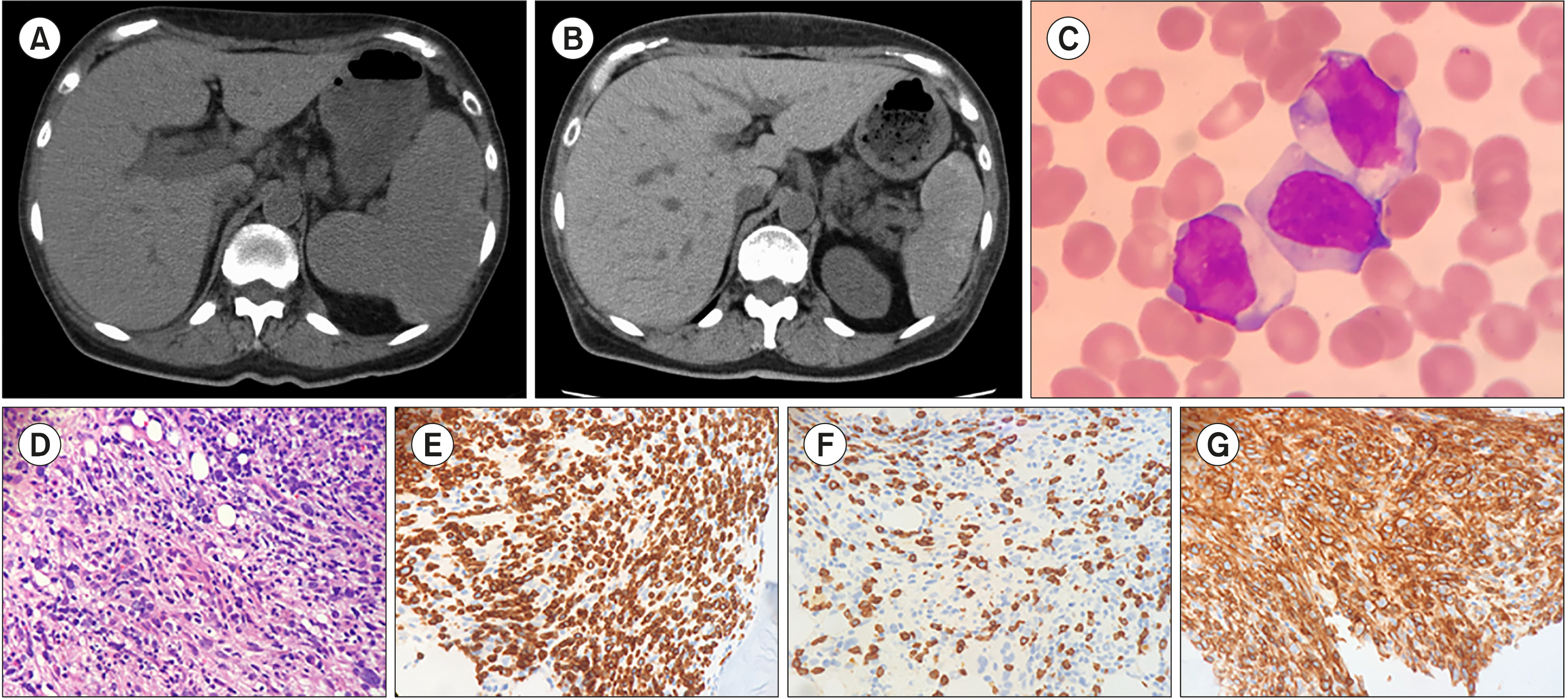
Blood Res.
2020 Sep;55(3):175-177. 10.5045/br.2020.2020045.
Successful response with romidepsin in relapsed peripheral T-cell lymphoma, not otherwise specified with leukemic progression
- Affiliations
-
- 1Division of Hematology, Shonan Kamakura General Hospital, Kanagawa, Japan
- KMID: 2507058
- DOI: http://doi.org/10.5045/br.2020.2020045
Abstract
- No abstract available.
Figure
Reference
-
1. Pileri SA, Orazi A, Falini B. Swerdlow SH, Campo E, Harris NL, editors. 2017. Myeloid sarcoma. WHO classification of tumours of haematopoietic and lymphoid tissues. Revised 4th ed. France: IARC Press;Lyon: p. 167–8.
Article2. Rizvi MA, Evens AM, Tallman MS, Nelson BP, Rosen ST. 2006; T-cell non-Hodgkin lymphoma. Blood. 107:1255–64. DOI: 10.1182/blood-2005-03-1306. PMID: 16210342.
Article3. Foss FM, Zinzani PL, Vose JM, Gascoyne RD, Rosen ST, Tobinai K. 2011; Peripheral T-cell lymphoma. Blood. 117:6756–67. DOI: 10.1182/blood-2010-05-231548. PMID: 30596214.
Article4. Kawamoto K, Miyoshi H, Yanagida E, et al. 2017; Comparison of clinicopathological characteristics between T-cell prolymphocytic leukemia and peripheral T-cell lymphoma, not otherwise specified. Eur J Haematol. 98:459–66. DOI: 10.1111/ejh.12856. PMID: 28129454.
Article5. Coiffier B, Pro B, Prince HM, et al. 2012; Results from a pivotal, open-label, phase II study of romidepsin in relapsed or refractory peripheral T-cell lymphoma after prior systemic therapy. J Clin Oncol. 30:631–6. DOI: 10.1200/JCO.2011.37.4223. PMID: 22271479.
Article6. National Comprehensive Cancer Network. 2020. NCCN Guidelines. T-cell lymphomas. National Comprehensive Cancer Network;Plymouth Meeting, PA: at https://www.nccn.org/professionals/physician_gls/default.aspx. Accessed February 12, 2020.7. Coiffier B, Pro B, Prince HM, et al. 2014; Romidepsin for the treatment of relapsed/refractory peripheral T-cell lymphoma: pivotal study update demonstrates durable responses. J Hematol Oncol. 7:11. DOI: 10.1186/1756-8722-7-11. PMID: 24456586. PMCID: PMC4016573.
Article8. Piekarz RL, Frye R, Prince HM, et al. 2011; Phase 2 trial of romidepsin in patients with peripheral T-cell lymphoma. Blood. 177:5827–34. DOI: 10.1182/blood-2010-10-312603. PMID: 21355097. PMCID: PMC3112033.9. Maruyama D, Tobinai K, Ogura M, et al. 2017; Romidepsin in Japanese patients with relapsed or refractory peripheral T-cell lymphoma: a phase I/II and pharmacokinetics study. Int J Hematol. 106:655–65. DOI: 10.1007/s12185-017-2286-1. PMID: 28664499.
Article10. Brunvand MW, Carson J. 2018; Complete remission with romidepsin in a patient with T-cell acute lymphoblastic leukemia refractory to induction hyper-CVAD. Hematol Oncol. 36:340–3. DOI: 10.1002/hon.2421. PMID: 28560733. PMCID: PMC5836898.
Article11. Marchi E, Zullo KM, Amengual JE, et al. 2015; The combination of hypomethylating agents and histone deacetylase inhibitors produce marked synergy in preclinical models of T-cell lymphoma. Br J Haematol. 171:215–26. DOI: 10.1111/bjh.13566. PMID: 26194163.
Article12. O'Connor OA, Zullo K, Marchi E, et al. 2015; Targeting epigenetic operations with HDAC inhibitor and hypomethylating drugs in combination exhibit synergy in preclinical and clinical experiences in drug resistant T-cell lymphoma (TCL): a translational focus on doublet development. Blood (ASH Annual Meeting Abstract). 126(Suppl):1282. DOI: 10.1182/blood.V126.23.1282.1282.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Circulating Lymphoma Cells in the Peripheral Blood from 4 Cases of Mantle and T Cell Types of Non-Hodgkin's Lymphoma: Light and Electron Microscopic Morphology
- Clinical Characteristics and Treatment Outcomes of Non-anaplastic Peripheral T-Cell Lymphoma in Children and Adolescents: A Single-center Experience
- BEAM conditioning regimen and autologous peripheral stem cell transplantation in patients with malignant lymphoma
- A Case of Neurolymphomatosis Relapsed as Mononeuropathy Multiplex after Hematologic Remission
- Assessing the Efficacy of Bortezomib and Dexamethasone for Induction and Maintenance Therapy in Relapsed/Refractory Cutaneous T-Cell Lymphoma: A Phase II CISL1701/BIC Study