Korean J Physiol Pharmacol.
2020 May;24(3):277-286. 10.4196/kjpp.2020.24.3.277.
Ca2+/calmodulin-dependent regulation of polycystic kidney disease 2-like-1 by binding at C-terminal domain
- Affiliations
-
- 1Department of Physiology, Seoul National University College of Medicine, Seoul 03080, Korea
- KMID: 2502510
- DOI: http://doi.org/10.4196/kjpp.2020.24.3.277
Abstract
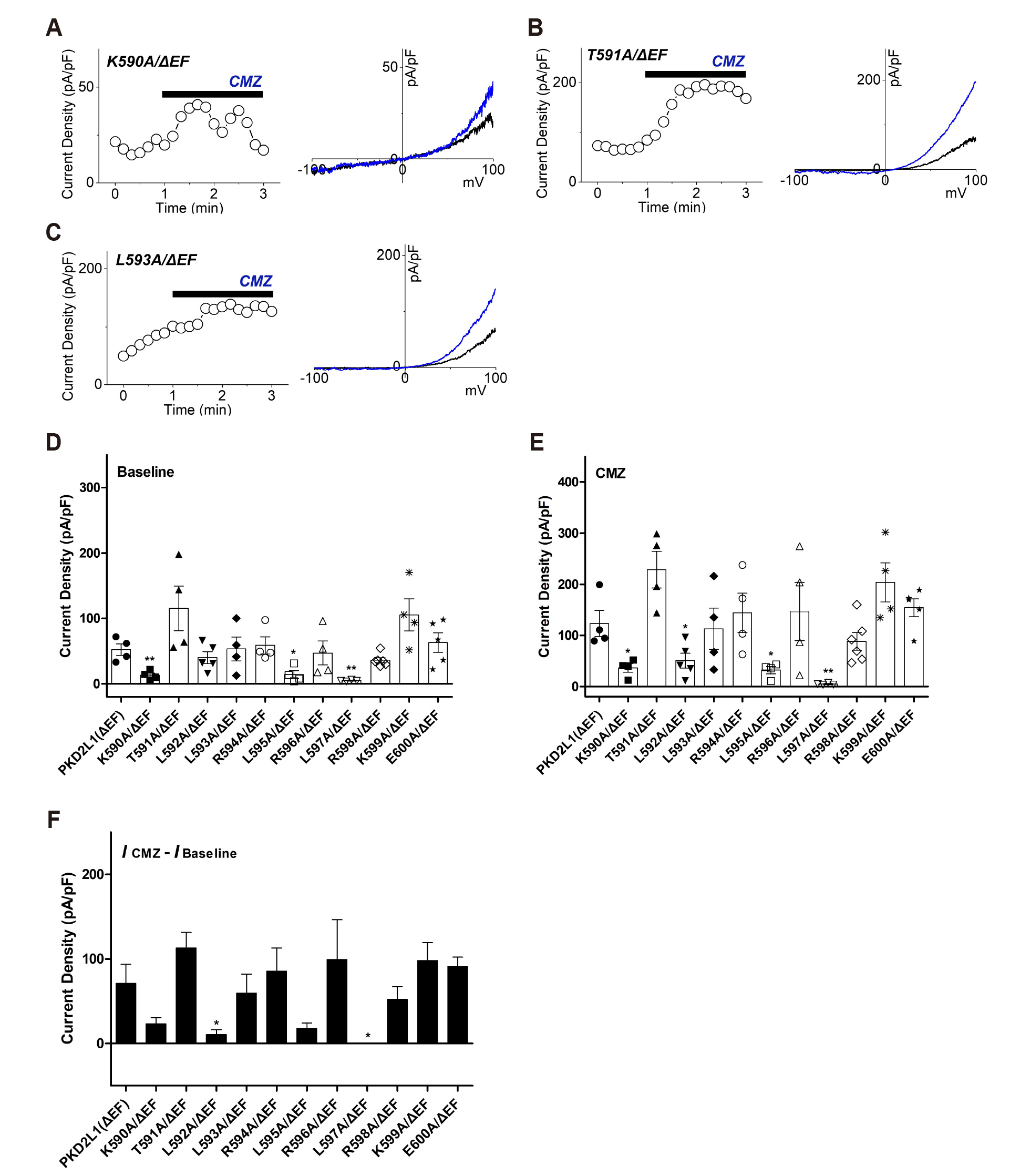
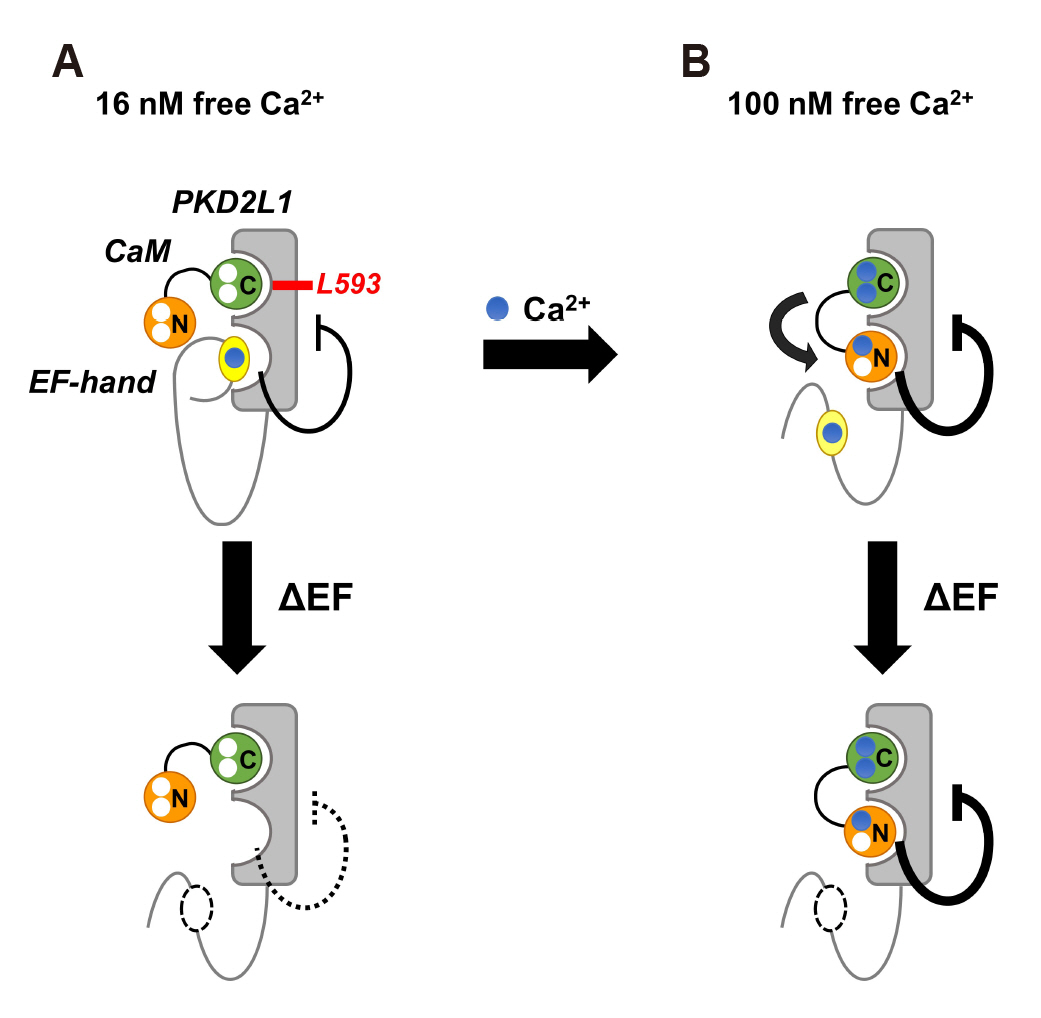
- Polycystic kidney disease 2-like-1 (PKD2L1), also known as polycystin- L or TRPP3, is a non-selective cation channel that regulates intracellular calcium concentration. Calmodulin (CaM) is a calcium binding protein, consisting of N-lobe and C-lobe with two calcium binding EF-hands in each lobe. In previous study, we confirmed that CaM is associated with desensitization of PKD2L1 and that CaM Nlobe and PKD2L1 EF-hand specifically are involved. However, the CaM-binding domain (CaMBD) and its inhibitory mechanism of PKD2L1 have not been identified. In order to identify CaM-binding anchor residue of PKD2L1, single mutants of putative CaMBD and EF-hand deletion mutants were generated. The current changes of the mutants were recorded with whole-cell patch clamp. The calmidazolium (CMZ), a calmodulin inhibitor, was used under different concentrations of intracellular. Among the mutants that showed similar or higher basal currents with that of the PKD2L1 wild type, L593A showed little change in current induced by CMZ. Co-expression of L593A with CaM attenuated the inhibitory effect of PKD2L1 by CaM. In the previous study it was inferred that CaM C-lobe inhibits channels by binding to PKD2L1 at 16 nM calcium concentration and CaM N-lobe at 100 nM. Based on the results at 16 nM calcium concentration condition, this study suggests that CaM C-lobe binds to Leu- 593, which can be a CaM C-lobe anchor residue, to regulate channel activity. Taken together, our results provide a model for the regulation of PKD2L1 channel activity by CaM.
Figure
Reference
-
1. Delling M, DeCaen PG, Doerner JF, Febvay S, Clapham DE. 2013; Primary cilia are specialized calcium signalling organelles. Nature. 504:311–314. DOI: 10.1038/nature12833. PMID: 24336288. PMCID: PMC4112737.
Article2. Sternberg JR, Prendergast AE, Brosse L, Cantaut-Belarif Y, Thouvenin O, Orts-Del'Immagine A, Castillo L, Djenoune L, Kurisu S, McDearmid JR, Bardet PL, Boccara C, Okamoto H, Delmas P, Wyart C. 2018; Pkd2l1 is required for mechanoception in cerebrospinal fluid-contacting neurons and maintenance of spine curvature. Nat Commun. 9:3804. DOI: 10.1038/s41467-018-06225-x. PMID: 30228263. PMCID: PMC6143598.
Article3. DeCaen PG, Delling M, Vien TN, Clapham DE. 2013; Direct recording and molecular identification of the calcium channel of primary cilia. Nature. 504:315–318. DOI: 10.1038/nature12832. PMID: 24336289. PMCID: PMC4073646.
Article4. Ishimaru Y, Inada H, Kubota M, Zhuang H, Tominaga M, Matsunami H. 2006; Transient receptor potential family members PKD1L3 and PKD2L1 form a candidate sour taste receptor. Proc Natl Acad Sci U S A. 103:12569–12574. DOI: 10.1073/pnas.0602702103. PMID: 16891422. PMCID: PMC1531643.
Article5. Zheng W, Hussein S, Yang J, Huang J, Zhang F, Hernandez-Anzaldo S, Fernandez-Patron C, Cao Y, Zeng H, Tang J, Chen XZ. 2015; A novel PKD2L1 C-terminal domain critical for trimerization and channel function. Sci Rep. 5:9460. DOI: 10.1038/srep09460. PMID: 25820328. PMCID: PMC4377555.
Article6. DeCaen PG, Liu X, Abiria S, Clapham DE. 2016; Atypical calcium regulation of the PKD2-L1 polycystin ion channel. Elife. 5:e13413. DOI: 10.7554/eLife.13413. PMID: 27348301. PMCID: PMC4922860.
Article7. Park EYJ, Kwak M, Ha K, So I. 2018; Identification of clustered phosphorylation sites in PKD2L1: how PKD2L1 channel activation is regulated by cyclic adenosine monophosphate signaling pathway. Pflugers Arch. 470:505–516. DOI: 10.1007/s00424-017-2095-7. PMID: 29230552.
Article8. Su Q, Hu F, Liu Y, Ge X, Mei C, Yu S, Shen A, Zhou Q, Yan C, Lei J, Zhang Y, Liu X, Wang T. 2018; Cryo-EM structure of the polycystic kidney disease-like channel PKD2L1. Nat Commun. 9:1192. DOI: 10.1038/s41467-018-03606-0. PMID: 29567962. PMCID: PMC5864754.
Article9. Hulse RE, Li Z, Huang RK, Zhang J, Clapham DE. 2018; Cryo-EM structure of the polycystin 2-l1 ion channel. Elife. 7:e36931. DOI: 10.7554/eLife.36931. PMID: 30004384. PMCID: PMC6056229.
Article10. Chattopadhyaya R, Meador WE, Means AR, Quiocho FA. 1992; Calmodulin structure refined at 1.7 A resolution. J Mol Biol. 228:1177–1192. DOI: 10.1016/0022-2836(92)90324-D. PMID: 1474585.11. Urrutia J, Aguado A, Muguruza-Montero A, Núñez E, Malo C, Casis O, Villarroel A. 2019; The crossroad of ion channels and calmodulin in disease. Int J Mol Sci. 20:E400. DOI: 10.3390/ijms20020400. PMID: 30669290. PMCID: PMC6359610.
Article12. Martin SR, Andersson Teleman A, Bayley PM, Drakenberg T, Forsen S. 1985; Kinetics of calcium dissociation from calmodulin and its tryptic fragments. A stopped-flow fluorescence study using Quin 2 reveals a two-domain structure. Eur J Biochem. 151:543–550. DOI: 10.1111/j.1432-1033.1985.tb09137.x. PMID: 4029146.
Article13. Liu XR, Zhang MM, Rempel DL, Gross ML. 2019; A single approach reveals the composite conformational changes, order of binding, and affinities for calcium binding to calmodulin. Anal Chem. 91:5508–5512. DOI: 10.1021/acs.analchem.9b01062. PMID: 30963760.
Article14. Kawasaki H, Soma N, Kretsinger RH. 2019; Molecular dynamics study of the changes in conformation of calmodulin with calcium binding and/or target recognition. Sci Rep. 9:10688. DOI: 10.1038/s41598-019-47063-1. PMID: 31337841. PMCID: PMC6650393.
Article15. Saimi Y, Kung C. 2002; Calmodulin as an ion channel subunit. Annu Rev Physiol. 64:289–311. DOI: 10.1146/annurev.physiol.64.100301.111649. PMID: 11826271.
Article16. Kovalevskaya NV, van de Waterbeemd M, Bokhovchuk FM, Bate N, Bindels RJ, Hoenderop JG, Vuister GW. 2013; Structural analysis of calmodulin binding to ion channels demonstrates the role of its plasticity in regulation. Pflugers Arch. 465:1507–1519. DOI: 10.1007/s00424-013-1278-0. PMID: 23609407.
Article17. Shah VN, Chagot B, Chazin WJ. 2006; Calcium-dependent regulation of ion channels. Calcium Bind Proteins. 1:203–212. PMID: 28757812. PMCID: PMC5531595.18. Budde T, Meuth S, Pape HC. 2002; Calcium-dependent inactivation of neuronal calcium channels. Nat Rev Neurosci. 3:873–883. DOI: 10.1038/nrn959. PMID: 12415295.
Article19. Lee CH, MacKinnon R. 2018; Activation mechanism of a human SK-calmodulin channel complex elucidated by cryo-EM structures. Science. 360:508–513. DOI: 10.1126/science.aas9466. PMID: 29724949. PMCID: PMC6241251.
Article20. Wang C, Chung BC, Yan H, Wang HG, Lee SY, Pitt GS. 2014; Structural analyses of Ca2+/CaM interaction with NaV channel C-termini reveal mechanisms of calcium-dependent regulation. Nat Commun. 5:4896. DOI: 10.1038/ncomms5896. PMID: 25232683. PMCID: PMC4170523.
Article21. Shah VN, Wingo TL, Weiss KL, Williams CK, Balser JR, Chazin WJ. 2006; Calcium-dependent regulation of the voltage-gated sodium channel hH1: intrinsic and extrinsic sensors use a common molecular switch. Proc Natl Acad Sci U S A. 103:3592–3597. DOI: 10.1073/pnas.0507397103. PMID: 16505387. PMCID: PMC1450128.
Article22. Ben Johny M, Yang PS, Bazzazi H, Yue DT. 2013; Dynamic switching of calmodulin interactions underlies Ca2+ regulation of CaV1.3 channels. Nat Commun. 4:1717. DOI: 10.1038/ncomms2727. PMID: 23591884. PMCID: PMC3856249.
Article23. Gordon-Shaag A, Zagotta WN, Gordon SE. 2008; Mechanism of Ca2+-dependent desensitization in TRP channels. Channels (Austin). 2:125–129. DOI: 10.4161/chan.2.2.6026. PMID: 18849652.24. Hasan R, Leeson-Payne AT, Jaggar JH, Zhang X. 2017; Calmodulin is responsible for Ca2+-dependent regulation of TRPA1 channels. Sci Rep. 7:45098. DOI: 10.1038/srep45098. PMID: 28332600. PMCID: PMC5362816.
Article25. Zhu MX. 2005; Multiple roles of calmodulin and other Ca2+-binding proteins in the functional regulation of TRP channels. Pflugers Arch. 451:105–115. DOI: 10.1007/s00424-005-1427-1. PMID: 15924238.26. Polat OK, Uno M, Maruyama T, Tran HN, Imamura K, Wong CF, Sakaguchi R, Ariyoshi M, Itsuki K, Ichikawa J, Morii T, Shirakawa M, Inoue R, Asanuma K, Reiser J, Tochio H, Mori Y, Mori MX. 2019; Contribution of coiled-coil assembly to Ca2+/calmodulin-dependent inactivation of TRPC6 channel and its impacts on FSGS-associated phenotypes. J Am Soc Nephrol. 30:1587–1603. DOI: 10.1681/ASN.2018070756. PMID: 31266820.27. Dang S, van Goor MK, Asarnow D, Wang Y, Julius D, Cheng Y, van der Wijst J. 2019; Structural insight into TRPV5 channel function and modulation. Proc Natl Acad Sci U S A. 116:8869–8878. DOI: 10.1073/pnas.1820323116. PMID: 30975749. PMCID: PMC6500171.
Article28. Singh AK, McGoldrick LL, Twomey EC, Sobolevsky AI. 2018; Mechanism of calmodulin inactivation of the calcium-selective TRP channel TRPV6. Sci Adv. 4:eaau6088. DOI: 10.1126/sciadv.aau6088. PMID: 30116787. PMCID: PMC6093632.
Article29. Park EYJ, Baik JY, Kwak M, So I. 2019; The role of calmodulin in regulating calcium-permeable PKD2L1 channel activity. Korean J Physiol Pharmacol. 23:219–227. DOI: 10.4196/kjpp.2019.23.3.219. PMID: 31080352. PMCID: PMC6488707.
Article30. Hoeflich KP, Ikura M. 2002; Calmodulin in action: diversity in target recognition and activation mechanisms. Cell. 108:739–742. DOI: 10.1016/S0092-8674(02)00682-7. PMID: 11955428.31. Tidow H, Nissen P. 2013; Structural diversity of calmodulin binding to its target sites. FEBS J. 280:5551–5565. DOI: 10.1111/febs.12296. PMID: 23601118.
Article32. Mruk K, Farley BM, Ritacco AW, Kobertz WR. 2014; Calmodulation meta-analysis: predicting calmodulin binding via canonical motif clustering. J Gen Physiol. 144:105–114. DOI: 10.1085/jgp.201311140. PMID: 24935744. PMCID: PMC4076516.
Article33. Sunagawa M, Kosugi T, Nakamura M, Sperelakis N. 2000; Pharmacological actions of calmidazolium, a calmodulin antagonist, in cardiovascular system. Cardiovasc Drug Rev. 18:211–221. DOI: 10.1111/j.1527-3466.2000.tb00044.x.
Article34. Kumar S, Kain V, Sitasawad SL. 2009; Cardiotoxicity of calmidazolium chloride is attributed to calcium aggravation, oxidative and nitrosative stress, and apoptosis. Free Radic Biol Med. 47:699–709. DOI: 10.1016/j.freeradbiomed.2009.05.028. PMID: 19497364.
Article35. Lau SY, Procko E, Gaudet R. 2012; Distinct properties of Ca2+-calmodulin binding to N- and C-terminal regulatory regions of the TRPV1 channel. J Gen Physiol. 140:541–555. DOI: 10.1085/jgp.201210810. PMID: 23109716. PMCID: PMC3483115.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Regulation of GTP-binding state in RalA through Ca2+ and calmodulin
- The role of calmodulin in regulating calcium-permeable PKD2L1 channel activity
- A Memory Molecule, Ca2+/Calmodulin-Dependent Protein Kinase II and Redox Stress; Key Factors for Arrhythmias in a Diseased Heart
- Role of Calmodulin in the Generation of Reactive Oxygen Species and Apoptosis Induced by Tamoxifen in HepG2 Human Hepatoma Cells
- Autosomal Dominant Polycystic Kidney Desease Coexisting with Renal Dysplasia. First Case Described and Followed Since Prenatal Period