Neurointervention.
2021 Mar;16(1):20-28. 10.5469/neuroint.2020.00199.
A Multicenter Survey of Acute Stroke Imaging Protocols for Endovascular Thrombectomy
- Affiliations
-
- 1Department of Radiology, Anam Hospital, Korea University College of Medicine, Seoul, Korea
- 2Department of Radiology and Research Institute of Radiology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- KMID: 2513339
- DOI: http://doi.org/10.5469/neuroint.2020.00199
Abstract
- Purpose
Identifying current practices in acute stroke imaging is essential for establishing optimal imaging protocols. We surveyed and assessed the current status of acute stroke imaging for endovascular thrombectomy (EVT) at tertiary hospitals throughout South Korea.
Materials and Methods
An electronic questionnaire on imaging protocols for EVT in patients with acute ischemic stroke was e-mailed to physicians at 42 registered tertiary hospitals, and their responses were collected between February and March 2020.
Results
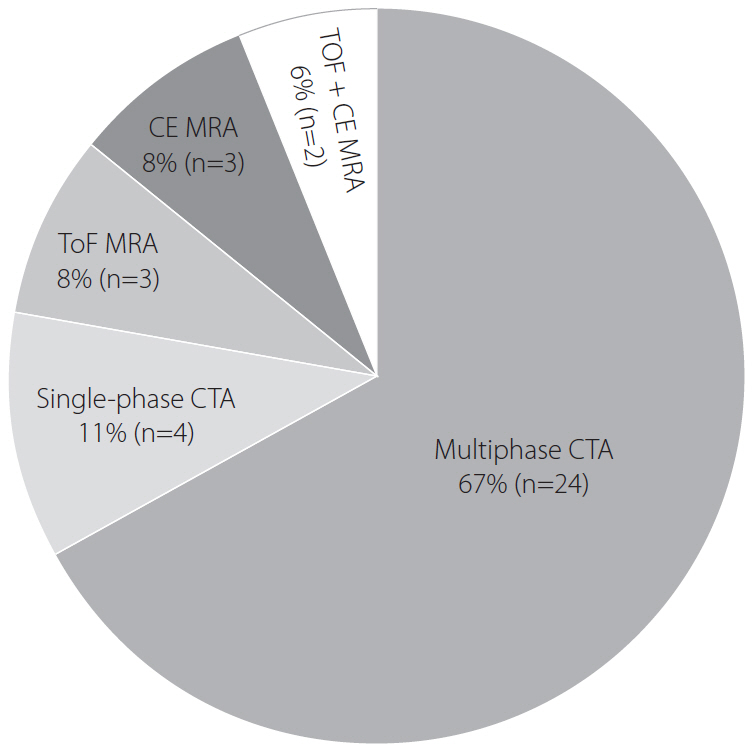
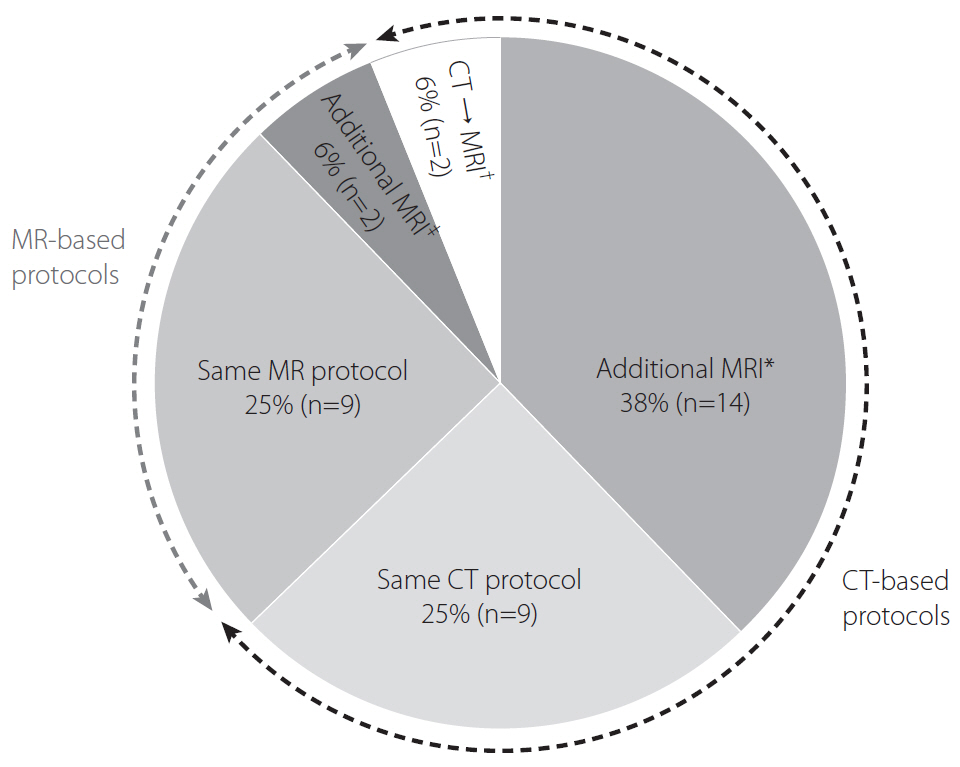
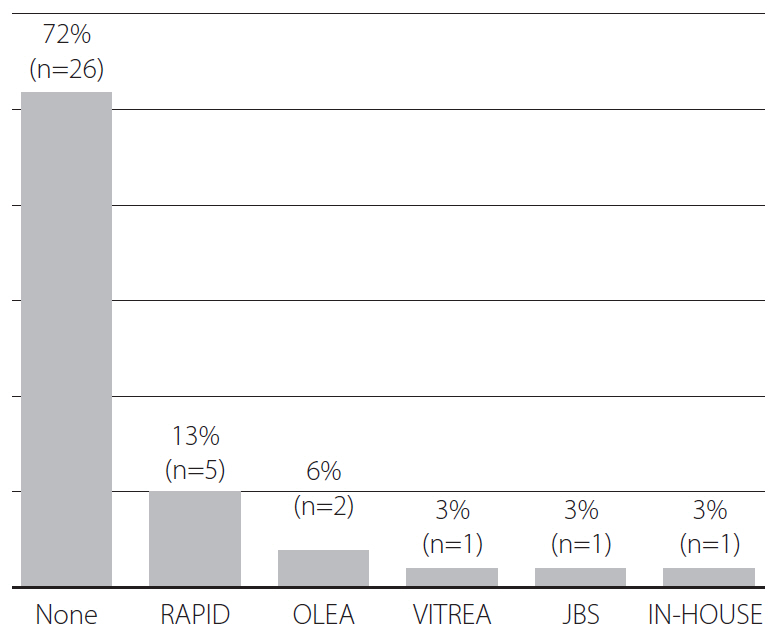
Of the 36 hospitals participating in the survey, 69% (25/36) adopted computed tomography (CT)-based protocols, whereas 31% (11/36) adopted magnetic resonance (MR)-based protocols. Non-enhanced CT (NECT) was the initial imaging study at 28%, NECT with CT angiography (CTA) at 36%, and NECT with CTA and CT perfusion (CTP) at 33% of hospitals. Perfusion imaging was performed at 61% (22/36), CTP at 44% (16/36), and MR perfusion at 17% (6/36) of hospitals. Multiphase CTA was performed at 67%, single-phase CTA at 11%, time-of-flight MR angiography (MRA) at 8%, contrast-enhanced MRA at 8%, and both at 6% of hospitals. For late time window stroke, 50% of hospitals used identical imaging protocols to those for early time window stroke, 39% used additional MR imaging (MRI), and 6% converted the imaging strategy from CT to MRI. Post-processing programs were used at 28% (10/36), and RAPID software at 14% (5/36) of hospitals, respectively. Most hospitals (92%) used the same imaging protocols for posterior and anterior circulation strokes.
Conclusion
Our multicenter survey demonstrated considerable heterogeneity in acute stroke imaging protocols across South Korean tertiary hospitals, suggesting that hospitals refine their imaging protocols according to hospital-specific conditions.
Keyword
Figure
Cited by 1 articles
-
Proposed Protocols for Artificial Intelligence Imaging Database in Acute Stroke Imaging
Minjae Kim, Seung Chai Jung, Soo Chin Kim, Bum Joon Kim, Woo-Keun Seo, Byungjun Kim
Neurointervention. 2023;18(3):149-158. doi: 10.5469/neuroint.2023.00339.
Reference
-
1. Aghaebrahim A, Streib C, Rangaraju S, Kenmuir CL, Giurgiutiu DV, Horev A, et al. Streamlining door to recanalization processes in endovascular stroke therapy. J Neurointerv Surg. 2017; 9:340–345.
Article2. Menon BK, Sajobi TT, Zhang Y, Rempel JL, Shuaib A, Thornton J, et al. Analysis of workflow and time to treatment on thrombectomy outcome in the endovascular treatment for small core and proximal occlusion ischemic stroke (ESCAPE) randomized, controlled trial. Circulation. 2016; 133:2279–2286.
Article3. Goyal M, Jadhav AP, Bonafe A, Diener H, Mendes Pereira V, Levy E, SWIFT PRIME investigators, et al. Analysis of workflow and time to treatment and the effects on outcome in endovascular treatment of acute ischemic stroke: results from the SWIFT PRIME randomized controlled trial. Radiology. 2016; 279:888–897.
Article4. Menon BK, Goyal M. Imaging paradigms in acute ischemic stroke: a pragmatic evidence-based approach. Radiology. 2015; 277:7–12.
Article5. Saver JL, Goyal M, Bonafe A, Diener HC, Levy EI, Pereira VM, SWIFT PRIME Investigators, et al. Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N Engl J Med. 2015; 372:2285–2295.
Article6. Jovin TG, Chamorro A, Cobo E, de Miquel MA, Molina CA, Rovira A, REVASCAT Trial Investigators, et al. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N Engl J Med. 2015; 372:2296–2306.
Article7. Goyal M, Demchuk AM, Menon BK, Eesa M, Rempel JL, Thornton J, ESCAPE Trial Investigators, et al. Randomized assessment of rapid endovascular treatment of ischemic stroke. N Engl J Med. 2015; 372:1019–1030.8. Campbell BC, Mitchell PJ, Kleinig TJ, Dewey HM, Churilov L, Yassi N, EXTEND-IA Investigators, et al. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med. 2015; 372:1009–1018.
Article9. Berkhemer OA, Fransen PS, Beumer D, van den Berg LA, Lingsma HF, Yoo AJ, MR CLEAN Investigators, et al. A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med. 2015; 372:11–20.10. Nogueira RG, Jadhav AP, Haussen DC, Bonafe A, Budzik RF, Bhuva P, DAWN Trial Investigators, et al. Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct. N Engl J Med. 2018; 378:11–21.11. Albers GW, Marks MP, Kemp S, Christensen S, Tsai JP, Ortega-Gutierrez S, DEFUSE 3 Investigators, et al. Thrombectomy for stroke at 6 to 16 hours with selection by perfusion imaging. N Engl J Med. 2018; 378:708–718.
Article12. Warach SJ, Luby M, Albers GW, Bammer R, Bivard A, Campbell BC, Stroke Imaging Research (STIR) and VISTA-Imaging Investigators, et al. Acute stroke imaging research roadmap III imaging selection and outcomes in acute stroke reperfusion clinical trials: consensus recommendations and further research priorities. Stroke. 2016; 47:1389–1398.13. Wintermark M, Luby M, Bornstein NM, Demchuk A, Fiehler J, Kudo K, et al. International survey of acute stroke imaging used to make revascularization treatment decisions. Int J Stroke. 2015; 10:759–762.
Article14. Warach S, Gaa J, Siewert B, Wielopolski P, Edelman RR. Acute human stroke studied by whole brain echo planar diffusion-weighted magnetic resonance imaging. Ann Neurol. 1995; 37:231–241.
Article15. Provost C, Soudant M, Legrand L, Ben Hassen W, Xie Y, Soize S, et al. Magnetic resonance imaging or computed tomography before treatment in acute ischemic stroke. Stroke. 2019; 50:659–664.
Article16. Goyal M, Menon BK, Hill MD, Demchuk A. Consistently achieving computed tomography to endovascular recanalization <90 minutes: solutions and innovations. Stroke. 2014; 45:e252–e256.
Article17. Rocha M, Jovin TG. Fast versus slow progressors of infarct growth in large vessel occlusion stroke: clinical and research implications. Stroke. 2017; 48:2621–2627.18. Ko SB, Park HK, Kim BM, Heo JH, Rha JH, Kwon SU, et al. 2019 update of the Korean clinical practice guidelines of stroke for endovascular recanalization therapy in patients with acute ischemic stroke. Neurointervention. 2019; 14:71–81.
Article19. Seo KD, Suh SH. Endovascular treatment in acute ischemic stroke: a nationwide survey in Korea. Neurointervention. 2018; 13:84–89.
Article20. Vergouwen MD, Algra A, Pfefferkorn T, Weimar C, Rueckert CM, Thijs V, Basilar Artery International Cooperation Study (BASICS) Study Group, et al. Time is brain(stem) in basilar artery occlusion. Stroke. 2012; 43:3003–3006.
Article21. Guillaume M, Lapergue B, Gory B, Labreuche J, Consoli A, Mione G, Endovascular Treatment in Ischemic Stroke (ETIS) Investigators, et al. Rapid successful reperfusion of basilar artery occlusion strokes with pretreatment diffusion-weighted imaging posterior-circulation aspects <8 is associated with good outcome. J Am Heart Assoc. 2019; 8:e010962.
Article22. Goyal N, Tsivgoulis G, Nickele C, Doss VT, Hoit D, Alexandrov AV, et al. Posterior circulation CT angiography collaterals predict outcome of endovascular acute ischemic stroke therapy for basilar artery occlusion. J Neurointerv Surg. 2016; 8:783–786.
Article23. McTaggart RA, Ansari SA, Goyal M, Abruzzo TA, Albani B, Arthur AJ, Standards and Guidelines Committee of the Society of NeuroInterventional Surgery (SNIS), et al. Initial hospital management of patients with emergent large vessel occlusion (ELVO): report of the standards and guidelines committee of the Society of NeuroInterventional Surgery. J Neurointerv Surg. 2017; 9:316–323.
Article24. Demeestere J, Wouters A, Christensen S, Lemmens R, Lansberg MG. Review of perfusion imaging in acute ischemic stroke: from time to tissue. Stroke. 2020; 51:1017–1024.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Choosing a Hyperacute Stroke Imaging Protocol for Proper Patient Selection and Time Efficient Endovascular Treatment: Lessons from Recent Trials
- Endovascular Treatment of Acute Ischemic Stroke
- Intravenous Thrombolysis and Endovascular Thrombectomy in Acute Ischemic Stroke with Minor Symptom
- Differences in Effectiveness among Devices for Endovascular Thrombectomy in Patients with Acute Ischemic Stroke
- Endovascular Mechanical Thrombectomy for Acute Ischemic Stroke: A New Standard of Care