J Korean Med Sci.
2021 Nov;36(44):e299. 10.3346/jkms.2021.36.e299.
Data Pseudonymization in a Range That Does Not Affect Data Quality: Correlation with the Degree of Participation of Clinicians
- Affiliations
-
- 1Department of Digital Health, Samsung Advanced Institute for Health Sciences & Technology (SAIHST), Sungkyunkwan University, Seoul, Korea
- 2Center for Research Resource Standardization, Samsung Medical Center, Seoul, Korea
- 3Department of Medical Informatics, College of Medicine, The Catholic University of Korea, Seoul, Korea
- 4Division of Endocrinology and Metabolism, Department of Internal Medicine, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
- KMID: 2522362
- DOI: http://doi.org/10.3346/jkms.2021.36.e299
Abstract
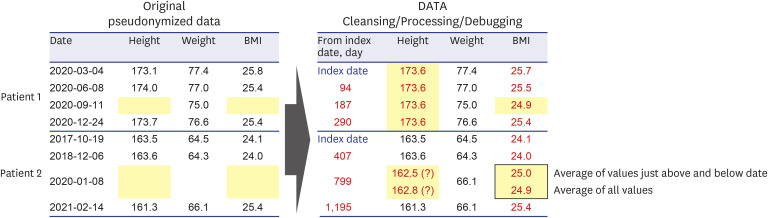
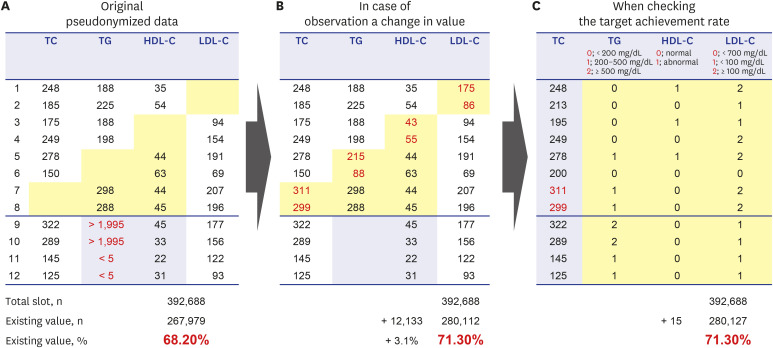
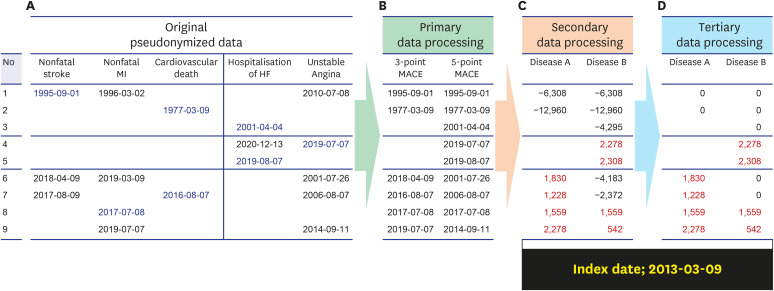
- Personal medical information is an essential resource for research; however, there are laws that regulate its use, and it typically has to be pseudonymized or anonymized. When data are anonymized, the quantity and quality of extractable information decrease significantly. From the perspective of a clinical researcher, a method of achieving pseudonymized data without degrading data quality while also preventing data loss is proposed herein. As the level of pseudonymization varies according to the research purpose, the pseudonymization method applied should be carefully chosen. Therefore, the active participation of clinicians is crucial to transform the data according to the research purpose. This can contribute to data security by simply transforming the data through secondary data processing. Case studies demonstrated that, compared with the initial baseline data, there was a clinically significant difference in the number of datapoints added with the participation of a clinician (from 267,979 to 280,127 points, P < 0.001). Thus, depending on the degree of clinician participation, data anonymization may not affect data quality and quantity, and proper data quality management along with data security are emphasized. Although the pseudonymization level and clinical use of data have a trade-off relationship, it is possible to create pseudonymized data while maintaining the data quality required for a given research purpose. Therefore, rather than relying solely on security guidelines, the active participation of clinicians is important.
Keyword
Figure
Cited by 2 articles
-
A Study on Methodologies of Drug Repositioning Using Biomedical Big Data: A Focus on Diabetes Mellitus
Suehyun Lee, Seongwoo Jeon, Hun-Sung Kim
Endocrinol Metab. 2022;37(2):195-207. doi: 10.3803/EnM.2022.1404.Long-Term Changes in HbA1c According to Blood Glucose Control Status During the First 3 Months After Visiting a Tertiary University Hospital
Hyunah Kim, Da Young Jung, Seung-Hwan Lee, Jae-Hyoung Cho, Hyeon Woo Yim, Hun-Sung Kim
J Korean Med Sci. 2022;37(38):e281. doi: 10.3346/jkms.2022.37.e281.
Reference
-
1. Franklin JM, Schneeweiss S. When and how can real world data analyses substitute for randomized controlled trials? Clin Pharmacol Ther. 2017; 102(6):924–933. PMID: 28836267.
Article2. Kim HS, Kim JH. Proceed with caution when using real world data and real world evidence. J Korean Med Sci. 2019; 34(4):e28. PMID: 30686950.
Article3. Kim HS, Lee S, Kim JH. Real-world evidence versus randomized controlled trial: clinical research based on electronic medical records. J Korean Med Sci. 2018; 33(34):e213. PMID: 30127705.
Article4. Mahmoudi E, Kamdar N, Kim N, Gonzales G, Singh K, Waljee AK. Use of electronic medical records in development and validation of risk prediction models of hospital readmission: systematic review. BMJ. 2020; 369:m958. PMID: 32269037.
Article5. Kim HS, Kim H, Jeong YJ, Kim TM, Yang SJ, Baik SJ, et al. Development of clinical data mart of HMG-CoA reductase inhibitor for varied clinical research. Endocrinol Metab. 2017; 32(1):90–98.
Article6. Lee J, Kim TM, Kim H, Lee SH, Cho JH, Lee H, et al. Differences in clinical outcomes between patients with and without hypoglycemia during hospitalization: a retrospective study using real-world evidence. Diabetes Metab J. 2020; 44(4):555–565. PMID: 32431110.
Article7. Choi J, Bove LA, Tarte V, Choi WJ. Impact of simulated electronic health records on informatics competency of students in informatics course. Healthc Inform Res. 2021; 27(1):67–72. PMID: 33611878.
Article8. Shin SY. Privacy protection and data utilization. Healthc Inform Res. 2021; 27(1):1–2. PMID: 33611870.
Article9. Shin SY, Park YR, Shin Y, Choi HJ, Park J, Lyu Y, et al. A de-identification method for bilingual clinical texts of various note types. J Korean Med Sci. 2015; 30(1):7–15. PMID: 25552878.
Article10. Chevrier R, Foufi V, Gaudet-Blavignac C, Robert A, Lovis C. Use and understanding of anonymization and de-identification in the biomedical literature: scoping review. J Med Internet Res. 2019; 21(5):e13484. PMID: 31152528.
Article11. Korea Legislation Research Institute. Personal Information Protection Act. Updated 2020. Accessed Mar 1, 2021. https://elaw.klri.re.kr/eng_service/lawView.do?hseq=53044&lang=ENG .12. Ministry of Culture, Sports and Tourism (KR). Three Data Bills. Updated 2020. Accessed Mar 1, 2021. http://www.korea.kr/special/policyCurationView.do?newsId=148867915 .13. Lee D, Park M, Chang S, Ko H. Protecting and utilizing health and medical big data: policy perspectives from Korea. Healthc Inform Res. 2019; 25(4):239–247. PMID: 31777667.
Article14. Choi HJ, Lee MJ, Choi CM, Lee J, Shin SY, Lyu Y, et al. Establishing the role of honest broker: bridging the gap between protecting personal health data and clinical research efficiency. PeerJ. 2015; 3:e1506. PMID: 26713253.
Article15. Personal Information Protection Commission (KR). Pseudonymization, Combination of Pseudonymized Information. Updated 2020. Accessed Mar 1, 2021. https://www.pipc.go.kr/eng/user/lgp/bnp/pseudonymization.do .16. Ministry of Health and Welfare (KR). Establish guidelines for the use of health care data for safe use of pseudonym information in the field of health care. Updated 2020. Accessed Mar 1, 2021. http://www.mohw.go.kr/react/al/sal0301vw.jsp?PAR_MENU_ID=04&MENU_ID=0403&page=2&CONT_SEQ=360056 .17. The Hankyoreh. Civil society “The government encourages commercial use of medical information”. Updated 2020. Accessed Mar 1, 2021. http://www.hani.co.kr/arti/economy/it/963693.html .18. Kim HS, Kim DJ, Yoon KH. Medical big data is not yet available: Why we need realism rather than exaggeration. Endocrinol Metab. 2019; 34(4):349–354.
Article19. Shin SY. Issues and solutions of healthcare data de-identification: the case of South Korea. J Korean Med Sci. 2018; 33(5):e41. PMID: 29349950.
Article20. Jones W, Bruce H, Bates MJ, Belkin N, Bergman O, Marshall C. Personal information management in the present and future perfect: reports from a special NSF-sponsored workshop. Proc Am Soc Info Sci Tech. 2005; 42(1):
Article21. Waling L, Sell A. A new vision on personal information managing and sharing using instant messaging. Updated 2004. Accessed Mar 1, 2021. https://www.researchgate.net/publication/31597236_A_New_Vision_on_Personal_Information_Managing_and_Sharing_Using_Instant_Messaging .22. Mandl KD, Perakslis ED. HIPAA and the leak of “deidentified” EHR data. N Engl J Med. 2021; 384(23):2171–2173. PMID: 34110112.
Article23. Kim H, Baik SY, Yang SJ, Kim TM, Lee SH, Cho JH, et al. Clinical experiences and case review of angiotensin II receptor blocker-related angioedema in Korea. Basic Clin Pharmacol Toxicol. 2019; 124(1):115–122. PMID: 30003686.
Article24. Mehra MR, Ruschitzka F, Patel AN. Retraction-Hydroxychloroquine or chloroquine with or without a macrolide for treatment of COVID-19: a multinational registry analysis. Lancet. 2020; 395(10240):1820. PMID: 32511943.
Article25. Kim HS, Kim H, Lee H, Park B, Park S, Lee SH, et al. Analysis and comparison of statin prescription patterns and outcomes according to clinical department. J Clin Pharm Ther. 2016; 41(1):70–77. PMID: 26791968.
Article26. Huh S. Protection of personal information in medical journal publications. Neurointervention. 2019; 14(1):1–8. PMID: 30776876.
Article27. Galloway A. Estimating actual height in the older individual. J Forensic Sci. 1988; 33(1):126–136. PMID: 3351449.
Article28. Hartman T, Howell MD, Dean J, Hoory S, Slyper R, Laish I, et al. Customization scenarios for de-identification of clinical notes. BMC Med Inform Decis Mak. 2020; 20(1):14. PMID: 32000770.
Article29. Purdam K, Elliot M. A case study of the impact of statistical disclosure control on data quality in the individual UK samples of anonymised records. Environ Plan A Econ Space. 2007; 39(5):1101–1118.
Article30. Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972; 18(6):499–502. PMID: 4337382.
Article31. Nam GE, Park HS. Perspective on diagnostic criteria for obesity and abdominal obesity in Korean adults. J Obes Metab Syndr. 2018; 27(3):134–142. PMID: 31089555.
Article32. El Sanadi CE, Ji X, Kattan MW. 3-point major cardiovascular event outcome for patients with T2D treated with dipeptidyl peptidase-4 inhibitor or glucagon-like peptide-1 receptor agonist in addition to metformin monotherapy. Ann Transl Med. 2020; 8(21):1345. PMID: 33313090.
Article33. Hermans WR, Foley DP, Rensing BJ, Rutsch W, Heyndrickx GR, Danchin N, et al. Usefulness of quantitative and qualitative angiographic lesion morphology, and clinical characteristics in predicting major adverse cardiac events during and after native coronary balloon angioplasty. Am J Cardiol. 1993; 72(1):14–20. PMID: 8517422.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Safe Utilization and Sharing of Genomic Data: Amendment to the Health and Medical Data Utilization Guidelines of South Korea
- Effect of Social Support Group on Quality of Life and Burden of Primary Family Caregivers of Patients with Brain Injury
- The Outcomes of Cardiac Rehabilitation Program in the post Myocardial Infarction patient
- Research on the Quality of Life of the Parents by Functions, Activities, Participation, and Environmental Factors of Children with Cerebral Palsy Using ICF-CY Checklist
- API Driven On-Demand Participant ID Pseudonymization in Heterogeneous Multi-Study Research