Yonsei Med J.
2020 Apr;61(4):284-290. 10.3349/ymj.2020.61.4.284.
A Single-Center, Retrospective Study of Bevacizumab-Containing Neoadjuvant Chemotherapy followed by Interval Debulking Surgery for Ovarian Cancer
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Institute of Women's Life Medical Science, Yonsei University College of Medicine, Seoul, Korea. jungyunlee@yuhs.ac
- KMID: 2471913
- DOI: http://doi.org/10.3349/ymj.2020.61.4.284
Abstract
- PURPOSE
We evaluated whether adding bevacizumab to current platinum-based chemotherapy could improve clinical outcomes without affecting safety.
MATERIALS AND METHODS
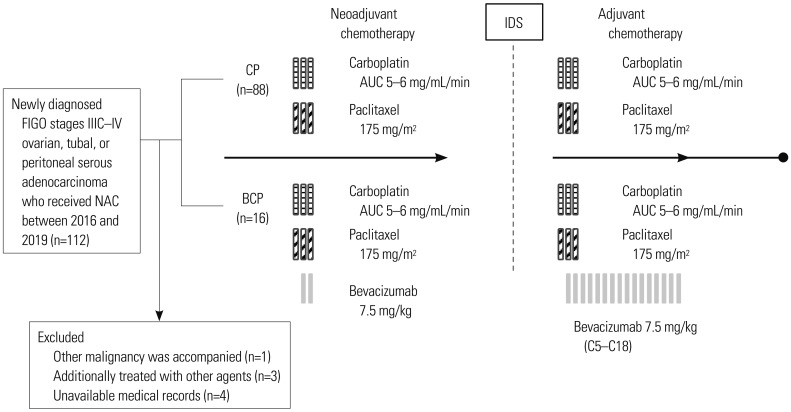
We retrospectively reviewed medical records of patients with pathologically confirmed ovarian cancer who received neoadjuvant chemotherapy (NAC) at Yonsei Cancer Hospital. We divided the patients into groups based on the use of bevacizumab for NAC (CP group: carboplatin+paclitaxel vs. BCP group: bevacizumab+carboplatin+paclitaxel) and compared patient characteristics, responses to NAC, and surgical and survival outcomes between the two groups. Overall, 88 patients in the CP group and 16 patients in the BCP group received NAC. The primary endpoint was survival outcomes. Complete resection rate after interval debulking surgery (IDS), cancer antigen 125 (CA-125) normalization after NAC, and chemotherapy response score were secondary endpoints.
RESULTS
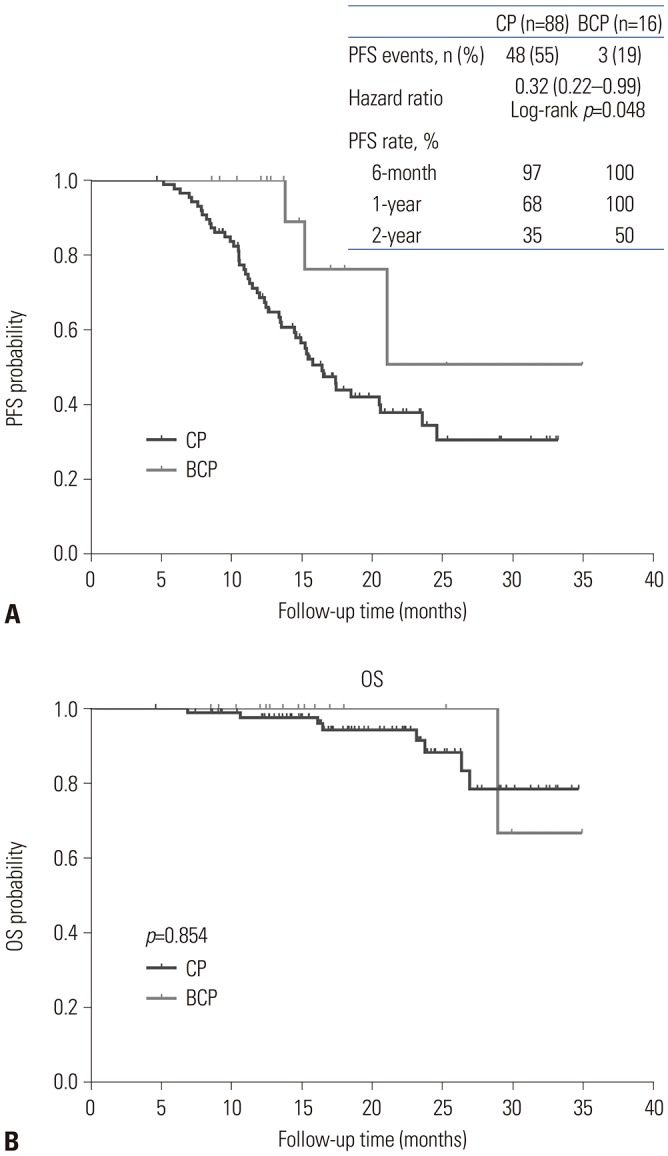
After NAC treatment, all patients underwent IDS. There were no significant differences in adverse events during NAC or postoperative complications between the two groups (p=0.293 and p=0.485, respectively). There were also no significant differences in CA-125 normalization after NAC (42.0% vs. 43.8%, p=0.899) or complete resection rate after IDS (47.7% vs. 56.3%, p=0.530). However, although the BCP group did not show longer overall survival (OS) (log-rank p=0.854), they had significantly longer progression-free survival (PFS) than the CP group (log-rank p=0.048).
CONCLUSION
Bevacizumab-containing NAC might be safe and provide longer PFS than chemotherapy alone in patients with advanced ovarian cancer. However, further study is necessary to investigate the impact of bevacizumab-containing NAC on OS.
Keyword
MeSH Terms
Figure
Reference
-
1. Vergote I, Tropé CG, Amant F, Kristensen GB, Ehlen T, Johnson N, et al. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N Engl J Med. 2010; 363:943–953. PMID: 20818904.
Article2. Kehoe S, Hook J, Nankivell M, Jayson GC, Kitchener H, Lopes T, et al. Primary chemotherapy versus primary surgery for newly diagnosed advanced ovarian cancer (CHORUS): an open-label, randomised, controlled, non-inferiority trial. Lancet. 2015; 386:249–257. PMID: 26002111.
Article3. Onda T, Satoh T, Saito T, Kasamatsu T, Nakanishi T, Nakamura K, et al. Comparison of treatment invasiveness between upfront debulking surgery versus interval debulking surgery following neoadjuvant chemotherapy for stage III/IV ovarian, tubal, and peritoneal cancers in a phase III randomised trial: Japan Clinical Oncology Group Study JCOG0602. Eur J Cancer. 2016; 64:22–31. PMID: 27323348.
Article4. Wright AA, Bohlke K, Armstrong DK, Bookman MA, Cliby WA, Coleman RL, et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: Society of Gynecologic Oncology and American Society of Clinical Oncology Clinical Practice Guideline. Gynecol Oncol. 2016; 143:3–15. PMID: 27650684.
Article5. Perren TJ, Swart AM, Pfisterer J, Ledermann JA, Pujade-Lauraine E, Kristensen G, et al. A phase 3 trial of bevacizumab in ovarian cancer. N Engl J Med. 2011; 365:2484–2496. PMID: 22204725.
Article6. Burger RA, Brady MF, Bookman MA, Fleming GF, Monk BJ, Huang H, et al. Incorporation of bevacizumab in the primary treatment of ovarian cancer. N Engl J Med. 2011; 365:2473–2483. PMID: 22204724.
Article7. Rouzier R, Gouy S, Selle F, Lambaudie E, Floquet A, Fourchotte V, et al. Efficacy and safety of bevacizumab-containing neoadjuvant therapy followed by interval debulking surgery in advanced ovarian cancer: results from the ANTHALYA trial. Eur J Cancer. 2017; 70:133–142. PMID: 27914243.
Article8. Lee YJ, Lee JY, Cho MS, Nam EJ, Kim SW, Kim S, et al. Incorporation of paclitaxel-based hyperthermic intraperitoneal chemotherapy in patients with advanced-stage ovarian cancer treated with neoadjuvant chemotherapy followed by interval debulking surgery: a protocol-based pilot study. J Gynecol Oncol. 2019; 30:e3. PMID: 30479087.
Article9. Fagotti A, Vizzielli G, Fanfani F, Costantini B, Ferrandina G, Gallotta V, et al. Introduction of staging laparoscopy in the management of advanced epithelial ovarian, tubal and peritoneal cancer: impact on prognosis in a single institution experience. Gynecol Oncol. 2013; 131:341–346. PMID: 23938372.
Article10. Fagotti A, Ferrandina G, Fanfani F, Garganese G, Vizzielli G, Carone V, et al. Prospective validation of a laparoscopic predictive model for optimal cytoreduction in advanced ovarian carcinoma. Am J Obstet Gynecol. 2008; 199:642.e1–642.e6. PMID: 18801470.
Article11. Chi DS, Eisenhauer EL, Zivanovic O, Sonoda Y, Abu-Rustum NR, Levine DA, et al. Improved progression-free and overall survival in advanced ovarian cancer as a result of a change in surgical paradigm. Gynecol Oncol. 2009; 114:26–31. PMID: 19395008.
Article12. Wimberger P, Lehmann N, Kimmig R, Burges A, Meier W, Du Bois A. Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Cancer Study Group. Prognostic factors for complete debulking in advanced ovarian cancer and its impact on survival. An exploratory analysis of a prospectively randomized phase III study of the Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Cancer Study Group (AGO-OVAR). Gynecol Oncol. 2007; 106:69–74. PMID: 17397910.
Article13. Eisenhauer EL, Abu-Rustum NR, Sonoda Y, Levine DA, Poynor EA, Aghajanian C, et al. The addition of extensive upper abdominal surgery to achieve optimal cytoreduction improves survival in patients with stages IIIC-IV epithelial ovarian cancer. Gynecol Oncol. 2006; 103:1083–1090. PMID: 16890277.
Article14. Strong VE, Selby LV, Sovel M, Disa JJ, Hoskins W, Dematteo R, et al. Development and assessment of Memorial Sloan Kettering Cancer Center's Surgical Secondary Events grading system. Ann Surg Oncol. 2015; 22:1061–1067. PMID: 25319579.
Article15. Chi DS, Franklin CC, Levine DA, Akselrod F, Sabbatini P, Jarnagin WR, et al. Improved optimal cytoreduction rates for stages IIIC and IV epithelial ovarian, fallopian tube, and primary peritoneal cancer: a change in surgical approach. Gynecol Oncol. 2004; 94:650–654. PMID: 15350354.
Article16. Coleman RL, Brady MF, Herzog TJ, Sabbatini P, Armstrong DK, Walker JL, et al. Bevacizumab and paclitaxel-carboplatin chemotherapy and secondary cytoreduction in recurrent, platinum-sensitive ovarian cancer (NRG Oncology/Gynecologic Oncology Group study GOG-0213): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2017; 18:779–791. PMID: 28438473.
Article17. Aghajanian C, Blank SV, Goff BA, Judson PL, Teneriello MG, Husain A, et al. OCEANS: a randomized, double-blind, placebo-controlled phase III trial of chemotherapy with or without bevacizumab in patients with platinum-sensitive recurrent epithelial ovarian, primary peritoneal, or fallopian tube cancer. J Clin Oncol. 2012; 30:2039–2045. PMID: 22529265.
Article18. Pujade-Lauraine E, Hilpert F, Weber B, Reuss A, Poveda A, Kristensen G, et al. Bevacizumab combined with chemotherapy for platinum-resistant recurrent ovarian cancer: the AURELIA openlabel randomized phase III trial. J Clin Oncol. 2014; 32:1302–1308. PMID: 24637997.
Article19. Oza AM, Cook AD, Pfisterer J, Embleton A, Ledermann JA, Pujade-Lauraine E, et al. Standard chemotherapy with or without bevacizumab for women with newly diagnosed ovarian cancer (ICON7): overall survival results of a phase 3 randomised trial. Lancet Oncol. 2015; 16:928–936. PMID: 26115797.
Article20. Daniele G, Lorusso D, Scambia G, Cecere SC, Nicoletto MO, Breda E, et al. Feasibility and outcome of interval debulking surgery (IDS) after carboplatin-paclitaxel-bevacizumab (CPB): a subgroup analysis of the MITO-16A-MaNGO OV2A phase 4 trial. Gynecol Oncol. 2017; 144:256–259. PMID: 27993479.
Article21. Garcia Garcia Y, de Juan Ferré A, Mendiola C, Barretina-Ginesta MP, Gaba Garcia L, Santaballa Bertrán A, et al. Efficacy and safety results from GEICO 1205, a randomized phase II trial of neoadjuvant chemotherapy with or without bevacizumab for advanced epithelial ovarian cancer. Int J Gynecol Cancer. 2019; 29:1050–1056. PMID: 31263024.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Neoadjuvant Chemotherapy in Patients with Advanced Epithelial Ovarian Cancer
- Negative peritoneal washing cytology during interval debulking surgery predicts overall survival after neoadjuvant chemotherapy for ovarian cancer
- Neoadjuvant chemotherapy in ovarian, primary peritoneal and tubal carcinoma: can imaging results prior to interval debulking predict survival?
- CA-125 cut-off value as a predictor for complete interval debulking surgery after neoadjuvant chemotherapy in patients with advanced ovarian cancer
- Treatment strategies for patients with advanced ovarian cancer undergoing neoadjuvant chemotherapy: interval debulking surgery or additional chemotherapy?