Korean J Physiol Pharmacol.
2021 Nov;25(6):603-611. 10.4196/kjpp.2021.25.6.603.
Influences of ethanol and temperature on sucrose-evoked response of gustatory neurons in the hamster solitary nucleus
- Affiliations
-
- 1Department of Anatomy, School of Medicine, Southern Illinois University, Carbondale, IL 62901, USA
- 2Department of Physiology and Neuroscience, College of Dentistry and Research Institute of Oral Science, Gangneung-Wonju National University, Gangneung 25457, Korea
- KMID: 2521486
- DOI: http://doi.org/10.4196/kjpp.2021.25.6.603
Abstract
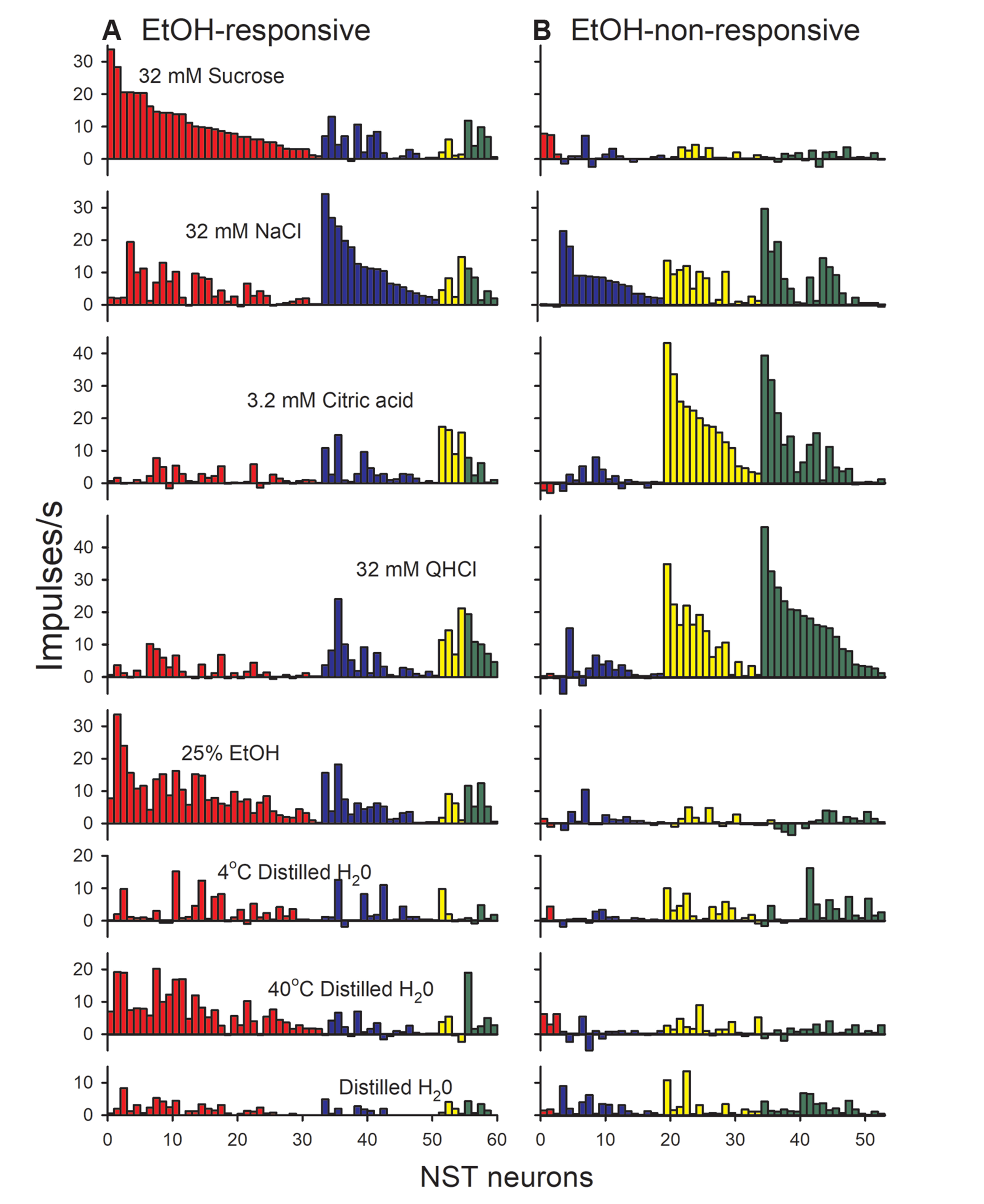
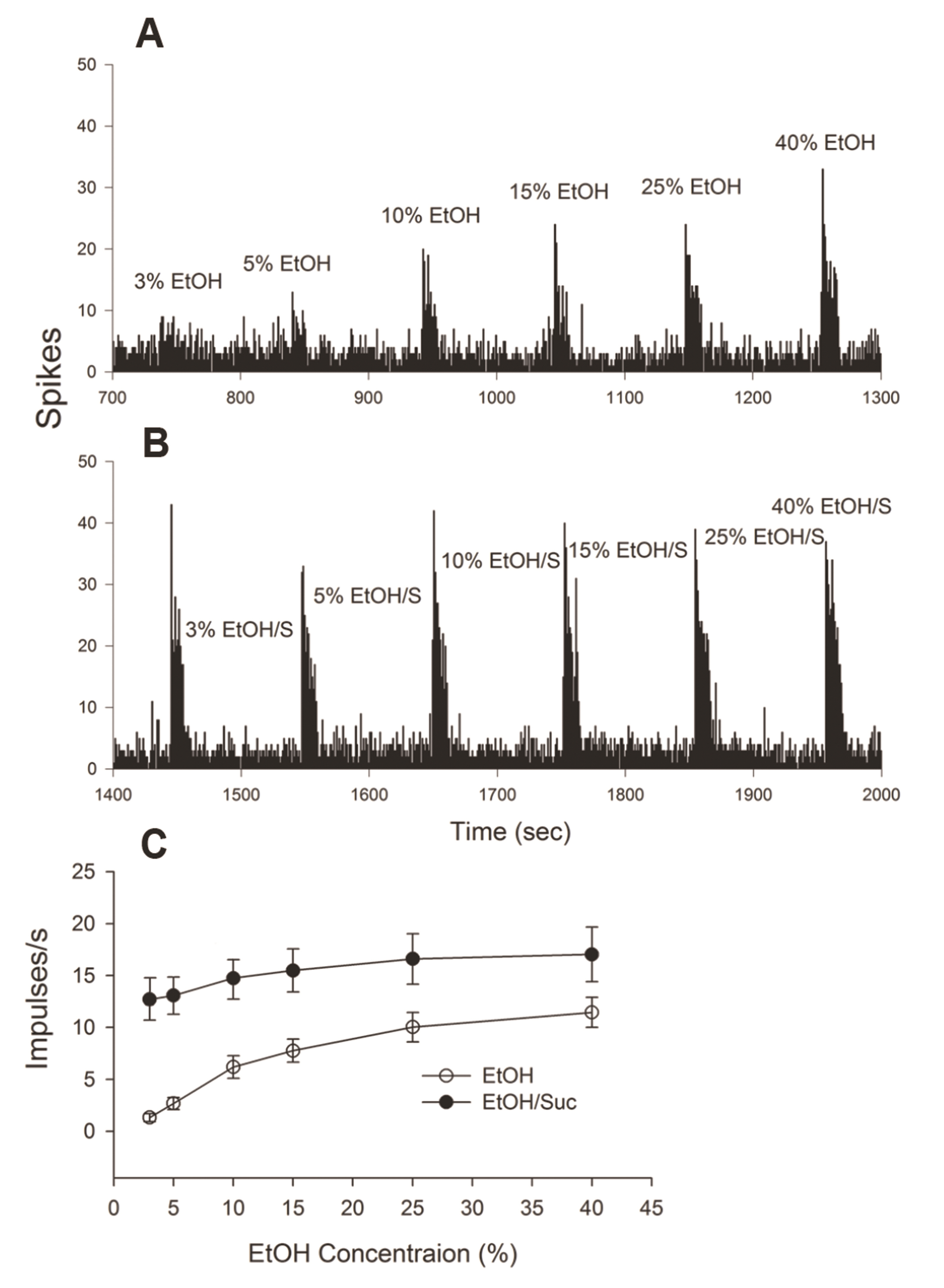
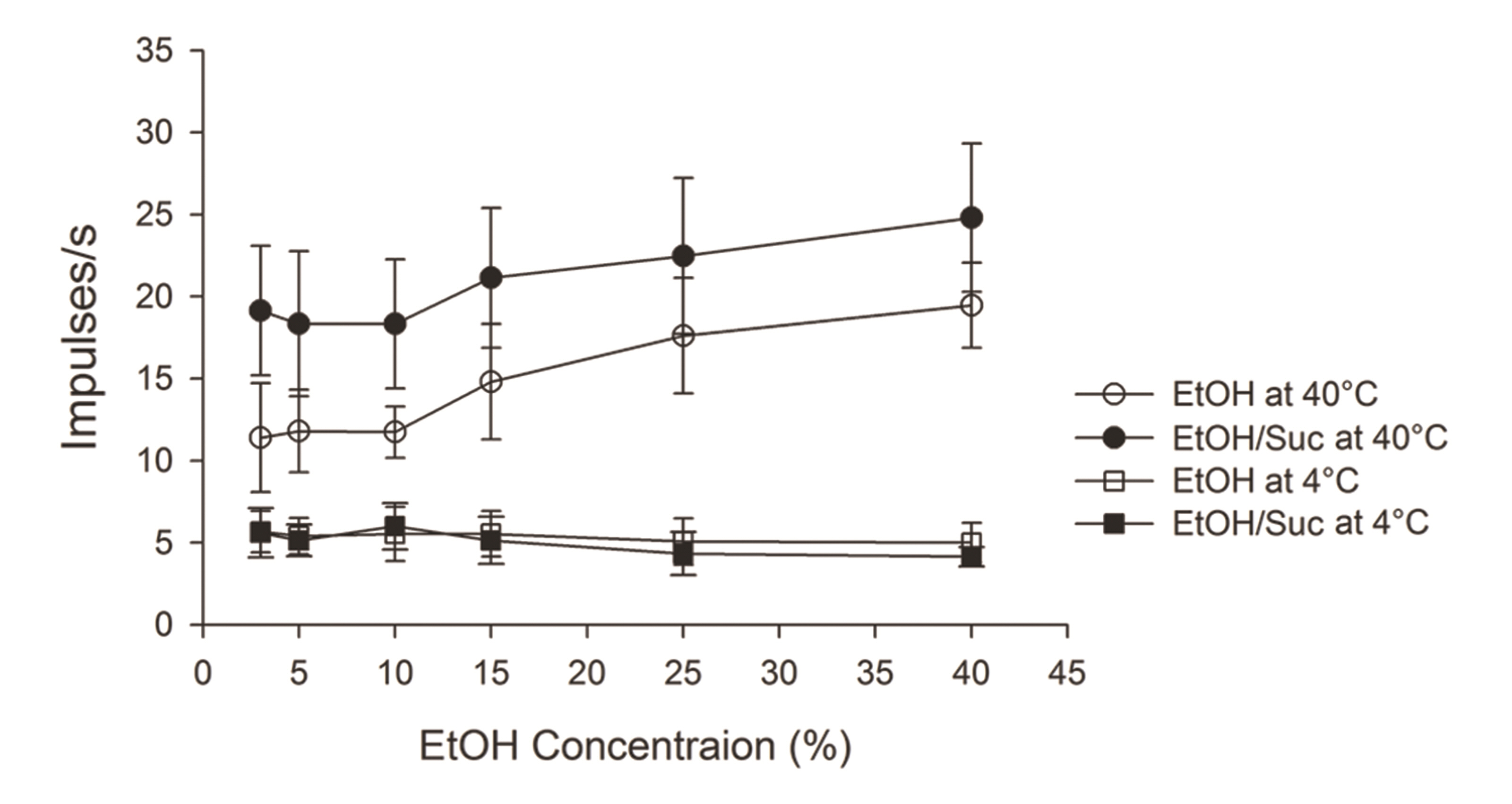
- Taste-responsive neurons in the nucleus of the solitary tract (NST), the first gustatory nucleus, often respond to thermal or mechanical stimulation. Alcohol, not a typical taste modality, is a rewarding stimulus. In this study, we aimed to investigate the effects of ethanol (EtOH) and/or temperature as stimuli to the tongue on the activity of taste-responsive neurons in hamster NST. In the first set of experiments, we recorded the activity of 113 gustatory NST neurons in urethane-anesthetized hamsters and evaluated responses to four basic taste stimuli, 25% EtOH, and 40°C and 4°C distilled water (dH2O). Sixty cells responded to 25% EtOH, with most of them also being sucrose sensitive. The response to 25% EtOH was significantly correlated with the sucrose-evoked response. A significant correlation was also observed between sucrose- and 40°C dH2O- and between 25% EtOH- and 40°C dH2O-evoked firings. In a subset of the cells, we evaluated neuronal activities in response to a series of EtOH concentrations, alone and in combination with 32 mM sucrose (EtOH/Suc) at room temperature (RT, 22°C–23°C), 40°C, and 4°C. Neuronal responses to EtOH at RT and 40°C increased as the concentrations increased. The firing rates to EtOH/Suc were greater than those to EtOH or sucrose alone. The responses were enhanced when solutions were applied at 40°C but diminished at 4°C. In summary, EtOH activates most sucrose-responsive NST gustatory cells, and the concomitant presence of sucrose or warm temperatures enhance this response. Our findings may contribute to elucidate the neural mechanisms underlying appetitive alcohol consumption.
Keyword
Figure
Reference
-
1. Lundy RF Jr. Paxinos G, editor. 2004. Gustatory system. The rat nervous system. 3rd ed. Elsevier Academic Press;Cambridge: p. 891–921. DOI: 10.1016/B978-012547638-6/50029-8.
Article2. Smith DV, Margolskee RF. 2001; Making sense of taste. Sci Am. 284:32–39. DOI: 10.1038/scientificamerican0301-32. PMID: 11234504.
Article3. Rolls ET. 2004; Smell, taste, texture, and temperature multimodal representations in the brain, and their relevance to the control of appetite. Nutr Rev. 62(11 Pt 2):S193–S204. discussion S224–S241. DOI: 10.1301/nr.2004.nov.S193-S204. PMID: 15630935.
Article4. Rogers PJ, Hardman CA. 2015; Food reward. What it is and how to measure it. Appetite. 90:1–15. DOI: 10.1016/j.appet.2015.02.032. PMID: 25728883.
Article5. Rolls ET. 1999. The brain and emotion. Oxford University Press;New York:6. Saper CB, Chou TC, Elmquist JK. 2002; The need to feed: homeostatic and hedonic control of eating. Neuron. 36:199–211. DOI: 10.1016/S0896-6273(02)00969-8. PMID: 12383777.7. Giza BK, Deems RO, Vanderweele DA, Scott TR. 1993; Pancreatic glucagon suppresses gustatory responsiveness to glucose. Am J Physiol. 265(6 Pt 2):R1231–R1237. DOI: 10.1152/ajpregu.1993.265.6.R1231. PMID: 8285262.
Article8. Giza BK, Scott TR. 1983; Blood glucose selectively affects taste-evoked activity in rat nucleus tractus solitarius. Physiol Behav. 31:643–650. PMID: 6665054.9. Giza BK, Scott TR. 1987; Intravenous insulin infusions in rats decrease gustatory-evoked responses to sugars. Am J Physiol. 252(5 Pt 2):R994–R1002. DOI: 10.1152/ajpregu.1987.252.5.R994. PMID: 3555122.
Article10. Whitehead MC. 1988; Neuronal architecture of the nucleus of the solitary tract in the hamster. J Comp Neurol. 276:547–572. DOI: 10.1002/cne.902760409. PMID: 2461969.
Article11. Chang FC, Scott TR. 1984; Conditioned taste aversions modify neural responses in the rat nucleus tractus solitarius. J Neurosci. 4:1850–1862. DOI: 10.1523/JNEUROSCI.04-07-01850.1984. PMID: 6737042. PMCID: PMC6564891.
Article12. Giza BK, Ackroff K, McCaughey SA, Sclafani A, Scott TR. 1997; Preference conditioning alters taste responses in the nucleus of the solitary tract of the rat. Am J Physiol. 273:R1230–R1240. DOI: 10.1152/ajpregu.1997.273.4.R1230. PMID: 9362285.13. Brasser SM, Castro N, Feretic B. 2015; Alcohol sensory processing and its relevance for ingestion. Physiol Behav. 148:65–70. DOI: 10.1016/j.physbeh.2014.09.004. PMID: 25304192. PMCID: PMC4388769.
Article14. Bachmanov AA, Kiefer SW, Molina JC, Tordoff MG, Duffy VB, Bartoshuk LM, Mennella JA. 2003; Chemosensory factors influencing alcohol perception, preferences, and consumption. Alcohol Clin Exp Res. 27:220–231. DOI: 10.1097/01.ALC.0000051021.99641.19. PMID: 12605071. PMCID: PMC1940064.
Article15. Mattes RD, DiMeglio D. 2001; Ethanol perception and ingestion. Physiol Behav. 72:217–229. DOI: 10.1016/S0031-9384(00)00397-8. PMID: 11240000.
Article16. Kiefer SW, Morrow NS, Metzler CW. 1988; Alcohol aversion generalization in rats: specific disruption of taste and odor cues with gustatory neocortex or olfactory bulb ablations. Behav Neurosci. 102:733–739. DOI: 10.1037/0735-7044.102.5.733. PMID: 2848538.
Article17. Kampov-Polevoy AB, Garbutt JC, Janowsky DS. 1999; Association between preference for sweets and excessive alcohol intake: a review of animal and human studies. Alcohol Alcohol. 34:386–395. DOI: 10.1093/alcalc/34.3.386. PMID: 10414615.
Article18. Hellekant G, Danilova V, Roberts T, Ninomiya Y. 1997; The taste of ethanol in a primate model: I. Chorda tympani nerve response in Macaca mulatta. Alcohol. 14:473–484. DOI: 10.1016/S0741-8329(96)00215-7. PMID: 9305463.
Article19. Sako N, Yamamoto T. 1999; Electrophysiological and behavioral studies on taste effectiveness of alcohols in rats. Am J Physiol. 276:R388–R396. DOI: 10.1152/ajpregu.1999.276.2.R388. PMID: 9950916.
Article20. Lemon CH, Brasser SM, Smith DV. 2004; Alcohol activates a sucrose-responsive gustatory neural pathway. J Neurophysiol. 92:536–544. DOI: 10.1152/jn.00097.2004. PMID: 14985409.
Article21. Lemon CH, Wilson DM, Brasser SM. 2011; Differential neural representation of oral ethanol by central taste-sensitive neurons in ethanol-preferring and genetically heterogeneous rats. J Neurophysiol. 106:3145–3156. DOI: 10.1152/jn.00580.2011. PMID: 21918002. PMCID: PMC3234093.
Article22. Cho YK. 2007; Relationship between alcohol and sucrose/thermal stimulation in pontine taste neurons in the hamster. Exp Neurobiol. 16:79–87.23. Cruz A, Green BG. 2000; Thermal stimulation of taste. Nature. 403:889–892. DOI: 10.1038/35002581. PMID: 10706285.
Article24. Bartoshuk LM, Rennert K, Rodin J, Stevens JC. 1982; Effects of temperature on the perceived sweetness of sucrose. Physiol Behav. 28:905–910. DOI: 10.1016/0031-9384(82)90212-8. PMID: 7100291.
Article25. Green BG, Frankmann SP. 1988; The effect of cooling on the perception of carbohydrate and intensive sweeteners. Physiol Behav. 43:515–519. DOI: 10.1016/0031-9384(88)90127-8. PMID: 3194473.
Article26. Cho YK, Li CS, Smith DV. 2002; Gustatory projections from the nucleus of the solitary tract to the parabrachial nuclei in the hamster. Chem Senses. 27:81–90. DOI: 10.1093/chemse/27.1.81. PMID: 11751472.
Article27. Cho YK, Li CS, Smith DV. 2002; Taste responses of neurons of the hamster solitary nucleus are enhanced by lateral hypothalamic stimulation. J Neurophysiol. 87:1981–1992. DOI: 10.1152/jn.00765.2001. PMID: 11929917.
Article28. Li CS, Lu DP, Cho YK. 2015; Descending projections from the nucleus accumbens shell excite activity of taste-responsive neurons in the nucleus of the solitary tract in the hamster. J Neurophysiol. 113:3778–3786. DOI: 10.1152/jn.00362.2014. PMID: 25744880. PMCID: PMC4468968.
Article29. Duncan HJ, Smith DV. 1992; Concentration-response functions for thirty chemical stimuli in the hamster solitary nucleus. Chem Senses. 17:616.30. Smith DV, Travers JB. 1979; A metric for the breadth of tuning of gustatory neurons. Chem Senses. 4:215–229. DOI: 10.1093/chemse/4.3.215.
Article31. McPheeters M, Hettinger TP, Nuding SC, Savoy LD, Whitehead MC, Frank ME. 1990; Taste-responsive neurons and their locations in the solitary nucleus of the hamster. Neuroscience. 34:745–758. DOI: 10.1016/0306-4522(90)90179-8. PMID: 2352650.
Article32. Di Lorenzo PM, Kiefer SW, Rice AG, Garcia J. 1986; Neural and behavioral responsivity to ethyl alcohol as a tastant. Alcohol. 3:55–61. DOI: 10.1016/0741-8329(86)90071-6. PMID: 3964438.
Article33. Danilova V, Hellekant G. 2000; The taste of ethanol in a primate model. II. Glossopharyngeal nerve response in Macaca mulatta. Alcohol. 21:259–269. DOI: 10.1016/S0741-8329(00)00094-X. PMID: 11091030.34. Hellekant G. 1965; Electrophysiological investigation of the gustatory effect of ethyl alcohol. II. A single fibre analysis in the cat. Acta Physiol Scand. 64:398–406. DOI: 10.1111/j.1748-1716.1965.tb04197.x. PMID: 5853033.
Article35. Lemon CH, Imoto T, Smith DV. 2003; Differential gurmarin suppression of sweet taste responses in rat solitary nucleus neurons. J Neurophysiol. 90:911–923. DOI: 10.1152/jn.00215.2003. PMID: 12702710.36. Kampov-Polevoy AB, Kasheffskaya OP, Sinclair JD. 1990; Initial acceptance of ethanol: gustatory factors and patterns of alcohol drinking. Alcohol. 7:83–85. DOI: 10.1016/0741-8329(90)90065-K. PMID: 2328091.
Article37. Kampov-Polevoy AB, Overstreet DH, Rezvani AH, Janowsky DS. 1995; Saccharin-induced increase in daily fluid intake as a predictor of voluntary alcohol intake in alcohol-preferring rats. Physiol Behav. 57:791–795. DOI: 10.1016/0031-9384(94)00389-0. PMID: 7777619.
Article38. Berridge KC. 2003; Pleasures of the brain. Brain Cogn. 52:106–128. DOI: 10.1016/S0278-2626(03)00014-9. PMID: 12812810.
Article39. Levine AS, Kotz CM, Gosnell BA. 2003; Sugars: hedonic aspects, neuroregulation, and energy balance. Am J Clin Nutr. 78:834S–842S. DOI: 10.1093/ajcn/78.4.834S. PMID: 14522747.
Article40. Hajnal A, Smith GP, Norgren R. 2004; Oral sucrose stimulation increases accumbens dopamine in the rat. Am J Physiol Regul Integr Comp Physiol. 286:R31–R37. DOI: 10.1152/ajpregu.00282.2003. PMID: 12933362.
Article41. Norgren R, Hajnal A, Mungarndee SS. 2006; Gustatory reward and the nucleus accumbens. Physiol Behav. 89:531–535. DOI: 10.1016/j.physbeh.2006.05.024. PMID: 16822531. PMCID: PMC3114426.
Article42. Smith GP. 2004; Accumbens dopamine mediates the rewarding effect of orosensory stimulation by sucrose. Appetite. 43:11–13. DOI: 10.1016/j.appet.2004.02.006. PMID: 15262012.
Article43. Weiss F, Lorang MT, Bloom FE, Koob GF. 1993; Oral alcohol self-administration stimulates dopamine release in the rat nucleus accumbens: genetic and motivational determinants. J Pharmacol Exp Ther. 267:250–258. PMID: 8229752.44. Zhang M, Kelley AE. 2002; Intake of saccharin, salt, and ethanol solutions is increased by infusion of a mu opioid agonist into the nucleus accumbens. Psychopharmacology (Berl). 159:415–423. DOI: 10.1007/s00213-001-0932-y. PMID: 11823894.
Article45. Loriaux AL, Roitman JD, Roitman MF. 2011; Nucleus accumbens shell, but not core, tracks motivational value of salt. J Neurophysiol. 106:1537–1544. DOI: 10.1152/jn.00153.2011. PMID: 21697439. PMCID: PMC3174809.
Article46. Li CS, Chung S, Lu DP, Cho YK. 2012; Descending projections from the nucleus accumbens shell suppress activity of taste-responsive neurons in the hamster parabrachial nuclei. J Neurophysiol. 108:1288–1298. DOI: 10.1152/jn.00121.2012. PMID: 22696536.
Article47. Hayama T, Ito S, Ogawa H. 1985; Responses of solitary tract nucleus neurons to taste and mechanical stimulations of the oral cavity in decerebrate rats. Exp Brain Res. 60:235–242. DOI: 10.1007/BF00235918. PMID: 4054268.
Article48. Ogawa H, Hayama T, Yamashita Y. 1988; Thermal sensitivity of neurons in a rostral part of the rat solitary tract nucleus. Brain Res. 454:321–331. DOI: 10.1016/0006-8993(88)90833-5. PMID: 3409015.
Article49. Ogawa H, Imoto T, Hayama T. 1984; Responsiveness of solitario-parabrachial relay neurons to taste and mechanical stimulation applied to the oral cavity in rats. Exp Brain Res. 54:349–358. DOI: 10.1007/BF00236236. PMID: 6723854.
Article50. Travers SP, Norgren R. 1995; Organization of orosensory responses in the nucleus of the solitary tract of rat. J Neurophysiol. 73:2144–2162. DOI: 10.1152/jn.1995.73.6.2144. PMID: 7666129.
Article51. Green BG, George P. 2004; 'Thermal taste' predicts higher responsiveness to chemical taste and flavor. Chem Senses. 2:617–628. DOI: 10.1093/chemse/bjh065. PMID: 15337686.
Article52. Ogawa H, Sato M, Yamashita S. 1968; Multiple sensitivity of chordat typani fibres of the rat and hamster to gustatory and thermal stimuli. J Physiol. 199:223–240. DOI: 10.1113/jphysiol.1968.sp008650. PMID: 5684036. PMCID: PMC1365355.53. Lundy RF Jr, Contreras RJ. 1997; Temperature and amiloride alter taste nerve responses to Na+, K+, and NH4+salts in rats. Brain Res. 744:309–317. DOI: 10.1016/S0006-8993(96)01118-3.54. Li J, Lemon CH. 2015; Influence of stimulus and oral adaptation temperature on gustatory responses in central taste-sensitive neurons. J Neurophysiol. 113:2700–2712. DOI: 10.1152/jn.00736.2014. PMID: 25673737. PMCID: PMC4416558.
Article55. Wilson DM, Lemon CH. 2014; Temperature systematically modifies neural activity for sweet taste. J Neurophysiol. 112:1667–1677. DOI: 10.1152/jn.00368.2014. PMID: 24966301. PMCID: PMC4157175.
Article56. Breza JM, Curtis KS, Contreras RJ. 2006; Temperature modulates taste responsiveness and stimulates gustatory neurons in the rat geniculate ganglion. J Neurophysiol. 95:674–685. DOI: 10.1152/jn.00793.2005. PMID: 16267112.
Article57. Cho YK, Smith ME, Norgren R. 2004; Low-dose furosemide modulates taste responses in the nucleus of the solitary tract of the rat. Am J Physiol Regul Integr Comp Physiol. 287:R706–R714. DOI: 10.1152/ajpregu.00090.2004. PMID: 15155275.
Article58. Cho YK, Mao L, Li CS. 2008; Modulation of solitary taste neurons by electrical stimulation of the ventroposteromedial nucleus of the thalamus in the hamster. Brain Res. 1221:67–79. DOI: 10.1016/j.brainres.2008.05.006. PMID: 18565498.
Article59. Li CS, Cho YK, Smith DV. 2002; Taste responses of neurons in the hamster solitary nucleus are modulated by the central nucleus of the amygdala. J Neurophysiol. 88:2979–2992. DOI: 10.1152/jn.00239.2002. PMID: 12466423.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Centrifugal Influence on Gustatory Neurons in the Nucleus of the Solitary Tract
- Comparison of sucrose and ethanol-induced c-Fos-like immunoreactivity in the parabrachial nuclei and accumbens nucleus
- Developmental changes of gustatory neurons in nucleus of solitary tract in rats
- c-Fos-like Immunoreactivity in the nucleus of the solitary tract following taste stimulation of the contralateral side of the tongue
- Immunohistochemical study on the distribution of serotonergic neurons in the raphe nucleus of the Hamster brainstem