix Biolimus A9-eluting coronary stent: a next-generation drug-eluting stent for coronary artery disease. Expert Rev Med Devices. 2006; 3:731–741. PMID: 17280537.25. Ahmad WA, Hai KT, Santoso T, et al. Long-term outcomes with Biolimus A9-elutiong stents in real-world, all-comers Asia Pacific patients. Final 5-year report of the BEACON (Biolimus Eluting A9 Coronary Stent Obviating Luminal Narrowing) II clinical registry. AsiaIntervention. 2015; 1:102–108.26. Maehara A, Ben-Yehuda O, Ali Z, et al. Comparison of stent expansion guided by optical coherence tomography versus intravascular ultrasound: the ILUMIEN II study (observational study of optical coherence tomography [OCT] in patients undergoing fractional flow reserve [FFR] and percutaneous coronary intervention). JACC Cardiovasc Inter. 2015; 8:1704–1714.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Very Late Stent Thrombosis Related to Fracture of a Sirolimus-Eluting Stent
- Reduction of In-Stent Restenosis and Inflammation with One Stent: New Concept of Sirolimus and Ascorbic Acid-Eluting Coronary Stent
- A Case of Stent Thrombosis Occurred at 5 Years after Sirolimus-Eluting Stent Implantation
- Drug-Eluting Stent Strut Fracture as a Cause of Restenosis
- A Case of Coronary Artery Aneurysm after Sirolimus-Eluting Stent Implantation