World J Mens Health.
2020 Apr;38(2):208-219. 10.5534/wjmh.180124.
Diagnostic Performance of 68Gallium Labelled Prostate-Specific Membrane Antigen Positron Emission Tomography/Computed Tomography and Magnetic Resonance Imaging for Staging the Prostate Cancer with Intermediate or High Risk Prior to Radical Prostatectomy: A Systematic Review and Meta-analysis
- Affiliations
-
- 1Department of Urology, Affiliated Hospital of Qingdao University, Qingdao, China. niuht0532@126.com, jiaowei3929@163.com
- 2Key Laboratory of Urinary System Diseases, Qingdao, China.
- 3Department of Geratology, The 971th Hospital of PLA, Qingdao, China.
- KMID: 2471827
- DOI: http://doi.org/10.5534/wjmh.180124
Abstract
- PURPOSE
To compare the diagnostic efficiency of â¶â¸Gallium labelled prostate-specific membrane antigen positron emission tomography (â¶â¸Ga-PSMA PET) and magnetic resonance imaging (MRI) for staging the lymph node metastases (LNMs) in the prostate cancer.
MATERIALS AND METHODS
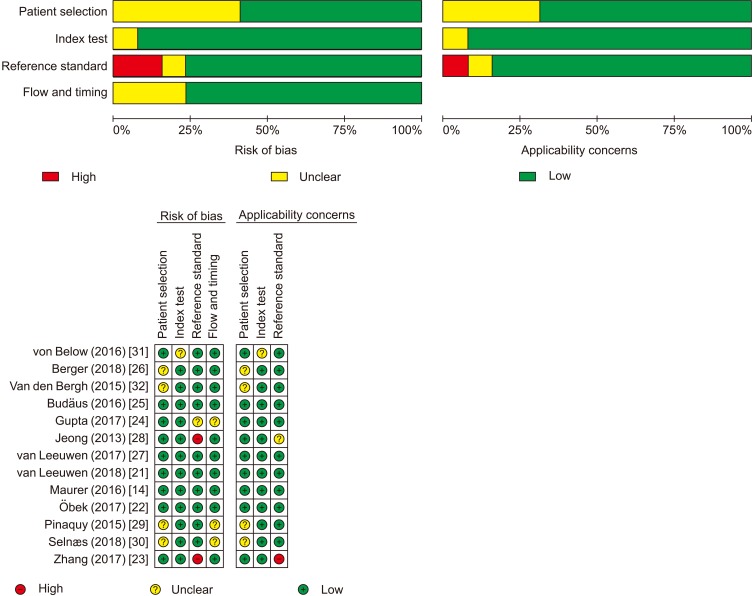
A broad search of scientific databases including PubMed, EMBASE, Web of Science, Cochrane Database, and Chinese Biomedicine Literature Database (updated prior to November 1st, 2018) was conducted systematically by two reviewers. In this paper, we evaluated the methodological quality of each included article independently and performed a systematic review and meta-analysis to reveal the summary of the diagnostic performance of â¶â¸Ga-PSMA PET and MRI in properly identifying LNMs of intermediate- and/or high-risk prostate cancer.
RESULTS
Thirteen eligible articles comprising 1,597 patients were included. For LNMs detection, the pooled sensitivity and specificity of â¶â¸Ga-PSMA PET were 0.65 (95% confidence interval [CI]: 0.49-0.79) and 0.94 (95% CI: 0.88-0.97), respectively, while the corresponding values of MRI were 0.41 (95% CI: 0.26-0.57) and 0.92 (95% CI: 0.86-0.95). The area under the symmetric receiver-operating characteristic (SROC) curve for â¶â¸Ga-PSMA PET and MRI were 0.92 and 0.83, respectively.
CONCLUSIONS
In intermediate- or high-risk pre-treatment prostate cancer, â¶â¸Ga-PSMA PET had a higher sensitivity and a slightly different specificity in probing the LNMs when comparing with MRI. Moreover, the area under the SROC curve indicated that â¶â¸Ga-PSMA PET was a more effective weapon for predicting the LNMs prior to radical surgery.
Keyword
MeSH Terms
Figure
Reference
-
1. Babb C, Urban M, Kielkowski D, Kellett P. Prostate cancer in South Africa: pathology based national cancer registry data (1986-2006) and mortality rates (1997-2009). Prostate Cancer. 2014; 2014:419801. PMID: 24955252.
Article2. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018; 68:394–424. PMID: 30207593.
Article3. Loeb S, Smith ND, Roehl KA, Catalona WJ. Intermediate-term potency, continence, and survival outcomes of radical prostatectomy for clinically high-risk or locally advanced prostate cancer. Urology. 2007; 69:1170–1175. PMID: 17572209.
Article4. Bartsch G, Horninger W, Klocker H, Reissigl A, Oberaigner W, Schönitzer D, et al. Prostate cancer mortality after introduction of prostate-specific antigen mass screening in the Federal State of Tyrol, Austria. Urology. 2001; 58:417–424. PMID: 11549491.
Article5. de Rooij M, Hamoen EH, Witjes JA, Barentsz JO, Rovers MM. Accuracy of magnetic resonance imaging for local staging of prostate cancer: a diagnostic meta-analysis. Eur Urol. 2016; 70:233–245. PMID: 26215604.
Article6. Bostwick DG, Pacelli A, Blute M, Roche P, Murphy GP. Prostate specific membrane antigen expression in prostatic intraepithelial neoplasia and adenocarcinoma: a study of 184 cases. Cancer. 1998; 82:2256–2261. PMID: 9610707.7. Silver DA, Pellicer I, Fair WR, Heston WD, Cordon-Cardo C. Prostate-specific membrane antigen expression in normal and malignant human tissues. Clin Cancer Res. 1997; 3:81–85. PMID: 9815541.8. Minner S, Wittmer C, Graefen M, Salomon G, Steuber T, Haese A, et al. High level PSMA expression is associated with early PSA recurrence in surgically treated prostate cancer. Prostate. 2011; 71:281–288. PMID: 20809553.
Article9. Jadvar H. positron emission tomography in prostate cancer: summary of systematic reviews and meta-analysis. Tomography. 2015; 1:18–22. PMID: 26726317.10. Pfob CH, Ziegler S, Graner FP, Köhner M, Schachoff S, Blechert B, et al. Biodistribution and radiation dosimetry of (68) Ga-PSMA HBED CC-a PSMA specific probe for PET imaging of prostate cancer. Eur J Nucl Med Mol Imaging. 2016; 43:1962–1970. PMID: 27207281.11. Herrmann K, Bluemel C, Weineisen M, Schottelius M, Wester HJ, Czernin J, et al. Biodistribution and radiation dosimetry for a probe targeting prostate-specific membrane antigen for imaging and therapy. J Nucl Med. 2015; 56:855–861. PMID: 25883128.
Article12. Afshar-Oromieh A, Zechmann CM, Malcher A, Eder M, Eisenhut M, Linhart HG, et al. Comparison of PET imaging with a (68)Ga-labelled PSMA ligand and (18)F-choline-based PET/CT for the diagnosis of recurrent prostate cancer. Eur J Nucl Med Mol Imaging. 2014; 41:11–20. PMID: 24072344.
Article13. Herlemann A, Wenter V, Kretschmer A, Thierfelder KM, Bartenstein P, Faber C, et al. (68)Ga-PSMA positron emission tomography/computed tomography provides accurate staging of lymph node regions prior to lymph node dissection in patients with prostate cancer. Eur Urol. 2016; 70:553–557. PMID: 26810345.
Article14. Maurer T, Gschwend JE, Rauscher I, Souvatzoglou M, Haller B, Weirich G, et al. Diagnostic efficacy of (68)Gallium-PSMA positron emission tomography compared to conventional imaging for lymph node staging of 130 consecutive patients with intermediate to high risk prostate cancer. J Urol. 2016; 195:1436–1443. PMID: 26682756.15. Perera M, Papa N, Christidis D, Wetherell D, Hofman MS, Murphy DG, et al. Sensitivity, specificity, and predictors of positive (68)Ga-Prostate-specific membrane antigen positron emission tomography in advanced prostate cancer: a systematic review and meta-analysis. Eur Urol. 2016; 70:926–937. PMID: 27363387.16. Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009; 339:b2700. PMID: 19622552.
Article17. Whiting PF, Rutjes AW, Westwood ME, Mallett S, Deeks JJ, Reitsma JB, et al. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med. 2011; 155:529–536. PMID: 22007046.
Article18. Van Houwelingen HC, Zwinderman KH, Stijnen T. A bivariate approach to meta-analysis. Stat Med. 1993; 12:2273–2284. PMID: 7907813.
Article19. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003; 327:557–560. PMID: 12958120.
Article20. Deeks JJ, Macaskill P, Irwig L. The performance of tests of publication bias and other sample size effects in systematic reviews of diagnostic test accuracy was assessed. J Clin Epidemiol. 2005; 58:882–893. PMID: 16085191.
Article21. van Leeuwen PJ, Donswijk M, Nandurkar R, Stricker P, Ho B, Heijmink S, et al. 68Ga PSMA PET/CT predicts complete biochemical response from radical prostatectomy and lymph node dissection in intermediate and high-risk prostate cancer. BJU Int. 2018; DOI: 10.1111/bju.14506. [Epub].22. Öbek C, Doğanca T, Demirci E, Ocak M, Kural AR, Yıldırım A, et al. The accuracy of 68Ga-PSMA PET/CT in primary lymph node staging in high-risk prostate cancer. Eur J Nucl Med Mol Imaging. 2017; 44:1806–1812. PMID: 28624849.
Article23. Zhang Q, Zang S, Zhang C, Fu Y, Lv X, Zhang Q, et al. Comparison of 68Ga-PSMA-11 PET-CT with mpMRI for preoperative lymph node staging in patients with intermediate to high-risk prostate cancer. J Transl Med. 2017; 15:230. PMID: 29115970.
Article24. Gupta M, Choudhury PS, Hazarika D, Rawal S. A comparative study of 68gallium-prostate specific membrane antigen positron emission tomography-computed tomography and magnetic resonance imaging for lymph node staging in high risk prostate cancer patients: an initial experience. World J Nucl Med. 2017; 16:186–191. PMID: 28670175.
Article25. Budäus L, Leyh-Bannurah SR, Salomon G, Michl U, Heinzer H, Huland H, et al. Initial experience of (68)Ga-PSMA PET/ct imaging in high-risk prostate cancer patients prior to radical prostatectomy. Eur Urol. 2016; 69:393–396. PMID: 26116958.
Article26. Berger I, Annabattula C, Lewis J, Shetty DV, Kam J, Maclean F, et al. 68Ga-PSMA PET/CT vs. mpMRI for locoregional prostate cancer staging: correlation with final histopathology. Prostate Cancer Prostatic Dis. 2018; 21:204–211. PMID: 29858591.27. van Leeuwen PJ, Emmett L, Ho B, Delprado W, Ting F, Nguyen Q, et al. Prospective evaluation of 68Gallium-prostate-specific membrane antigen positron emission tomography/computed tomography for preoperative lymph node staging in prostate cancer. BJU Int. 2017; 119:209–215. PMID: 27207581.
Article28. Jeong IG, Lim JH, You D, Kim MH, Choi HJ, Kim JK, et al. Incremental value of magnetic resonance imaging for clinically high risk prostate cancer in 922 radical prostatectomies. J Urol. 2013; 190:2054–2060. PMID: 23791890.
Article29. Pinaquy JB, De Clermont-Galleran H, Pasticier G, Rigou G, Alberti N, Hindie E, et al. Comparative effectiveness of [(18) F]-fluorocholine PET-CT and pelvic MRI with diffusion-weighted imaging for staging in patients with high-risk prostate cancer. Prostate. 2015; 75:323–331. PMID: 25393215.30. Selnæs KM, Krüger-Stokke B, Elschot M, Willoch F, Størkersen Ø, Sandsmark E, et al. 18F-Fluciclovine PET/MRI for preoperative lymph node staging in high-risk prostate cancer patients. Eur Radiol. 2018; 28:3151–3159. PMID: 29294158.
Article31. von Below C, Daouacher G, Wassberg C, Grzegorek R, Gestblom C, Sörensen J, et al. Validation of 3 T MRI including diffusion-weighted imaging for nodal staging of newly diagnosed intermediate- and high-risk prostate cancer. Clin Radiol. 2016; 71:328–334. PMID: 26774372.32. Van den Bergh L, Lerut E, Haustermans K, Deroose CM, Oyen R, Isebaert S, et al. Final analysis of a prospective trial on functional imaging for nodal staging in patients with prostate cancer at high risk for lymph node involvement. Urol Oncol. 2015; 33:109.e23–109.e31.
Article33. Van Poppel H, Joniau S. An analysis of radical prostatectomy in advanced stage and high-grade prostate cancer. Eur Urol. 2008; 53:253–259. PMID: 17949893.
Article34. Hövels AM, Heesakkers RA, Adang EM, Jager GJ, Strum S, Hoogeveen YL, et al. The diagnostic accuracy of CT and MRI in the staging of pelvic lymph nodes in patients with prostate cancer: a meta-analysis. Clin Radiol. 2008; 63:387–395. PMID: 18325358.
Article35. Hricak H, Dooms GC, Jeffrey RB, Avallone A, Jacobs D, Benton WK, et al. Prostatic carcinoma: staging by clinical assessment, CT, and MR imaging. Radiology. 1987; 162:331–336. PMID: 3797645.
Article36. Heesakkers RA, Hövels AM, Jager GJ, van den Bosch HC, Witjes JA, Raat HP, et al. MRI with a lymph-node-specific contrast agent as an alternative to CT scan and lymph-node dissection in patients with prostate cancer: a prospective multicohort study. Lancet Oncol. 2008; 9:850–856. PMID: 18708295.
Article37. Hofman MS, Murphy DG, Williams SG, Nzenza T, Herschtal A, Lourenco RA, et al. A prospective randomized multicentre study of the impact of gallium-68 prostate-specific membrane antigen (PSMA) PET/CT imaging for staging high-risk prostate cancer prior to curative-intent surgery or radiotherapy (proPSMA study): clinical trial protocol. BJU Int. 2018; 122:783–793. PMID: 29726071.
Article38. Hijazi S, Meller B, Leitsmann C, Strauss A, Meller J, Ritter CO, et al. Pelvic lymph node dissection for nodal oligometastatic prostate cancer detected by 68Ga-PSMA-positron emission tomography/computerized tomography. Prostate. 2015; 75:1934–1940. PMID: 26356236.
Article39. Hamed MAG, Basha MAA, Ahmed H, Obaya AA, Afifi AHM, Abdelbary EH. 68Ga-PSMA PET/CT in patients with rising prostatic-specific antigen after definitive treatment of prostate cancer: detection efficacy and diagnostic accuracy. Acad Radiol. 2018; DOI: 10.1016/j.acra.2018.05.020. [Epub].
Article40. Rauscher I, Maurer T, Beer AJ, Graner FP, Haller B, Weirich G, et al. Value of 68Ga-PSMA HBED-CC PET for the assessment of lymph node metastases in prostate cancer patients with biochemical recurrence: comparison with histopathology after salvage lymphadenectomy. J Nucl Med. 2016; 57:1713–1719. PMID: 27261524.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Updates of prostate cancer staging: Prostate-specific membrane antigen
- Role of Pelvic Lymph Node Dissection in Prostate Cancer Treatment
- An Update of Prostate-Specific Membrane Antigen Theranostics in Prostate Cancer
- Performance and Impact of Prostate Specific Membrane Antigen-Based Diagnostics in the Management of Men with Biochemical Recurrence of Prostate Cancer and its Role in Salvage Lymph Node Dissection
- Radical Prostatectomy