J Korean Med Sci.
2020 Nov;35(45):e370. 10.3346/jkms.2020.35.e370.
Growth and Bone Mineral Density Changes in Ovariectomized Rats Treated with Estrogen Receptor Alpha or Beta Agonists
- Affiliations
-
- 1Department of Pediatrics, Kyung Hee University Hospital at Gangdong, Kyung Hee University School of Medicine, Seoul, Korea
- 2Department of Nuclear Medicine, Kyung Hee University Hospital at Gangdong, Kyung Hee University School of Medicine, Seoul, Korea
- 3Department of Pathology, Kyung Hee University Hospital at Gangdong, Kyung Hee University School of Medicine, Seoul, Korea
- KMID: 2508801
- DOI: http://doi.org/10.3346/jkms.2020.35.e370
Abstract
- Background
Estrogen controls the pubertal growth spurt, growth plate closure, and accretion of bone mineral density (BMD) of long bones after biding estrogen receptor (ER). There are two subtypes of ER, ERα and ERβ. If each ER subtype has different effects, we may control those actions by manipulating the estrogen binding intensity to each ER subtype and increase the final adult height without markedly reducing BMD or impairing reproductive functions. The purpose of our study was to compare these effects of ERα and ERβ on long bones in ovariectomized rats.
Methods
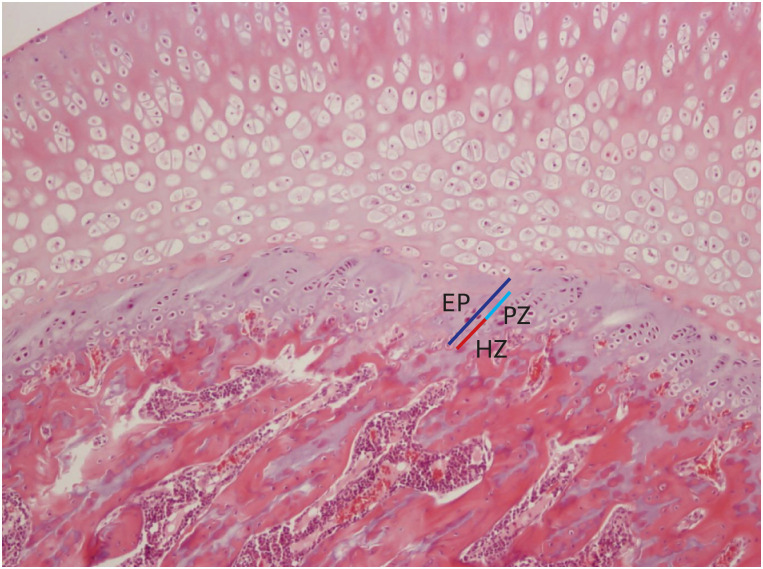
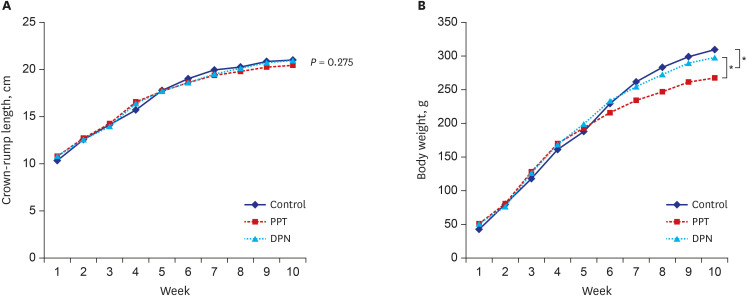
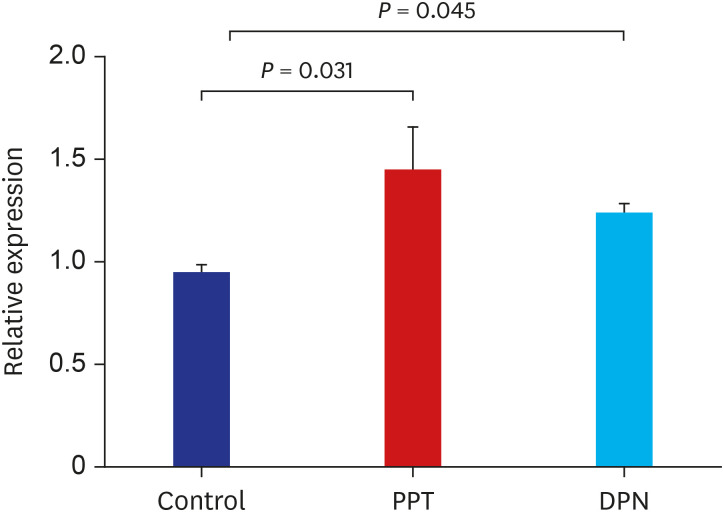
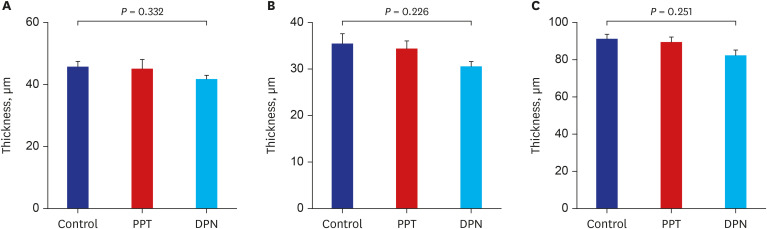
Thirty female rats were ovariectomized and randomly divided into 3 groups. The control, propylpyrazole triol (PPT), and 2,3-bis (4-hydroxyphenyl) propionitrile (DPN) groups were subcutaneously injected for 5 weeks with sesame oil, PPT as an ERα agonist, and DPN as an ERβ agonist, respectively. The crown-lump length and body weight were measured weekly. BMD, serum levels of growth hormone (GH) and estradiol were checked before and after 5 weeks of injections. Pituitary GH1 expression levels were determined with quantitative realtime polymerase chain reaction, the proximal tibias were dissected, decalcified and stained with hematoxylin-eosin, and the thicknesses of epiphyseal plates including proliferative and hypertrophic zones were measured in 20-evenly divided sites after 5 weeks of injections. Comparisons for auxological data, serum hormone and pituitary GH1 expression levels, BMD, and epiphyseal plate thicknesses among 3 groups before and after injections were conducted.
Results
There was no significant difference in body lengths among 3 groups. The body weights were significantly lower, but, serum GH, pituitary GH1 expression levels, and BMDs were higher in PPT group than the other 2 groups after 5 weeks of injections. There was no significant difference in the thicknesses of the total epiphyseal plate, proliferative, and hypertrophic zone among 3 groups.
Conclusion
ERα is more involved in pituitary GH secretion and bone mineral deposition than ERβ. Weight gain might be prevented with the ERα agonist.
Figure
Reference
-
1. Styne DM, Grumbach MM. Chapter 24. Puberty: ontogeny, neuroendocrinology, physiology, and disorders. In : Kronenberg HM, Melmed S, Polonsky KS, Larsen PR, editors. Williams Textbook of Endocrinology. 11th ed. Philadelphia, PA: Saunders Co.;2008. p. 969–1166.2. Krum SA. Direct transcriptional targets of sex steroid hormones in bone. J Cell Biochem. 2011; 112(2):401–408. PMID: 21268060.
Article3. Emons J, Chagin AS, Sävendahl L, Karperien M, Wit JM. Mechanisms of growth plate maturation and epiphyseal fusion. Horm Res Paediatr. 2011; 75(6):383–391. PMID: 21540578.
Article4. Nilsson O, Chrysis D, Pajulo O, Boman A, Holst M, Rubinstein J, et al. Localization of estrogen receptors-alpha and -beta and androgen receptor in the human growth plate at different pubertal stages. J Endocrinol. 2003; 177(2):319–326. PMID: 12740020.
Article5. Stavrou I, Zois C, Chatzikyriakidou A, Georgiou I, Tsatsoulis A. Combined estrogen receptor α and estrogen receptor β genotypes influence the age of menarche. Hum Reprod. 2006; 21(2):554–557. PMID: 16210384.
Article6. van der Eerden BC, Karperien M, Wit JM. Systemic and local regulation of the growth plate. Endocr Rev. 2003; 24(6):782–801. PMID: 14671005.
Article7. Carel JC, Léger J. Clinical practice. Precocious puberty. N Engl J Med. 2008; 358(22):2366–2377. PMID: 18509122.8. Image J. Contributors. Accessed April 15, 2014. http://imagej.nih.gov/ij/download.html.9. Shim KS. The growth and pubertal development in female mice with tissue-specific knock out of estrogen receptor. J Korean Soc Pediatr Endocrinol. 2011; 16(2):67–72.
Article10. MacGillivray MH, Morishima A, Conte F, Grumbach M, Smith EP. Pediatric endocrinology update: an overview. The essential roles of estrogens in pubertal growth, epiphyseal fusion and bone turnover: lessons from mutations in the genes for aromatase and the estrogen receptor. Horm Res. 1998; 49 Suppl 1:2–8. PMID: 9554463.11. Kronenberg HM. Developmental regulation of the growth plate. Nature. 2003; 423(6937):332–336. PMID: 12748651.
Article12. Hiney JK, Ojeda SR, Dees WL. Insulin-like growth factor I: a possible metabolic signal involved in the regulation of female puberty. Neuroendocrinology. 1991; 54(4):420–423. PMID: 1758585.
Article13. Juul A. The effects of oestrogens on linear bone growth. Hum Reprod Update. 2001; 7(3):303–313. PMID: 11392377.
Article14. Ohlsson C, Mohan S, Sjögren K, Tivesten A, Isgaard J, Isaksson O, et al. The role of liver-derived insulin-like growth factor-I. Endocr Rev. 2009; 30(5):494–535. PMID: 19589948.
Article15. Baron J, Klein KO, Yanovski JA, Novosad JA, Bacher JD, Bolander ME, et al. Induction of growth plate cartilage ossification by basic fibroblast growth factor. Endocrinology. 1994; 135(6):2790–2793. PMID: 7988472.
Article16. Bord S, Horner A, Beavan S, Compston J. Estrogen receptors α and β are differentially expressed in developing human bone. J Clin Endocrinol Metab. 2001; 86(5):2309–2314. PMID: 11344243.
Article17. Ohlsson C, Engdahl C, Börjesson AE, Windahl SH, Studer E, Westberg L, et al. Estrogen receptor-α expression in neuronal cells affects bone mass. Proc Natl Acad Sci U S A. 2012; 109(3):983–988. PMID: 22215598.
Article18. Börjesson AE, Lagerquist MK, Liu C, Shao R, Windahl SH, Karlsson C, et al. The role of estrogen receptor α in growth plate cartilage for longitudinal bone growth. J Bone Miner Res. 2010; 25(12):2690–2700. PMID: 20564247.
Article19. Weise M, De-Levi S, Barnes KM, Gafni RI, Abad V, Baron J. Effects of estrogen on growth plate senescence and epiphyseal fusion. Proc Natl Acad Sci U S A. 2001; 98(12):6871–6876. PMID: 11381135.
Article20. Zhao C, Dahlman-Wright K, Gustafsson JA. Estrogen receptor β: an overview and update. Nucl Recept Signal. 2008; 6(1):e003. PMID: 18301783.
Article21. Börjesson AE, Lagerquist MK, Windahl SH, Ohlsson C. The role of estrogen receptor α in the regulation of bone and growth plate cartilage. Cell Mol Life Sci. 2013; 70(21):4023–4037. PMID: 23516016.
Article22. Chagin AS, Sävendahl L. Oestrogen receptors and linear bone growth. Acta Paediatr. 2007; 96(9):1275–1279. PMID: 17718780.
Article23. Stauffer SR, Coletta CJ, Tedesco R, Nishiguchi G, Carlson K, Sun J, et al. Pyrazole ligands: structure-affinity/activity relationships and estrogen receptor-α-selective agonists. J Med Chem. 2000; 43(26):4934–4947. PMID: 11150164.
Article24. Kraichely DM, Sun J, Katzenellenbogen JA, Katzenellenbogen BS. Conformational changes and coactivator recruitment by novel ligands for estrogen receptor-α and estrogen receptor-β: correlations with biological character and distinct differences among SRC coactivator family members. Endocrinology. 2000; 141(10):3534–3545. PMID: 11014206.
Article25. Frasor J, Barnett DH, Danes JM, Hess R, Parlow AF, Katzenellenbogen BS. Response-specific and ligand dose-dependent modulation of estrogen receptor (ER) α activity by ERβ in the uterus. Endocrinology. 2003; 144(7):3159–3166. PMID: 12810572.
Article26. Meyers MJ, Sun J, Carlson KE, Marriner GA, Katzenellenbogen BS, Katzenellenbogen JA. Estrogen receptor-β potency-selective ligands: structure-activity relationship studies of diarylpropionitriles and their acetylene and polar analogues. J Med Chem. 2001; 44(24):4230–4251. PMID: 11708925.
Article27. Butler MJ, Hildebrandt RP, Eckel LA. Selective activation of estrogen receptors, ERα and GPER-1, rapidly decreases food intake in female rats. Horm Behav. 2018; 103:54–61. PMID: 29807036.
Article28. Heine PA, Taylor JA, Iwamoto GA, Lubahn DB, Cooke PS. Increased adipose tissue in male and female estrogen receptor-alpha knockout mice. Proc Natl Acad Sci U S A. 2000; 97(23):12729–12734. PMID: 11070086.
Article29. Avtanski D, Novaira HJ, Wu S, Romero CJ, Kineman R, Luque RM, et al. Both estrogen receptor α and β stimulate pituitary GH gene expression. Mol Endocrinol. 2014; 28(1):40–52. PMID: 24284820.
Article30. Khalid AB, Krum SA. Estrogen receptors alpha and beta in bone. Bone. 2016; 87:130–135. PMID: 27072516.
Article31. Lindberg MK, Weihua Z, Andersson N, Movérare S, Gao H, Vidal O, et al. Estrogen receptor specificity for the effects of estrogen in ovariectomized mice. J Endocrinol. 2002; 174(2):167–178. PMID: 12176656.
Article32. Hertrampf T, Schleipen B, Velders M, Laudenbach U, Fritzemeier KH, Diel P. Estrogen receptor subtype-specific effects on markers of bone homeostasis. Mol Cell Endocrinol. 2008; 291(1-2):104–108. PMID: 18433985.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Growth and Pubertal Development in Female Mice with Tissue-specific Knock out of Estrogen Receptor
- Effect of dietary legumes on bone-specific gene expression in ovariectomized rats
- Differential Skeletal Response to Ovariectomy in Young and Old Rats
- Evidence for estrogen receptor expression during medullary bone formation and resorption in estrogen-treated male Japanese quails (Coturnix coturnix japonica)
- Erratum: Title Correction. The Role of The Effects of Vitamin D Supplementation on Bone Mineral Density and Bone Mineral Content in Ovariectomized Rats Compensation in Rats