J Neurocrit Care.
2020 Jun;13(1):49-56. 10.18700/jnc.190104.
Predicting parenchymal hematoma associated with endovascular thrombectomy for acute occlusion of anterior circulation large vessel: the GuEss-MALiGn scale
- Affiliations
-
- 1Department of Neurology and Institute of Health Science, Gyeongsang National University Hospital, Gyeongsang National University College of Medicine, Jinju, Republic of Korea
- KMID: 2503385
- DOI: http://doi.org/10.18700/jnc.190104
Abstract
- Background
Endovascular thrombectomy (EVT) is an emergency treatment for stroke caused by anterior circulation large vessel occlusion (ACLVO). This study aimed to identify the predictors for post-EVT parenchymal hematoma (PH) and to develop a predictive tool using the identified factors.
Methods
Using the clinical and imaging data of consecutive patients with acute ACLVO who underwent EVT, we performed a multivariate binary logistic regression analysis to identify predictors for PH. With the predictors proved by the regression, we developed a scale for predicting PH using receiver operating characteristic (ROC) curve analyses.
Results
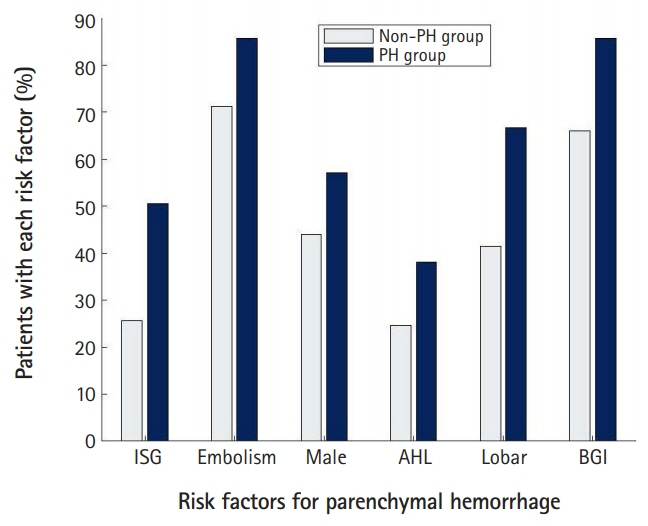
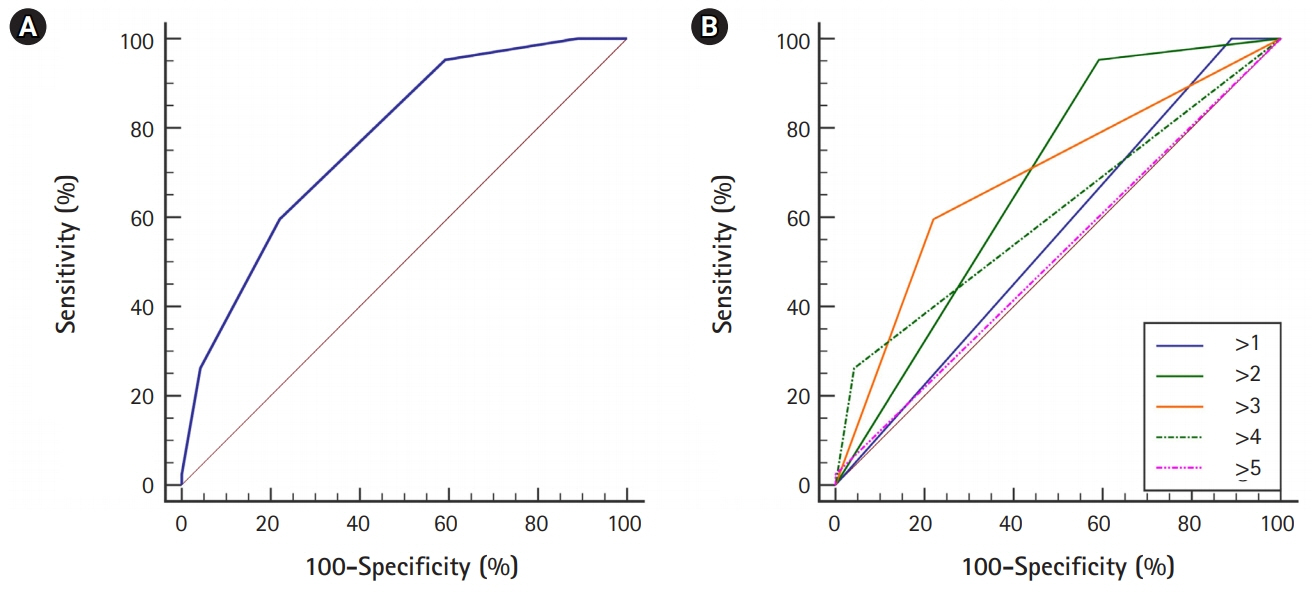
In 233 enrolled patients, the mean age was 72.3 years old, and the male proportion was 46.4%. The rate of PH after EVT was 18.0%: the rate of type 1 PH was 12.9%, and the rate of type 2 PH was 5.2%. The significant predictors for PH were basal ganglia involvement, embolism, male sex, antihyperlipidemic use, lobar infarction, and serum glucose level. We developed the GuEss-MALiGn scale with the six significant predictors. Each of these six items was placed on a Likert scale and scored as a 0 or 1. The ROC curve analysis revealed that the area under the curve was 0.771. The cutoff score for the risk of PH was >3. The sensitivity was 59.5%, and the specificity was 78.0%.
Conclusion
We propose the GuEss-MALiGn scale as a tool for predicting PH associated with EVT. Future external validation is needed to determine the reliability of this scale.
Keyword
Figure
Reference
-
1. Berkhemer OA, Fransen PS, Beumer D, van den Berg LA, Lingsma HF, Yoo AJ, et al. A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med. 2015; 372:11–20.2. Goyal M, Demchuk AM, Menon BK, Eesa M, Rempel JL, Thornton J, et al. Randomized assessment of rapid endovascular treatment of ischemic stroke. N Engl J Med. 2015; 372:1019–30.3. Jovin TG, Chamorro A, Cobo E, de Miquel MA, Molina CA, Rovira A, et al. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N Engl J Med. 2015; 372:2296–306.
Article4. Saver JL, Goyal M, Bonafe A, Diener HC, Levy EI, Pereira VM, et al. Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N Engl J Med. 2015; 372:2285–95.
Article5. Campbell BC, Mitchell PJ, Kleinig TJ, Dewey HM, Churilov L, Yassi N, et al. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med. 2015; 372:1009–18.
Article6. Nogueira RG, Gupta R, Jovin TG, Levy EI, Liebeskind DS, Zaidat OO, et al. Predictors and clinical relevance of hemorrhagic transformation after endovascular therapy for anterior circulation large vessel occlusion strokes: a multicenter retrospective analysis of 1122 patients. J Neurointerv Surg. 2015; 7:16–21.
Article7. Lee YB, Yoon W, Lee YY, Kim SK, Baek BH, Kim JT, et al. Predictors and impact of hemorrhagic transformations after endovascular thrombectomy in patients with acute large vessel occlusions. J Neurointerv Surg. 2019; 11:469–73.
Article8. Fiorelli M, Bastianello S, von Kummer R, del Zoppo GJ, Larrue V, Lesaffre E, et al. Hemorrhagic transformation within 36 hours of a cerebral infarct: relationships with early clinical deterioration and 3-month outcome in the European Cooperative Acute Stroke Study I (ECASS I) cohort. Stroke. 1999; 30:2280–4.9. Hart RG, Catanese L, Perera KS, Ntaios G, Connolly SJ. Embolic stroke of undetermined source: a systematic review and clinical update. Stroke. 2017; 48:867–72.10. Higashida RT, Furlan AJ, Roberts H, Tomsick T, Connors B, Barr J, et al. Trial design and reporting standards for intra-arterial cerebral thrombolysis for acute ischemic stroke. Stroke. 2003; 34:e109–37.
Article11. Loh Y, Towfighi A, Liebeskind DS, MacArthur DL, Vespa P, Gonzalez NR, et al. Basal ganglionic infarction before mechanical thrombectomy predicts poor outcome. Stroke. 2009; 40:3315–20.
Article12. Bang OY, Saver JL, Alger JR, Shah SH, Buck BH, Starkman S, et al. Patterns and predictors of blood-brain barrier permeability derangements in acute ischemic stroke. Stroke. 2009; 40:454–61.
Article13. del Zoppo GJ, von Kummer R, Hamann GF. Ischaemic damage of brain microvessels: inherent risks for thrombolytic treatment in stroke. J Neurol Neurosurg Psychiatry. 1998; 65:1–9.
Article14. Wahlgren N, Ahmed N, Eriksson N, Aichner F, Bluhmki E, Dávalos A, et al. Multivariable analysis of outcome predictors and adjustment of main outcome results to baseline data profile in randomized controlled trials: Safe Implementation of Thrombolysis in Stroke-MOnitoring STudy (SITS-MOST). Stroke. 2008; 39:3316–22.15. Okada Y, Yamaguchi T, Minematsu K, Miyashita T, Sawada T, Sadoshima S, et al. Hemorrhagic transformation in cerebral embolism. Stroke. 1989; 20:598–603.
Article16. Nagura J, Suzuki K, Hayashi M, Sakamoto T, Shindo K, Oishi H, et al. Stroke subtypes and lesion sites in Akita, Japan. J Stroke Cerebrovasc Dis. 2005; 14:1–7.
Article17. Wang X, Dong Y, Qi X, Huang C, Hou L. Cholesterol levels and risk of hemorrhagic stroke: a systematic review and meta-analysis. Stroke. 2013; 44:1833–9.18. Konishi M, Iso H, Komachi Y, Iida M, Shimamoto T, Jacobs DR Jr, et al. Associations of serum total cholesterol, different types of stroke, and stenosis distribution of cerebral arteries: the Akita Pathology Study. Stroke. 1993; 24:954–64.
Article19. Kim JH, Bang OY, Liebeskind DS, Ovbiagele B, Kim GM, Chung CS, et al. Impact of baseline tissue status (diffusion-weighted imaging lesion) versus perfusion status (severity of hypoperfusion) on hemorrhagic transformation. Stroke. 2010; 41:e135–42.
Article20. Tong DC, Adami A, Moseley ME, Marks MP. Prediction of hemorrhagic transformation following acute stroke: role of diffusion- and perfusion-weighted magnetic resonance imaging. Arch Neurol. 2001; 58:587–93.
Article21. Wang BG, Yang N, Lin M, Lu B. Analysis of risk factors of hemorrhagic transformation after acute ischemic stroke: cerebral microbleeds do not correlate with hemorrhagic transformation. Cell Biochem Biophys. 2014; 70:135–42.
Article22. Kerenyi L, Kardos L, Szász J, Szatmári S, Bereczki D, Hegedüs K, et al. Factors influencing hemorrhagic transformation in ischemic stroke: a clinicopathological comparison. Eur J Neurol. 2006; 13:1251–5.
Article23. Hafer-Macko CE, Ivey FM, Gyure KA, Sorkin JD, Macko RF. Thrombomodulin deficiency in human diabetic nerve microvasculature. Diabetes. 2002; 51:1957–63.
Article24. Ceriello A, Giugliano D, Quatraro A, Marchi E, Barbanti M, Lefèbvre P. Evidence for a hyperglycaemia-dependent decrease of antithrombin III-thrombin complex formation in humans. Diabetologia. 1990; 33:163–7.
Article25. Ceriello A, Giacomello R, Stel G, Motz E, Taboga C, Tonutti L, et al. Hyperglycemia-induced thrombin formation in diabetes: the possible role of oxidative stress. Diabetes. 1995; 44:924–8.
Article26. Pandolfi A, Cetrullo D, Polishuck R, Alberta MM, Calafiore A, Pellegrini G, et al. Plasminogen activator inhibitor type 1 is increased in the arterial wall of type II diabetic subjects. Arterioscler Thromb Vasc Biol. 2001; 21:1378–82.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Suction thrombectomy of distal medium vessel occlusion using microcatheter during mechanical thrombectomy for acute ischemic stroke: A case series
- Predictors of Catastrophic Outcome after Endovascular Thrombectomy in Elderly Patients with Acute Anterior Circulation Stroke
- Differences in mechanical thrombectomy for acute ischemic stroke on weekdays versus nights/ weekends in a Japanese primary stroke core center
- Intravenous Thrombolysis and Endovascular Thrombectomy in Acute Ischemic Stroke with Minor Symptom
- Endovascular Revascularization Therapy in a Patient with Acute Ischemic Stroke due to Intracranial Multiple Large-vessel Occlusion