Cancer Res Treat.
2020 Oct;52(4):1211-1218. 10.4143/crt.2019.506.
Early Assessment of Response to Neoadjuvant Chemotherapy with 18F-FDG-PET/CT in Patients with Advanced-Stage Ovarian Cancer
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Institute of Women’s Life Medical Science, Yonsei University College of Medicine, Seoul, Korea
- 2Department of Pathology and Translational Genomics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
- 3Department of Nuclear Medicine, Yonsei University College of Medicine, Seoul, Korea
- KMID: 2507946
- DOI: http://doi.org/10.4143/crt.2019.506
Abstract
- Purpose
The aim of this study was to evaluate the ability of sequential 18F-fluorodeoxyglucose positron emission tomography/computed tomography (18F-FDG-PET/CT) after one cycle of neoadjuvant chemotherapy (NAC) to predict chemotherapy response before interval debulking surgery (IDS) in advanced-stage ovarian cancer patients.
Materials and Methods
Forty consecutive patients underwent 18F-FDG-PET/CT at baseline and after one cycle of NAC. Metabolic responses were assessed by quantitative decrease in the maximum standardized uptake value (SUVmax) with PET/CT. Decreases in SUVmax were compared with cancer antigen 125 (CA-125) level before IDS, response rate by Response Evaluation Criteria in Solid Tumors criteria before IDS, residual tumor at IDS, and I chemotherapy response score (CRS) at IDS.
Results
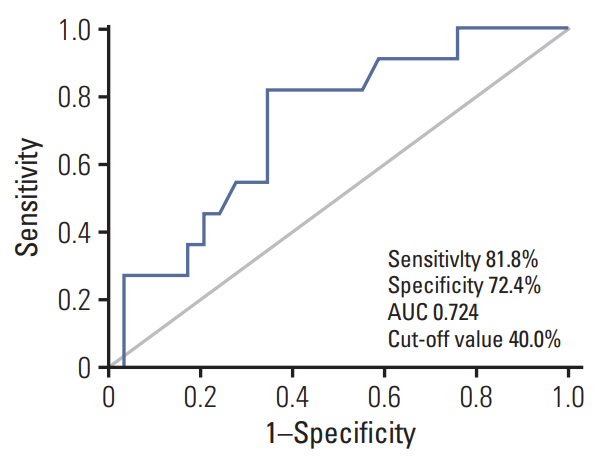
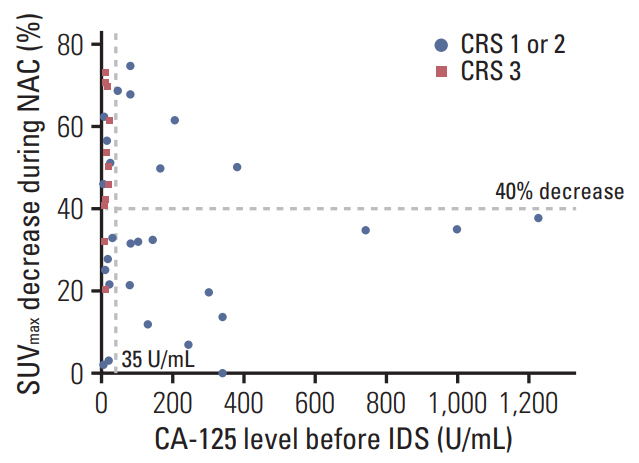
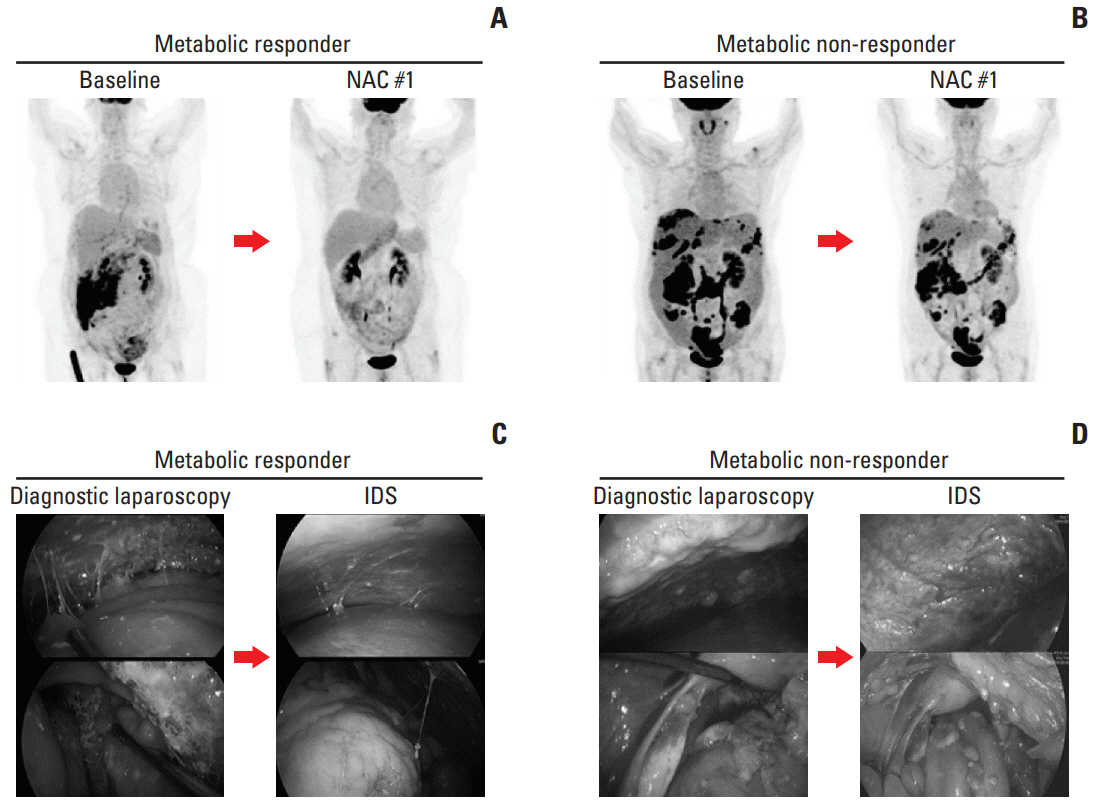
A 40% cut-off for the decrease in SUVmax provided the best performance to predict CRS 3 (compete or near-complete pathologic response), with sensitivity, specificity, and accuracy of 81.8%, 72.4%, and 72.4%, respectively. According to this 40% cut-off, there were 17 (42.5%) metabolic responders (≥ 40%) and 23 (57.5%) metabolic non-responders (< 40%). Metabolic responders had higher rate of CRS 3 (52.9% vs. 8.7%, p=0.003), CA-125 normalization (< 35 U/mL) before IDS (76.5% vs. 39.1%, p=0.019), and no residual tumor at IDS (70.6% vs. 31.8%, p=0.025) compared with metabolic non-responders. There were significant associations with progression-free survival (p=0.021) between metabolic responders and non-responders, but not overall survival (p=0.335).
Conclusion
Early assessment with 18F-FDG-PET/CT after one cycle of NAC can be useful to predic response to chemotherapy before IDS in patients with advanced-stage ovarian cancer.
Keyword
Figure
Reference
-
References
1. Kehoe S, Hook J, Nankivell M, Jayson GC, Kitchener H, Lopes T, et al. Primary chemotherapy versus primary surgery for newly diagnosed advanced ovarian cancer (CHORUS): an open-label, randomised, controlled, non-inferiority trial. Lancet. 2015; 386:249–57.
Article2. Vergote I, Trope CG, Amant F, Kristensen GB, Ehlen T, Johnson N, et al. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N Engl J Med. 2010; 363:943–53.
Article3. Groheux D, Hindie E, Giacchetti S, Hamy AS, Berger F, Merlet P, et al. Early assessment with 18F-fluorodeoxyglucose positron emission tomography/computed tomography can help predict the outcome of neoadjuvant chemotherapy in triple negative breast cancer. Eur J Cancer. 2014; 50:1864–71.4. Lordick F, Ott K, Krause BJ, Weber WA, Becker K, Stein HJ, et al. PET to assess early metabolic response and to guide treatment of adenocarcinoma of the oesophagogastric junction: the MUNICON phase II trial. Lancet Oncol. 2007; 8:797–805.
Article5. Evilevitch V, Weber WA, Tap WD, Allen-Auerbach M, Chow K, Nelson SD, et al. Reduction of glucose metabolic activity is more accurate than change in size at predicting histopathologic response to neoadjuvant therapy in high-grade soft-tissue sarcomas. Clin Cancer Res. 2008; 14:715–20.
Article6. Kostakoglu L, Goldsmith SJ. PET in the assessment of therapy response in patients with carcinoma of the head and neck and of the esophagus. J Nucl Med. 2004; 45:56–68.7. MacManus MP, Seymour JF, Hicks RJ. Overview of early response assessment in lymphoma with FDG-PET. Cancer Imaging. 2007; 7:10–8.
Article8. Burger IA, Schwarz EI, Samarin A, Breitenstein S, Weber A, Hany TF. Correlation between therapy response assessment using FDG PET/CT and histopathologic tumor regression grade in hepatic metastasis of colorectal carcinoma after neoadjuvant therapy. Ann Nucl Med. 2013; 27:177–83.
Article9. Park JS, Choi JY, Moon SH, Ahn YC, Lee J, Kim D, et al. Response evaluation after neoadjuvant chemoradiation by positron emission tomography-computed tomography for esophageal squamous cell carcinoma. Cancer Res Treat. 2013; 45:22–30.
Article10. Avril N, Sassen S, Schmalfeldt B, Naehrig J, Rutke S, Weber WA, et al. Prediction of response to neoadjuvant chemotherapy by sequential F-18-fluorodeoxyglucose positron emission tomography in patients with advanced-stage ovarian cancer. J Clin Oncol. 2005; 23:7445–53.
Article11. Nishiyama Y, Yamamoto Y, Kanenishi K, Ohno M, Hata T, Kushida Y, et al. Monitoring the neoadjuvant therapy response in gynecological cancer patients using FDG PET. Eur J Nucl Med Mol Imaging. 2008; 35:287–95.
Article12. Martoni AA, Fanti S, Zamagni C, Rosati M, De Iaco P, D’Errico Grigioni A, et al. [18F]FDG-PET/CT monitoring early identifies advanced ovarian cancer patients who will benefit from prolonged neo-adjuvant chemotherapy. Q J Nucl Med Mol Imaging. 2011; 55:81–90.13. Vallius T, Peter A, Auranen A, Carpen O, Kemppainen J, Matomaki J, et al. 18F-FDG-PET/CT can identify histopathological non-responders to platinum-based neoadjuvant chemotherapy in advanced epithelial ovarian cancer. Gynecol Oncol. 2016; 140:29–35.14. Muraji M, Sudo T, Iwasaki S, Ueno S, Wakahashi S, Yamaguchi S, et al. Histopathology predicts clinical outcome in advanced epithelial ovarian cancer patients treated with neo-adjuvant chemotherapy and debulking surgery. Gynecol Oncol. 2013; 131:531–4.
Article15. Petrillo M, Zannoni GF, Tortorella L, Pedone Anchora L, Salutari V, Ercoli A, et al. Prognostic role and predictors of complete pathologic response to neoadjuvant chemotherapy in primary unresectable ovarian cancer. Am J Obstet Gynecol. 2014; 211:632.
Article16. Le T, Williams K, Senterman M, Hopkins L, Faught W, Fung-Kee-Fung M. Histopathologic assessment of chemotherapy effects in epithelial ovarian cancer patients treated with neoadjuvant chemotherapy and delayed primary surgical debulking. Gynecol Oncol. 2007; 106:160–3.
Article17. Sassen S, Schmalfeldt B, Avril N, Kuhn W, Busch R, Hofler H, et al. Histopathologic assessment of tumor regression after neoadjuvant chemotherapy in advanced-stage ovarian cancer. Hum Pathol. 2007; 38:926–34.
Article18. Bohm S, Faruqi A, Said I, Lockley M, Brockbank E, Jeyarajah A, et al. Chemotherapy response score: development and validation of a system to quantify histopathologic response to neoadjuvant chemotherapy in tubo-ovarian high-grade serous carcinoma. J Clin Oncol. 2015; 33:2457–63.19. Lee JY, Chung YS, Na K, Kim HM, Park CK, Nam EJ, et al. External validation of chemotherapy response score system for histopathological assessment of tumor regression after neoadjuvant chemotherapy in tubo-ovarian high-grade serous carcinoma. J Gynecol Oncol. 2017; 28:e73.
Article20. Fagotti A, Ferrandina G, Fanfani F, Garganese G, Vizzielli G, Carone V, et al. Prospective validation of a laparoscopic predictive model for optimal cytoreduction in advanced ovarian carcinoma. Am J Obstet Gynecol. 2008; 199:642.
Article21. Lee YJ, Lee JY, Cho MS, Nam EJ, Kim SW, Kim S, et al. Incorporation of paclitaxel-based hyperthermic intraperitoneal che-motherapy in patients with advanced-stage ovarian cancer treated with neoadjuvant chemotherapy followed by interval debulking surgery: a protocol-based pilot study. J Gynecol Oncol. 2019; 30:e3.
Article22. Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009; 45:228–47.
Article23. Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J Nucl Med. 2009; 50 Suppl 1:122S–50S.
Article24. Lee JY, Yoon JK, Kim B, Kim S, Kim MA, Lim H, et al. Tumor evolution and intratumor heterogeneity of an epithelial ovarian cancer investigated using next-generation sequencing. BMC Cancer. 2015; 15:85.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- PET Imaging for Gynecologic Malignancy
- The role of positron emission tomography in the diagnosis and management of gynecologic malignancies
- Imaging of Gastric Cancer Metabolism Using 18 F-FDG PET/CT
- The Use of PET in Esophageal Cancer
- The Role of fluorodeoxyglucose PET in the management of breast cancer