Lab Med Online.
2020 Jul;10(3):214-220. 10.3343/lmo.2020.10.3.214.
Development and Performance Evaluation of a Quantitative Reverse Transcription PCR Kit for the Determination of prohibitin Gene Expression
- Affiliations
-
- 1Brain Korea 21 Plus Project, Chonnam National University Medical School, Gwangju, Korea
- 2Department of Laboratory Medicine, GwangYang Sarang General Hospital, GwangYang, Korea
- 3School of Electronics and Computer Engineering, College of Engineering, Chonnam National University, Gwangju, Korea
- 4College of Korean Medicine, Dongshin University, Naju, Korea
- 5Department of Laboratory Medicine, Chonnam National University Medical School and Chonnam National University Hwasun Hospital, Hwasun, Korea
- KMID: 2504131
- DOI: http://doi.org/10.3343/lmo.2020.10.3.214
Abstract
- Background
Prohibitin (PHB) regulates intracellular signal pathways, transcription, and cell cycles. Aberrant expression of the PHB gene is known to be related totumorigenesis, tumor progression, and chronic metabolic and inflammatory diseases. The present study aimed to develop a one-step quantitative reverse transcription PCR (RT-qPCR) kit for quantifying PHB mRNA levels and evaluate its performance in the laboratory.
Methods
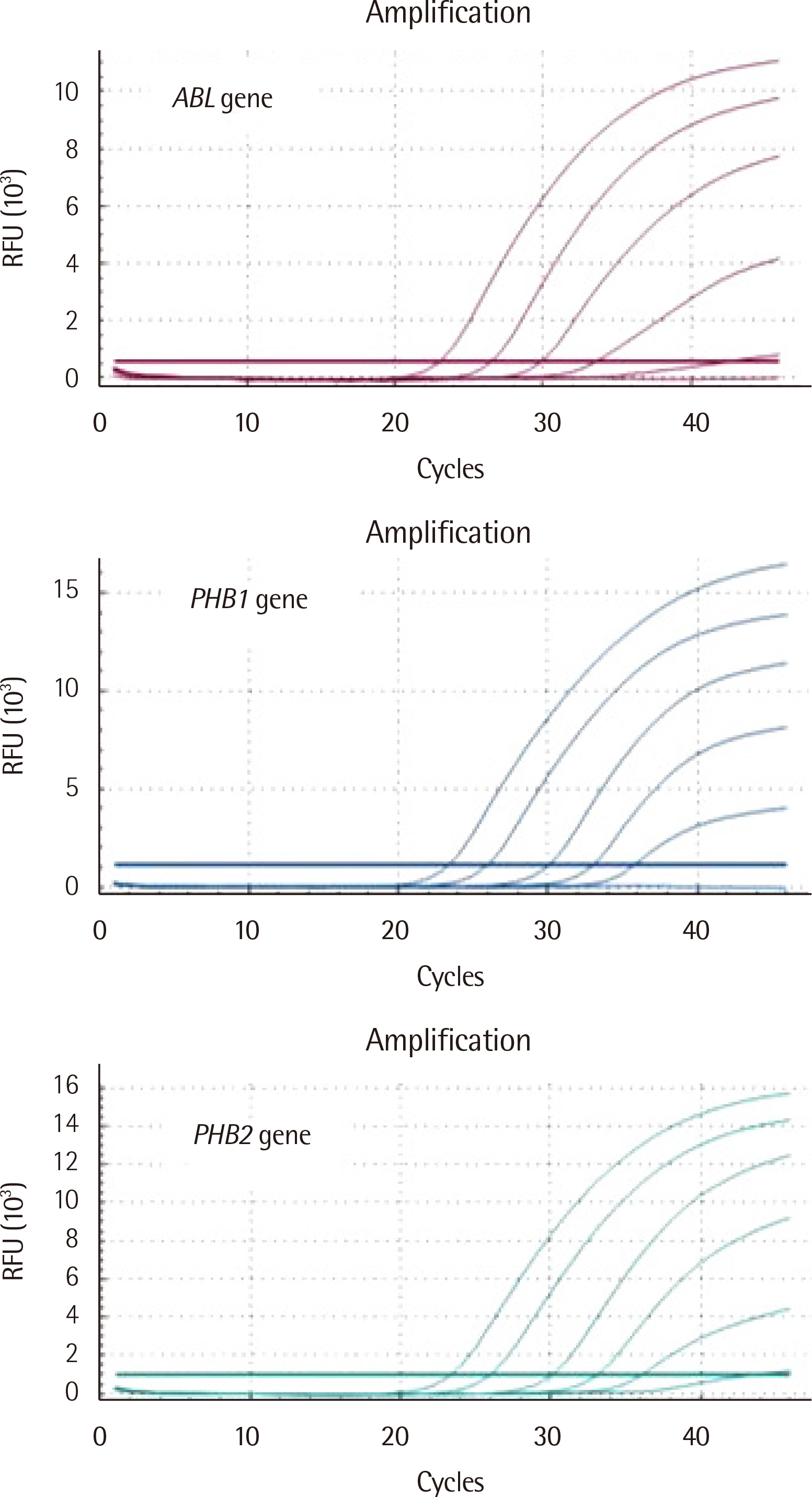
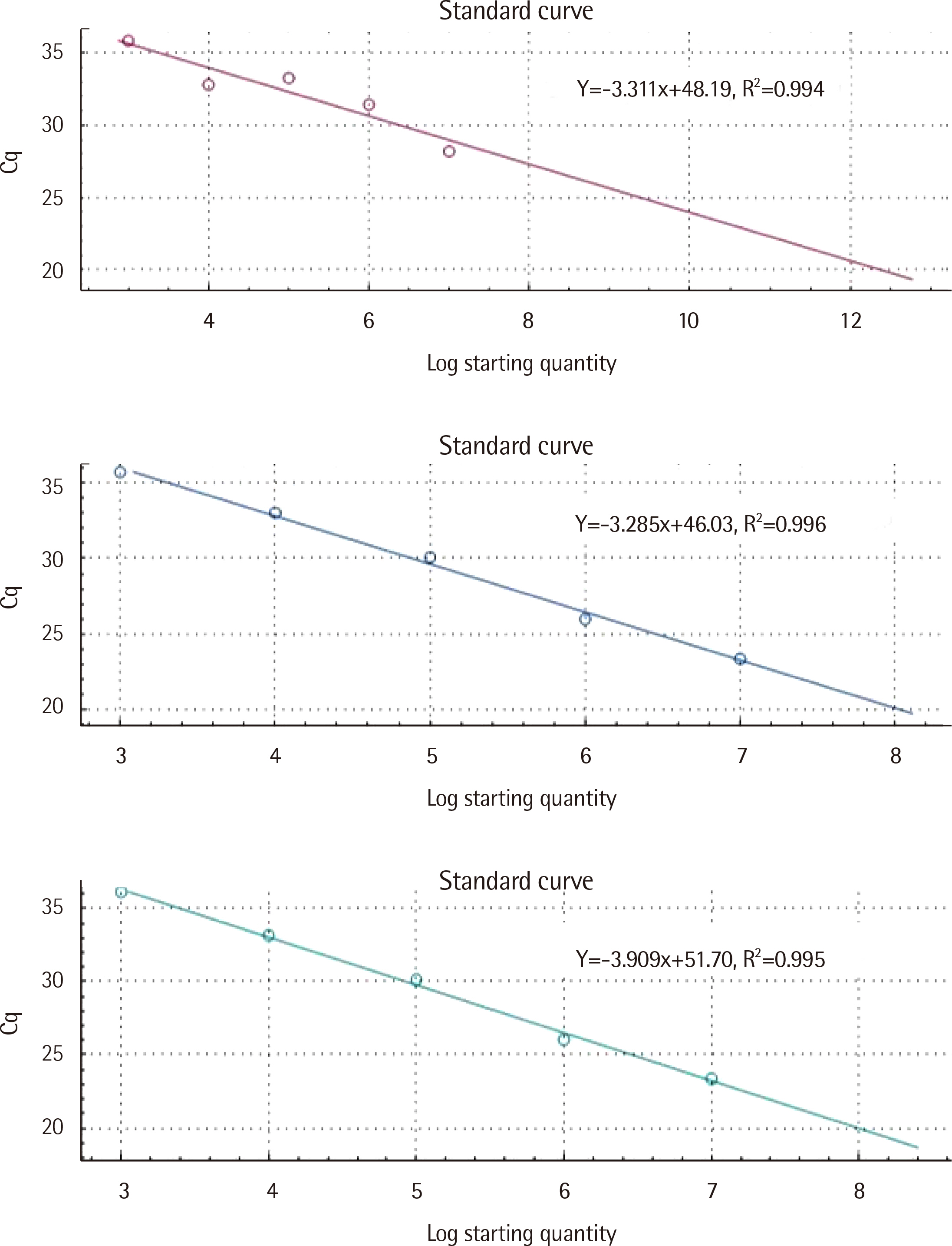
TaqMan chemistry was used to develop the one-step PHB1 and PHB2 RT-qPCR kit. Normal peripheral blood cells from healthy individuals (N=20) and leukemia cells from patients initially diagnosed with acute myeloid leukemia (AML, N=20), chronic myeloid leukemia (CML, N=13), and acute lymphoid leukemia (ALL, N=7) were enrolled to evaluate the laboratory performance of the kit using commercially available total human RNA controls.
Results
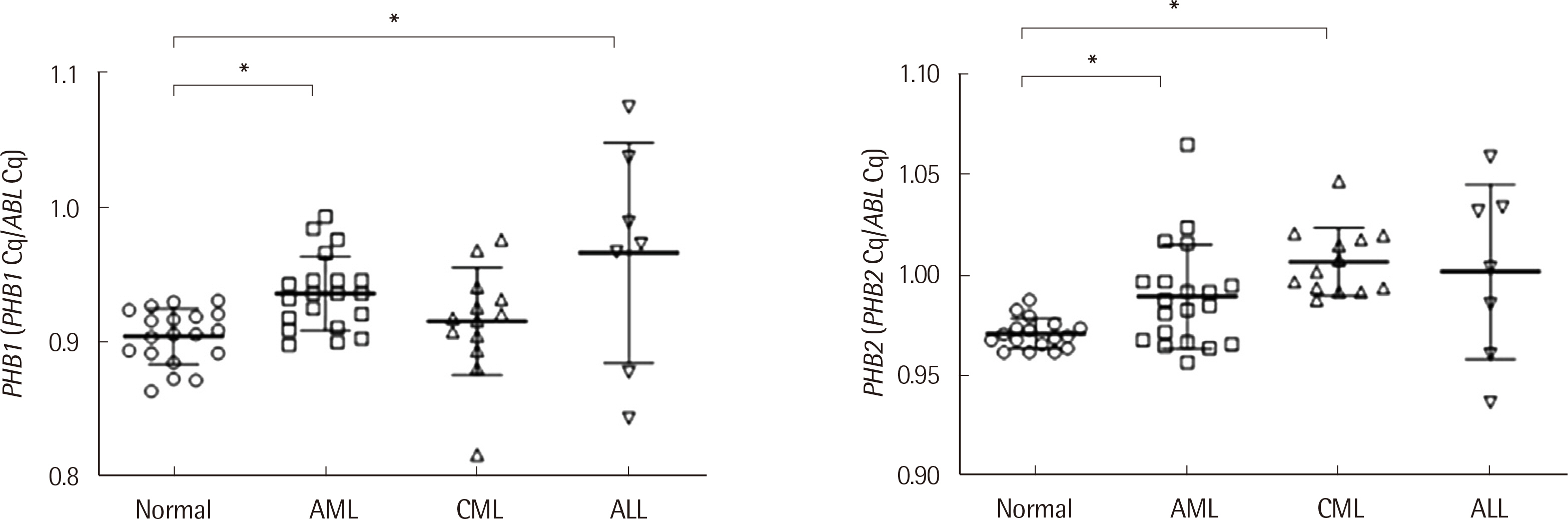
The intra-assay and inter-assay precision of the kit developed in this study was less than 2%. The distribution of PHB1 mRNA expression of AML, CML, and ALL was 0.898-0.993 (median: 0.936), 0.817-0.976 (0.918), and 0.844-1.074 (0.973), respectively. The distribution of PHB2 mRNA expression of AML, CML, and ALL was 0.957-1.024 (median: 0.985), 0.988-1.047 (1.002), and 0.937-1.059 (1.004), respectively. The sensitivity, specificity, positive and negative predictive value, and test effectiveness of the developed PHB1 and PHB2 kit were greater than 50% for each parameter.
Conclusions
Our developed kit would be useful for diagnosing leukemia as well as detecting residual disease. Additionally, this kit could be used for monitoring and conducting molecular pathophysiological studies of obesity, metabolic, and inflammatory diseases.
Keyword
Figure
Reference
-
1. Tatsuta T, Model K, Langer T. 2005; Formation of membrane-bound ring complexes by prohibitins in mitochondria. Mol Biol Cell. 16:248–59. DOI: 10.1091/mbc.e04-09-0807. PMID: 15525670. PMCID: PMC539169.
Article2. Kurtev V, Margueron R, Kroboth K, Ogris E, Cavailles V, Seiser C. 2004; Transcriptional regulation by the repressor of estrogen receptor activity via recruitment of histone deacetylases. J Biol Chem. 279:24834–43. DOI: 10.1074/jbc.M312300200. PMID: 15140878.
Article3. Signorile A, Sgaramella G, Bellomo F, De Rasmo D. 2019; Prohibitins: acritical role in mitochondrial functions and implication in diseases. Cells. 8:71. DOI: 10.3390/cells8010071. PMID: 30669391. PMCID: PMC6356732.4. Artal-Sanz M, Tavernarakis N. 2009; Prohibitin couples diapause signalling to mitochondrial metabolism during ageing in C. elegans. Nature. 461:793–7. DOI: 10.1038/nature08466. PMID: 19812672.5. Koushyar S, Jiang WG, Dart DA. 2015; Unveiling the potential of prohibitin in cancer. Cancer Lett. 369:316–22. DOI: 10.1016/j.canlet.2015.09.012. PMID: 26450374.
Article6. Dutta D, Ali N, Banerjee E, Singh R, Naskar A, Paidi RK, et al. 2018; Low levels of prohibitin in substantia nigra makes dopaminergic neurons vulnerable in Parkinson's disease. Mol Neurobiol. 55:804–21. DOI: 10.1007/s12035-016-0328-y. PMID: 28062948.
Article7. Supale S, Thorel F, Merkwirth C, Gjinovci A, Herrera PL, Scorrano L, et al. 2013; Loss of prohibitin induces mitochondrial damages altering beta-cell function and survival and is responsible for gradual diabetes development. Diabetes. 62:3488–99. DOI: 10.2337/db13-0152. PMID: 23863811. PMCID: PMC3781460.8. Ross JA, Robles-Escajeda E, Oaxaca DM, Padilla DL, Kirken RA. 2017; The prohibitin protein complex promotes mitochondrial stabilization and cell survival in hematologic malignancies. Oncotarget. 8:65445–56. DOI: 10.18632/oncotarget.18920. PMID: 29029444. PMCID: PMC5630344.
Article9. Peng YT, Chen P, Ouyang RY, Song L. 2015; Multifaceted role of prohibitin in cell survival and apoptosis. Apoptosis. 20:1135–49. DOI: 10.1007/s10495-015-1143-z. PMID: 26091791. PMCID: PMC4531144.
Article10. Mishra S, Nyomba BG. 2017; Prohibitin - At the crossroads of obesity-linked diabetes and cancer. Exp Biol Med (Maywood). 242:1170–7. DOI: 10.1177/1535370217703976. PMID: 28399645. PMCID: PMC5477999.
Article11. Nijtmans LG, Artal SM, Grivell LA, Coates PJ. 2002; The mitochondrial PHB complex: roles in mitochondrial respiratory complex assembly, ageing and degenerative disease. Cell Mol Life Sci. 59:143–55. DOI: 10.1007/s00018-002-8411-0. PMID: 11852914.12. Fan W, Yang H, Liu T, Wang J, Li TW, Mavila N, et al. 2017; Prohibitin 1 suppresses liver cancer tumorigenesis in mice and human hepatocellular and cholangiocarcinoma cells. Hepatology. 65:1249–66. DOI: 10.1002/hep.28964. PMID: 27981602. PMCID: PMC5360526.
Article13. Merkwirth C, Langer T. 2009; Prohibitin function within mitochondria: essential roles for cell proliferation and cristae morphogenesis. Biochim Biophys Acta. 1793:27–32. DOI: 10.1016/j.bbamcr.2008.05.013. PMID: 18558096.
Article14. Lahiri V, Klionsky DJ. 2017; PHB2/prohibitin 2: An inner membrane mitophagy receptor. Cell Res. 27:311–2. DOI: 10.1038/cr.2017.23. PMID: 28220775. PMCID: PMC5339840.
Article15. Wei Y, Chiang WC, Sumpter R Jr, Mishra P, Levine B. 2017; Prohibitin 2 is an inner mitochondrial membrane mitophagy receptor. Cell. 168:224–38.e10. DOI: 10.1016/j.cell.2016.11.042. PMID: 28017329. PMCID: PMC5235968.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Performance Evaluation of the CareGENETM Zika Virus Reverse Transcription-PCR Kit for Urine Specimen
- Gene Amplification and Overexpression in BPH by Means of One-step Real Time Quantitative PCR
- Genetic Architecture of Transcription and Chromatin Regulation
- Comparison Between the SFTS-QS Kit and the PowerChek SFTSV Real-time PCR Kit for the Detection of Severe Fever With Thrombocytopenia Syndrome Virus
- Development of Quantitative Reverse Transcription-Polymerase Chain Reaction for the Mesurement of Angiotensin Converting Enzyme mRNA