J Korean Med Sci.
2020 Jul;35(26):e225. 10.3346/jkms.2020.35.e225.
Anti-Allodynic Effects of Polydeoxyribonucleotide in an Animal Model of Neuropathic Pain and Complex Regional Pain Syndrome
- Affiliations
-
- 1Department of Anesthesiology and Pain Medicine, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul, Korea
- 2Department of Anesthesiology and Pain Medicine, Soonchunhyang University Cheonan Hospital, Soonchunhyang University College of Medicine, Cheonan, Korea
- 3Department of Anesthesiology and Pain Medicine, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
- 4Department of Urology, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
- KMID: 2503682
- DOI: http://doi.org/10.3346/jkms.2020.35.e225
Abstract
- Background
Spinal nerve ligation (SNL) model is one of the representative models of the neuropathic pain model. Neuropathic pain in a chronic post-ischemic pain (CPIP) mimics the symptoms of complex regional pain syndrome (CRPS). The administration of polydeoxyribonucleotide (PDRN), which has regenerative and anti-inflammatory effects, has been studied and is used in clinical practice treating various diseases. However, the analgesic effect of PDRN in a neuropathic pain or CRPS model remains unknown.
Methods
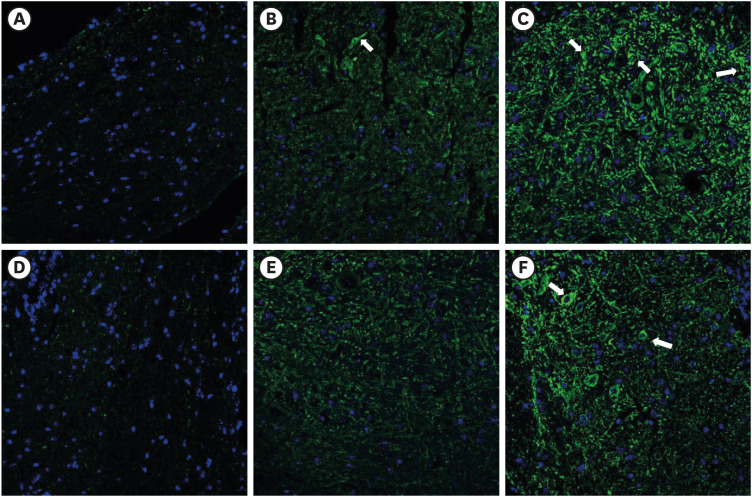
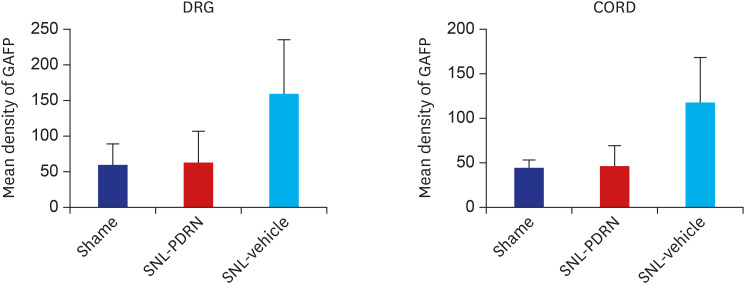
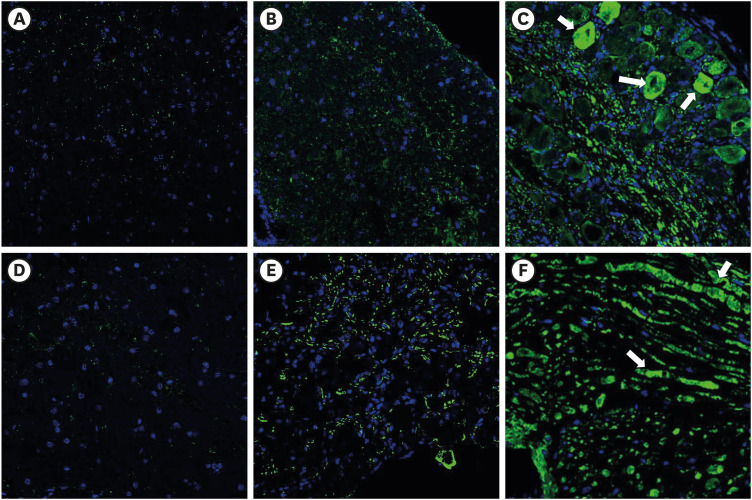
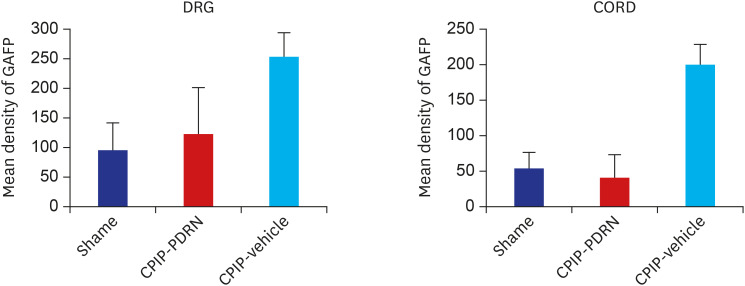
PDRN (3.3, 10, and 20 mg/kg) was administered into the subcutaneous (SC) layer of the hind paws of SNL and CPIP models. Mechanical anti-allodynic effects were then investigated using the von Frey test. In the immunohistochemical examination, dorsal root ganglia (DRG) and the spinal cord were harvested and examined for the expression of glial fibrillary acidic protein (GFAP) after the 20 mg PDRN injection.
Results
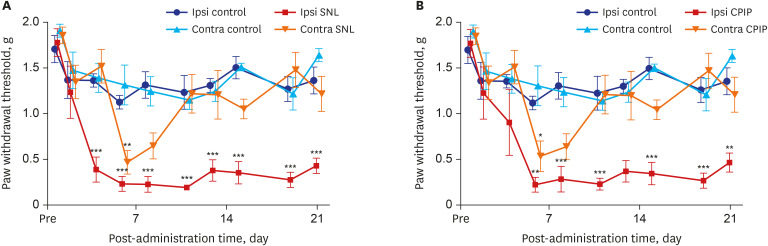
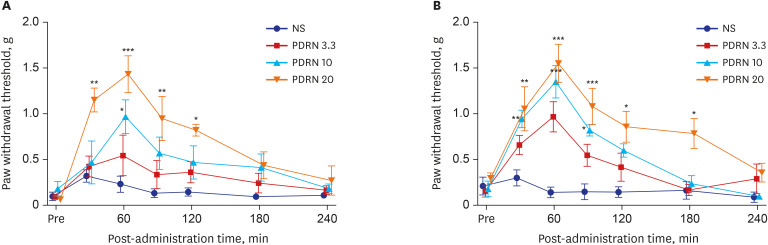
Mechanical allodynia was significantly alleviated by administration of PDRN in SNL and CPIP mice at all of the time point. As the dose of PDRN increased, the effect was greater. The 20 mg PDRN injection was found to have the most effective anti-allodynic effect. The increased expression of GFAP in DRG and the spinal cord of SNL and CPIP model decreased following the administration of PDRN than vehicle.
Conclusion
SC administration of PDRN results in the attenuation of allodynia and activation of astrocytes in neuropathic pain or CRPS models. We propose that PDRN can have significant potential advantages in neuropathic pain treatment.
Keyword
Figure
Cited by 1 articles
-
Effect of epidural polydeoxyribonucleotide in a rat model of lumbar foraminal stenosis
Ho-Jin Lee, Jiyoun Ju, Eunjoo Choi, Francis Sahngun Nahm, Ghee Young Choe, Pyung Bok Lee
Korean J Pain. 2021;34(4):394-404. doi: 10.3344/kjp.2021.34.4.394.
Reference
-
1. Zilliox LA. Neuropathic pain. Continuum (Minneap Minn). 2017; 23(2):512–532. PMID: 28375916.
Article2. Taha R, Blaise GA. Update on the pathogenesis of complex regional pain syndrome: role of oxidative stress. Can J Anaesth. 2012; 59(9):875–881. PMID: 22798149.
Article3. Mrabet D, Khemiri C, Ben Mrad I, Mrabet H, Essaddem H, Amel M, et al. Pathophysiology of complex regional pain syndrome (CRPS) type 1. Tunis Med. 2012; 90(4):278–281. PMID: 22535340.4. Yang L, Wang SH, Hu Y, Sui YF, Peng T, Guo TC. Effects of repetitive transcranial magnetic stimulation on astrocytes proliferation and nNOS expression in neuropathic pain rats. Curr Med Sci. 2018; 38(3):482–490. PMID: 30074216.
Article5. Garrison CJ, Dougherty PM, Kajander KC, Carlton SM. Staining of glial fibrillary acidic protein (GFAP) in lumbar spinal cord increases following a sciatic nerve constriction injury. Brain Res. 1991; 565(1):1–7. PMID: 1723019.
Article6. Shen J, Huo BB, Zheng MX, Hua XY, Shen H, Lu YC, et al. Evaluation of neuropathic pain in a rat model of total brachial plexus avulsion from behavior to brain metabolism. Pain Physician. 2019; 22(3):E215–24. PMID: 31151344.7. Vallejo R, Tilley DM, Vogel L, Benyamin R. The role of glia and the immune system in the development and maintenance of neuropathic pain. Pain Pract. 2010; 10(3):167–184. PMID: 20384965.
Article8. Raghavendra V, Tanga F, DeLeo JA. Inhibition of microglial activation attenuates the development but not existing hypersensitivity in a rat model of neuropathy. J Pharmacol Exp Ther. 2003; 306(2):624–630. PMID: 12734393.
Article9. Bitto A, Galeano M, Squadrito F, Minutoli L, Polito F, Dye JF, et al. Polydeoxyribonucleotide improves angiogenesis and wound healing in experimental thermal injury. Crit Care Med. 2008; 36(5):1594–1602. PMID: 18434887.
Article10. Galeano M, Bitto A, Altavilla D, Minutoli L, Polito F, Calò M, et al. Polydeoxyribonucleotide stimulates angiogenesis and wound healing in the genetically diabetic mouse. Wound Repair Regen. 2008; 16(2):208–217. PMID: 18318806.
Article11. Bitto A, Polito F, Altavilla D, Minutoli L, Migliorato A, Squadrito F. Polydeoxyribonucleotide (PDRN) restores blood flow in an experimental model of peripheral artery occlusive disease. J Vasc Surg. 2008; 48(5):1292–1300. PMID: 18971038.
Article12. Valdatta L, Thione A, Mortarino C, Buoro M, Tuinder S. Evaluation of the efficacy of polydeoxyribonucleotides in the healing process of autologous skin graft donor sites: a pilot study. Curr Med Res Opin. 2004; 20(3):403–408. PMID: 15025849.
Article13. Koo Y, Yun Y. Effects of polydeoxyribonucleotides (PDRN) on wound healing: Electric cell-substrate impedance sensing (ECIS). Mater Sci Eng C. 2016; 69:554–560.
Article14. Vanelli R, Costa P, Rossi SM, Benazzo F. Efficacy of intra-articular polynucleotides in the treatment of knee osteoarthritis: a randomized, double-blind clinical trial. Knee Surg Sports Traumatol Arthrosc. 2010; 18(7):901–907. PMID: 20111953.
Article15. Guizzardi S, Galli C, Govoni P, Boratto R, Cattarini G, Martini D, et al. Polydeoxyribonucleotide (PDRN) promotes human osteoblast proliferation: a new proposal for bone tissue repair. Life Sci. 2003; 73(15):1973–1983. PMID: 12899922.
Article16. Bitto A, Polito F, Irrera N, D'Ascola A, Avenoso A, Nastasi G, et al. Polydeoxyribonucleotide reduces cytokine production and the severity of collagen-induced arthritis by stimulation of adenosine A(2A) receptor. Arthritis Rheum. 2011; 63(11):3364–3371. PMID: 21769841.17. Bitto A, Oteri G, Pisano M, Polito F, Irrera N, Minutoli L, et al. Adenosine receptor stimulation by polynucleotides (PDRN) reduces inflammation in experimental periodontitis. J Clin Periodontol. 2013; 40(1):26–32. PMID: 23033941.
Article18. Pallio G, Bitto A, Pizzino G, Galfo F, Irrera N, Squadrito F, et al. Adenosine receptor stimulation by polydeoxyribonucleotide improves tissue repair and symptomology in experimental colitis. Front Pharmacol. 2016; 7:273. PMID: 27601997.
Article19. Ko IG, Kim SE, Jin JJ, Hwang L, Ji ES, Kim CJ, et al. Combination therapy with polydeoxyribonucleotide and proton pump inhibitor enhances therapeutic effectiveness for gastric ulcer in rats. Life Sci. 2018; 203:12–19. PMID: 29665355.
Article20. Ye GL, Savelieva KV, Vogel P, Baker KB, Mason S, Lanthorn TH, et al. Ligation of mouse L4 and L5 spinal nerves produces robust allodynia without major motor function deficit. Behav Brain Res. 2015; 276:99–110. PMID: 24786331.
Article21. Kim SH, Chung JM. An experimental model for peripheral neuropathy produced by segmental spinal nerve ligation in the rat. Pain. 1992; 50(3):355–363. PMID: 1333581.22. Choi BM, Lee SH, An SM, Park DY, Lee GW, Noh GJ. The time-course and RNA interference of TNF-α, IL-6, and IL-1β expression on neuropathic pain induced by L5 spinal nerve transection in rats. Korean J Anesthesiol. 2015; 68(2):159–169. PMID: 25844135.
Article23. Coderre TJ, Xanthos DN, Francis L, Bennett GJ. Chronic post-ischemia pain (CPIP): a novel animal model of complex regional pain syndrome-type I (CRPS-I; reflex sympathetic dystrophy) produced by prolonged hindpaw ischemia and reperfusion in the rat. Pain. 2004; 112(1-2):94–105. PMID: 15494189.
Article24. Ryu TH, Jung KY, Ha MJ, Kwak KH, Lim DG, Hong JG. Superoxide and nitric oxide involvement in enhancing of N-methyl-D-aspartate receptor-mediated central sensitization in the chronic post-ischemia pain model. Korean J Pain. 2010; 23(1):1–10. PMID: 20552066.
Article25. Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL. Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods. 1994; 53(1):55–63. PMID: 7990513.
Article26. Bonin RP, Bories C, De Koninck Y. A simplified up-down method (SUDO) for measuring mechanical nociception in rodents using von Frey filaments. Mol Pain. 2014; 10:26. PMID: 24739328.
Article27. David Clark J, Tawfik VL, Tajerian M, Kingery WS. Autoinflammatory and autoimmune contributions to complex regional pain syndrome. Mol Pain. 2018; 14:1744806918799127. PMID: 30124090.
Article28. Schlereth T, Drummond PD, Birklein F. Inflammation in CRPS: role of the sympathetic supply. Auton Neurosci. 2014; 182:102–107. PMID: 24411269.
Article29. Martínez-Lavín M, Vargas A, Silveira LH, Amezcua-Guerra LM, Martínez-Martínez LA, Pineda C. Complex regional pain syndrome evolving to full-blown fibromyalgia: a proposal of common mechanisms. J Clin Rheumatol. Forthcoming. 2020.30. Russo M, Georgius P, Santarelli DM. A new hypothesis for the pathophysiology of complex regional pain syndrome. Med Hypotheses. 2018; 119:41–53. PMID: 30122490.
Article31. Hansson E. Could chronic pain and spread of pain sensation be induced and maintained by glial activation? Acta Physiol (Oxf). 2006; 187(1-2):321–327. PMID: 16734769.
Article32. Milligan ED, Watkins LR. Pathological and protective roles of glia in chronic pain. Nat Rev Neurosci. 2009; 10(1):23–36. PMID: 19096368.
Article33. Cao H, Zhang YQ. Spinal glial activation contributes to pathological pain states. Neurosci Biobehav Rev. 2008; 32(5):972–983. PMID: 18471878.
Article34. Broadhead MJ, Miles GB. Bi-directional communication between neurons and astrocytes modulates spinal motor circuits. Front Cell Neurosci. 2020; 14:30. PMID: 32180706.
Article35. Ma Z, Stork T, Bergles DE, Freeman MR. Neuromodulators signal through astrocytes to alter neural circuit activity and behaviour. Nature. 2016; 539(7629):428–432. PMID: 27828941.
Article36. Ben Achour S, Pascual O. Glia: the many ways to modulate synaptic plasticity. Neurochem Int. 2010; 57(4):440–445. PMID: 20193723.
Article37. Kwilasz AJ, Ellis A, Wieseler J, Loram L, Favret J, McFadden A, et al. Sustained reversal of central neuropathic pain induced by a single intrathecal injection of adenosine A2A receptor agonists. Brain Behav Immun. 2018; 69:470–479. PMID: 29366930.38. Luongo L, Salvemini D. Targeting metabotropic adenosine receptors for neuropathic pain: focus on A2A. Brain Behav Immun. 2018; 69:60–61. PMID: 29499304.
Article39. Sawynok J. Adenosine receptor targets for pain. Neuroscience. 2016; 338:1–18. PMID: 26500181.
Article40. Zhang ZJ, Jiang BC, Gao YJ. Chemokines in neuron-glial cell interaction and pathogenesis of neuropathic pain. Cell Mol Life Sci. 2017; 74(18):3275–3291. PMID: 28389721.
Article41. Old EA, Malcangio M. Chemokine mediated neuron-glia communication and aberrant signalling in neuropathic pain states. Curr Opin Pharmacol. 2012; 12(1):67–73. PMID: 22056024.
Article42. Ren K, Dubner R. Neuron-glia crosstalk gets serious: role in pain hypersensitivity. Curr Opin Anaesthesiol. 2008; 21(5):570–579. PMID: 18784481.
Article43. Polito F, Bitto A, Galeano M, Irrera N, Marini H, Calò M, et al. Polydeoxyribonucleotide restores blood flow in an experimental model of ischemic skin flaps. J Vasc Surg. 2012; 55(2):479–488. PMID: 22051873.
Article44. Thellung S, Florio T, Maragliano A, Cattarini G, Schettini G. Polydeoxyribonucleotides enhance the proliferation of human skin fibroblasts: involvement of A2 purinergic receptor subtypes. Life Sci. 1999; 64(18):1661–1674. PMID: 10328526.
Article45. Loram LC, Taylor FR, Strand KA, Harrison JA, Rzasalynn R, Sholar P, et al. Intrathecal injection of adenosine 2A receptor agonists reversed neuropathic allodynia through protein kinase (PK)A/PKC signaling. Brain Behav Immun. 2013; 33:112–122. PMID: 23811314.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Atypical presentation of complex regional pain syndrome: neuropathic itching - A case report -
- Treatment for Acute Stage Complex Regional Pain Syndrome Type II with Polydeoxyribonucleotide Injection
- Intrathecal Gabapentin Increases Interleukin-10 Expression and Inhibits Pro-Inflammatory Cytokine in a Rat Model of Neuropathic Pain
- The Effect of Epidural Ketamine in Complex Regional Pain Syndrome Occuring after Lumbar Epidural Block
- Synergistic anti-allodynic effect between intraperitoneal thalidomide and morphine on rat spinal nerve ligation-induced neuropathic pain