Korean J Gastroenterol.
2020 Sep;76(3):142-149. 10.4166/kjg.2020.76.3.142.
Efficacy of Seven-day High-dose Esomeprazole-based Triple Therapy versus Seven-day Standard Dose Non-esomeprazole-based Triple Therapy as the First-line Treatment of Patients with Helicobacter pylori Infection
- Affiliations
-
- 1Departments of Internal Medicine, Kosin University College of Medicine, Busan, Korea
- 2Departments of Medicine, Kosin University College of Medicine, Busan, Korea
- KMID: 2506670
- DOI: http://doi.org/10.4166/kjg.2020.76.3.142
Abstract
- Background/Aims
The rates of Helicobacter pylori (H. pylori) eradication have declined with the use of proton pump inhibitor-amoxicillin-clarithromycin as the first-line triple therapy. On the other hand, several studies have suggested that high gastric pH levels could affect the H. pylori eradication rate by enhancing the efficacy of antimicrobials. This study compared the efficacy of seven-day high-dose esomeprazole-based triple therapy (7-HEAC) for first-line H. pylori eradication with the seven-day standard dose non-esomeprazole-based triple therapy (7-NEAC) to identify the risk factors related to eradication failure.
Methods
This study included 223 patients who were diagnosed with a H. pylori infection and received 7-HEAC or 7-NEAC between June 2016 and January 2017. The H. pylori eradication rates, as well as demographic and clinical factors, were investigated retrospectively. H. pylori eradication was confirmed by a 13C-urea breath test or rapid urease test at least 4 weeks after the completion of therapy.
Results
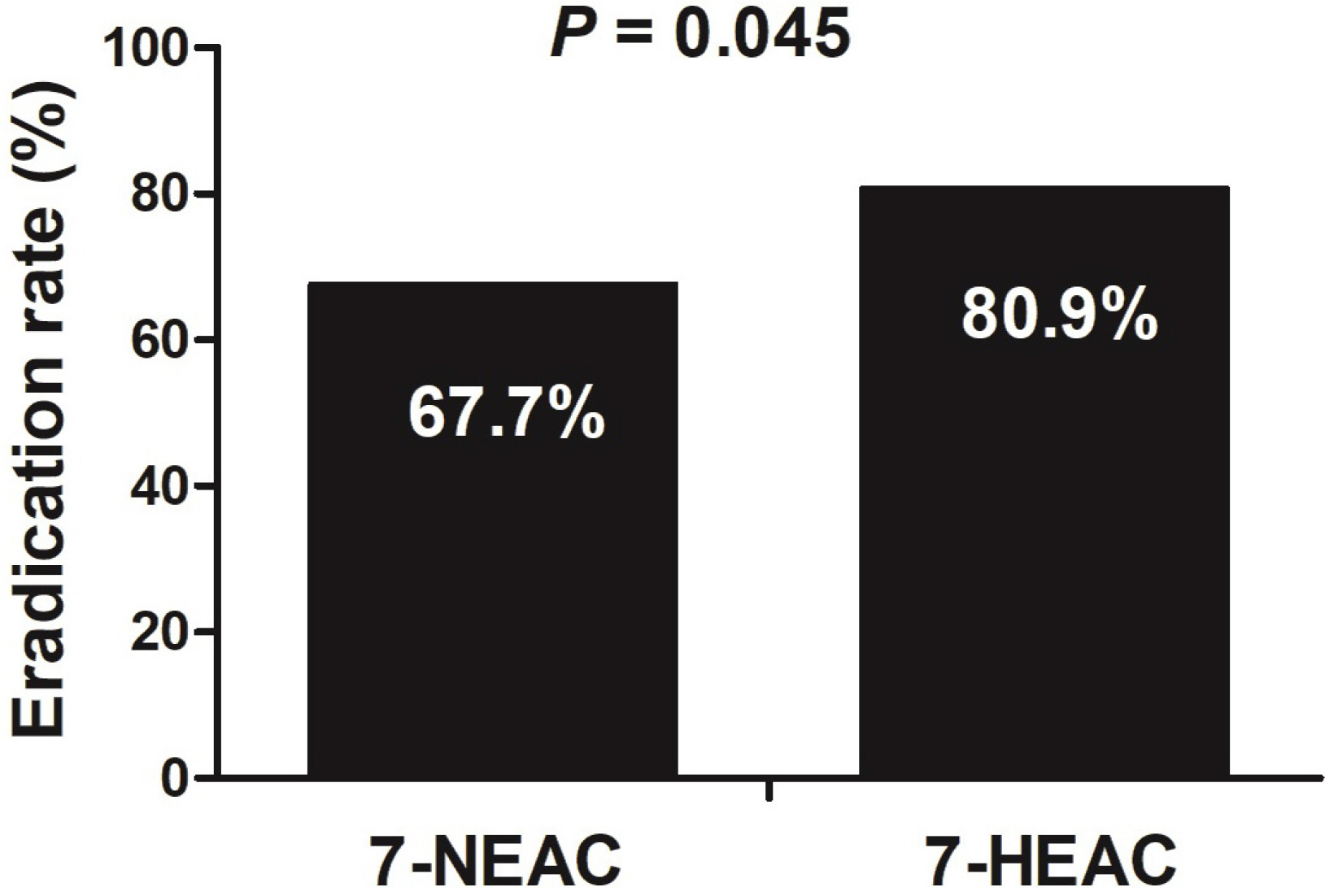
The eradication rates were 67.7% (105/155; 95% CI 59.5-74.8%) in the 7-NEAC group and 80.9% (55/68; 95% CI 69.9-89.8%) in the 7-HEAC group (p=0.045). The adverse event rates were 5.8% (9/155) in the 7-NEAC group and 7.4% (5/68) in the 7-HEAC group (p=0.661). Multivariate analysis revealed being female (OR 2.08; 95% CI 1.15-3.76) to be associated with the failure of H. pylori eradication therapy.
Conclusions
The eradication rate of the 7-HEAC group was higher than that of the 7-NEAC group. Nevertheless, more effective first-line therapies may be necessary for H. pylori eradication in the near future.
Figure
Reference
-
1. Yeo YH, Shiu SI, Ho HJ, et al. 2018; First-line Helicobacter pylori eradication therapies in countries with high and low clarithromycin resistance: a systematic review and network meta-analysis. Gut. 67:20–27. DOI: 10.1136/gutjnl-2016-311868. PMID: 27670375.
Article2. Shin WG, Lee SW, Baik GH, et al. 2016; Eradication rates of Helicobacter pylori in Korea over the past 10 years and correlation of the amount of antibiotics use: nationwide survey. Helicobacter. 21:266–278. DOI: 10.1111/hel.12279. PMID: 26470999.3. Lindberg P, Keeling D, Fryklund J, Andersson T, Lundborg P, Carlsson E. 2003; Review article: esomeprazole--enhanced bio-availability, specificity for the proton pump and inhibition of acid secretion. Aliment Pharmacol Ther. 17:481–488. DOI: 10.1046/j.1365-2036.2003.01481.x. PMID: 12622756.4. Hsu PI, Lai KH, Wu CJ, et al. 2007; High-dose versus low-dose esomeprazole-based triple therapy for Helicobacter pylori infection. Eur J Clin Invest. 37:724–730. DOI: 10.1111/j.1365-2362.2007.01852.x. PMID: 17696962.
Article5. Shin JM, Kim N. 2013; Pharmacokinetics and pharmacodynamics of the proton pump inhibitors. J Neurogastroenterol Motil. 19:25–35. DOI: 10.5056/jnm.2013.19.1.25. PMID: 23350044. PMCID: PMC3548122.
Article6. Oshima T, Miwa H. 2018; Potent potassium-competitive acid blockers: a new era for the treatment of acid-related diseases. J Neurogastroenterol Motil. 24:334–344. DOI: 10.5056/jnm18029. PMID: 29739175. PMCID: PMC6034668.
Article7. Choi HS, Park DI, Hwang SJ, et al. 2007; Double-dose, new-generation proton pump inhibitors do not improve Helicobacter pylori eradication rate. Helicobacter. 12:638–642. DOI: 10.1111/j.1523-5378.2007.00556.x. PMID: 18001407.
Article8. Anagnostopoulos GK, Tsiakos S, Margantinis G, Kostopoulos P, Arvanitidis D. 2004; Esomeprazole versus omeprazole for the eradication of Helicobacter pylori infection: results of a randomized controlled study. J Clin Gastroenterol. 38:503–506. DOI: 10.1097/01.mcg.0000129061.54277.c6. PMID: 15220685.9. Hsu PI, Lai KH, Lin CK, et al. 2005; A prospective randomized trial of esomeprazole-versus pantoprazole-based triple therapy for Helicobacter pylori eradication. Am J Gastroenterol. 100:2387–2392. DOI: 10.1111/j.1572-0241.2005.00264.x. PMID: 16279889.10. Villoria A, Garcia P, Calvet X, Gisbert JP, Vergara M. 2008; Meta-analysis: high-dose proton pump inhibitors vs. standard dose in triple therapy for Helicobacter pylori eradication. Aliment Pharmacol Ther. 28:868–877. DOI: 10.1111/j.1365-2036.2008.03807.x. PMID: 18644011.11. De Francesco V, Ridola L, Hassan C, et al. 2016; Two-week triple therapy with either standard or high-dose esomeprazole for first-line H. pylori eradication. J Gastrointestin Liver Dis. 25:147–150. DOI: 10.15403/jgld.2014.1121.252.2w3. PMID: 27308644.
Article12. Gisbert JP, Domínguez-Muñoz A, Domínguez-Martín A, Gisbert JL, Marcos S. 2005; Esomeprazole-based therapy in Helicobacter pylori eradication: any effect by increasing the dose of esomeprazole or prolonging the treatment? Am J Gastroenterol. 100:1935–1940. DOI: 10.1111/j.1572-0241.2005.00178.x. PMID: 16128936.
Article13. Ishizaki T, Horai Y. 1999; Review article: cytochrome P450 and the metabolism of proton pump inhibitors--emphasis on rabeprazole. Aliment Pharmacol Ther. 13(Suppl 3):27–36. DOI: 10.1046/j.1365-2036.1999.00022.x. PMID: 10491726.14. Scott LJ, Dunn CJ, Mallarkey G, Sharpe M. 2002; Esomeprazole: a review of its use in the management of acid-related disorders. Drugs. 62:1503–1538. DOI: 10.2165/00003495-200262100-00006. PMID: 12093317. PMCID: PMC3810197.15. Scott DR, Sachs G, Marcus EA. 2016; The role of acid inhibition in Helicobacter pylori eradication. F1000Res. 5:F1000 Faculty Rev-1747. DOI: 10.12688/f1000research.8598.1. PMID: 30023042. PMCID: PMC4957631.
Article16. Marcus EA, Inatomi N, Nagami GT, Sachs G, Scott DR. 2012; The effects of varying acidity on Helicobacter pylori growth and the bactericidal efficacy of ampicillin. Aliment Pharmacol Ther. 36:972–979. DOI: 10.1111/apt.12059. PMID: 23009227. PMCID: PMC3474890.17. Sugimoto M, Furuta T, Shirai N, et al. 2007; Evidence that the degree and duration of acid suppression are related to Helicobacter pylori eradication by triple therapy. Helicobacter. 12:317–323. DOI: 10.1111/j.1523-5378.2007.00508.x. PMID: 17669104.
Article18. Graham DY, Lu H, Yamaoka Y. 2007; A report card to grade Helicobacter pylori therapy. Helicobacter. 12:275–278. DOI: 10.1111/j.1523-5378.2007.00518.x. PMID: 17669098.
Article19. Gong EJ, Yun SC, Jung HY, et al. 2014; Meta-analysis of first-line triple therapy for Helicobacter pylori eradication in Korea: is it time to change? J Korean Med Sci. 29:704–713. DOI: 10.3346/jkms.2014.29.5.704. PMID: 24851029. PMCID: PMC4024949.
Article20. Lee JH, Ahn JY, Choi KD, et al. 2019; Nationwide antibiotic resistance mapping of Helicobacter pylori in Korea: a prospective multicenter study. Helicobacter. 24:e12592. DOI: 10.1111/hel.12592. PMID: 31111572.21. Ong S, Kim SE, Kim JH, et al. 2019; Helicobacter pylori eradication rates with concomitant and tailored therapy based on 23S rRNA point mutation: a multicenter randomized controlled trial. Helicobacter. 24:e12654. DOI: 10.1111/hel.12654. PMID: 31411793.
Article22. Osato MS, Reddy R, Reddy SG, Penland RL, Malaty HM, Graham DY. 2001; Pattern of primary resistance of Helicobacter pylori to metronidazole or clarithromycin in the United States. Arch Intern Med. 161:1217–1220. DOI: 10.1001/archinte.161.9.1217. PMID: 11343444.
Article23. Cai W, Zhou L, Ren W, Deng L, Yu M. 2009; Variables influencing outcome of Helicobacter pylori eradication therapy in South China. Helicobacter. 14:91–96. DOI: 10.1111/j.1523-5378.2009.00718.x. PMID: 19751433. PMCID: PMC3320751.
Article24. Kim SE, Park MI, Park SJ, et al. 2015; Trends in Helicobacter pylori eradication rates by first-line triple therapy and related factors in eradication therapy. Korean J Intern Med. 30:801–807. DOI: 10.3904/kjim.2015.30.6.801. PMID: 26552455. PMCID: PMC4642009.25. Chang YW, Ko WJ, Oh CH, et al. 2019; Clarithromycin resistance and female gender affect Helicobacter pylori eradication failure in chronic gastritis. Korean J Intern Med. 34:1022–1029. DOI: 10.3904/kjim.2018.054. PMID: 29898576. PMCID: PMC6718756.
Article26. Moayyedi P, Chalmers DM, Axon AT. 1997; Patient factors that predict failure of omeprazole, clarithromycin, and tinidazole to eradicate Helicobacter pylori. J Gastroenterol. 32:24–27. DOI: 10.1007/BF01213292. PMID: 9058291.27. Kim T, Song HJ, Shin SY, et al. 2013; Clarithromycin-resistant Helicobacter pylori associated with 23S rRNA point mutations in Jeju Island. Korean J Gastroenterol. 61:252–258. DOI: 10.4166/kjg.2013.61.5.252. PMID: 23756666. PMCID: PMC7140143.28. Mégraud F, Lehours P. 2007; Helicobacter pylori detection and antimicrobial susceptibility testing. Clin Microbiol Rev. 20:280–322. DOI: 10.1128/CMR.00033-06. PMID: 17428887. PMCID: PMC1865594.29. Versalovic J. 2003; Helicobacter pylori. Pathology and diagnostic strategies. Am J Clin Pathol. 119:403–412. DOI: 10.1309/5DTF5HT7NPLNA6J5. PMID: 12645343.30. Ferwana M, Abdulmajeed I, Alhajiahmed A, et al. 2015; Accuracy of urea breath test in Helicobacter pylori infection: meta-analysis. World J Gastroenterol. 21:1305–1314. DOI: 10.3748/wjg.v21.i4.1305. PMID: 25632206. PMCID: PMC4306177.31. Gatta L, Vakil N, Ricci C, et al. 2004; Effect of proton pump inhibitors and antacid therapy on 13C urea breath tests and stool test for Helicobacter pylori infection. Am J Gastroenterol. 99:823–829. DOI: 10.1111/j.1572-0241.2004.30162.x. PMID: 15128344.
Article32. Leodolter A, Wolle K, Malfertheiner P. 2001; Current standards in the diagnosis of Helicobacter pylori infection. Dig Dis. 19:116–122. DOI: 10.1159/000050665. PMID: 11549820.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- First-line Helicobacter pylori Eradication with Standard Triple Therapy and Concomitant Therapy: A Retrospective Study
- Proton Pump Inhibitor Switching Strategy after Failure of Standard Triple Therapy for Helicobacter pylori Infection
- Comparison of the Efficacy of 12-day Concomitant Quadruple Therapy versus 14-day High dose Dual Therapy as a First-line H. pylori Eradication Regimen
- Helicobacter pylori Treatment Strategies in Singapore
- The Efficacy of Levofloxacin Based Triple Therapy for Helicobacter pylori Eradication