Korean J Radiol.
2020 Feb;21(2):228-235. 10.3348/kjr.2019.0188.
Alpha Stent for Coiling of Unruptured, Wide-Necked, Distal Internal Carotid Artery Aneurysms: Safety and Effectiveness at 6 Months
- Affiliations
-
- 1Department of Radiology, University of Ulsan College of Medicine, Asan Medical Center, Seoul, Korea. dhlee@amc.seoul.kr
- 2Department of Neurosurgery, University of Ulsan College of Medicine, Asan Medical Center, Seoul, Korea.
- 3Department of Neurology, Jeju National University Hospital, Jeju, Korea.
- 4Department of Neurology, Korea University Ansan Hospital, Ansan, Korea.
- 5Department of Neurosurgery, Gangneung Asan Hospital, Gangneung, Korea.
- KMID: 2471640
- DOI: http://doi.org/10.3348/kjr.2019.0188
Abstract
OBJECTIVE
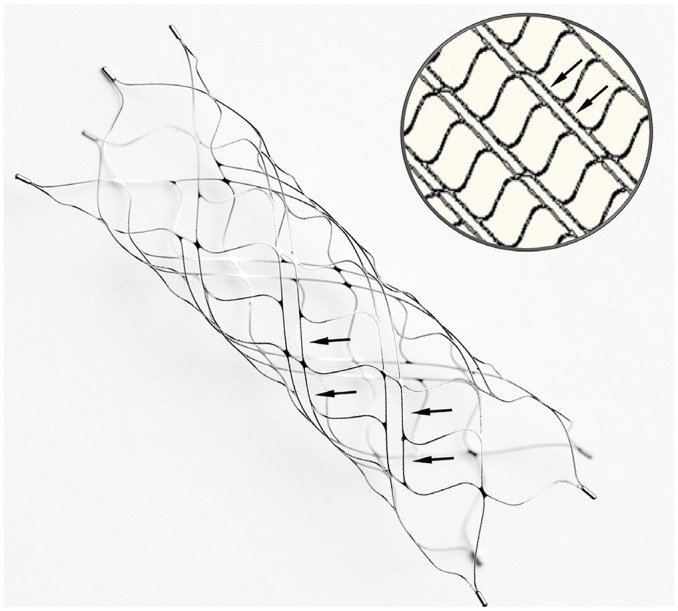
The Alpha stent (CGBio), a new intracranial stent featuring a re-sheathable mesh design with improved wall apposition at the curved segment, was clinically evaluated. We report the 6-month follow-up results from a prospective, single-center study in which the stent was used for coiling of wide-necked distal internal carotid artery (ICA) aneurysms.
MATERIALS AND METHODS
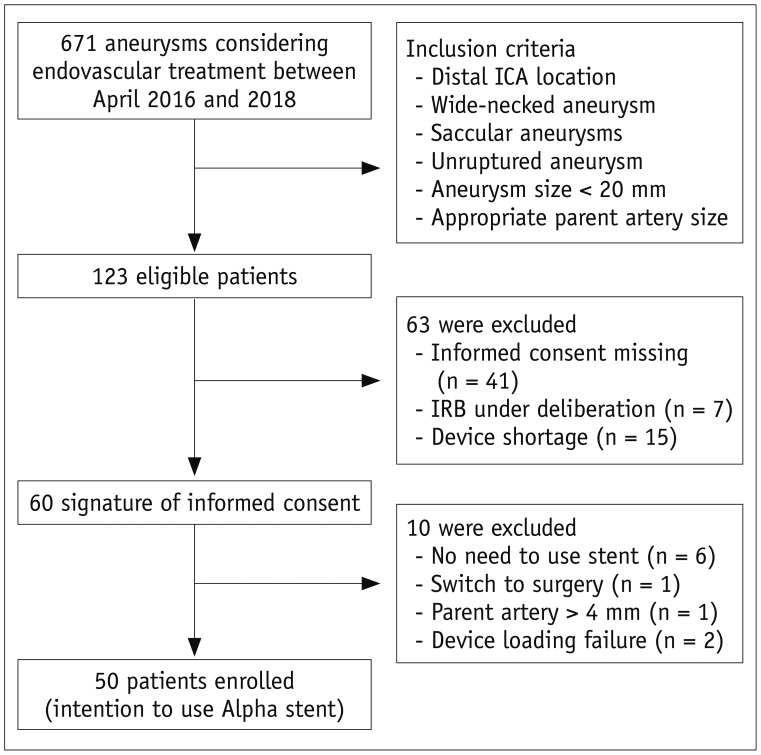
Between April 2016 and 2018, 50 patients (mean age, 56.5 years, 45 females [90%]) with 54 unruptured distal ICA aneurysms (average diameter: 5.6 ± 1.7 mm) were enrolled. The primary endpoint for effectiveness was successful coil embolization with the Alpha stent, and subsequent complete or near-complete occlusion at the 6-month magnetic resonance angiography assessment. The primary safety endpoint was the absence of serious adverse events (SAEs) up to 6 months from the procedure.
RESULTS
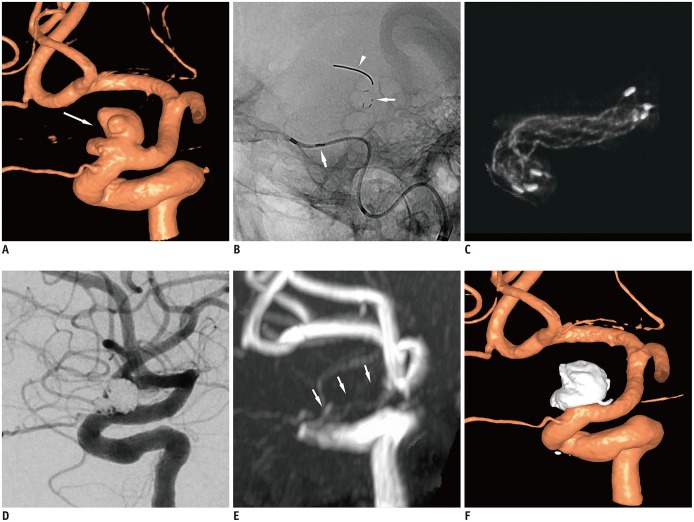
The primary effectiveness endpoint was observed in 94.4% (51/54) aneurysms. In one patient with technical failure, the stent could not be deployed because of parent artery tortuosity; therefore, a different type of stent was used. Of the 53 aneurysms treated with the Alpha stent, complete occlusion was achieved in 64.1% (34/53) cases, and near-complete occlusion was achieved in 32.0% (17/53) cases by the 6-month follow-up. Two cases (3.7%) required retreatment because of major recurrence. In 4% (2/50) patients, SAEs, i.e., retinal artery thromboembolism and corona radiata lacunar infarction, were reported after the procedure.
CONCLUSION
For endovascular treatment of unruptured, wide-necked, distal ICA aneurysms, coil embolization using the newly developed Alpha stent showed excellent procedural and mid-term clinical follow-up results in terms of effectiveness and safety.
Keyword
MeSH Terms
Figure
Reference
-
1. Dholakia RJ, Kappel AD, Pagano A, Woo HH, Lieber BB, Fiorella DJ, et al. In vitro angiographic comparison of the flow-diversion performance of five neurovascular stents. Interv Neuroradiol. 2018; 24:150–161. PMID: 29239685.2. Cho SH, Jo WI, Jo YE, Yang KH, Park JC, Lee DH. Bench-top comparison of physical properties of 4 commercially-available self-expanding intracranial stents. Neurointervention. 2017; 12:31–39. PMID: 28316867.
Article3. Kole MK, Pelz DM, Kalapos P, Lee DH, Gulka IB, Lownie SP. Endovascular coil embolization of intracranial aneurysms: important factors related to rates and outcomes of incomplete occlusion. J Neurosurg. 2005; 102:607–615. PMID: 15871501.
Article4. Raymond J, Guilbert F, Weill A, Georganos SA, Juravsky L, Lambert A, et al. Long-term angiographic recurrences after selective endovascular treatment of aneurysms with detachable coils. Stroke. 2003; 34:1398–1403. PMID: 12775880.
Article5. Lee JW, Woo JM, Lim OK, Jo YE, Kim JK, Kim ES, et al. Enlarged parent artery lumen at aneurysmal-neck segment in wide-necked distal internal carotid artery aneurysms. Neurointervention. 2015; 10:82–88. PMID: 26389011.
Article6. Ulfert C, Pham M, Sonnberger M, Amaya F, Trenkler J, Bendszus M, et al. The Neuroform Atlas stent to assist coil embolization of intracranial aneurysms: a multicentre experience. J Neurointerv Surg. 2018; 10:1192–1196. PMID: 29678886.
Article7. Ten Brinck MFM, de Vries J, Bartels RHMA, Grotenhuis JA, Boogaarts HD. NeuroForm Atlas stent-assisted coiling: preliminary results. Neurosurgery. 2019; 84:179–189. PMID: 29579261.
Article8. Iosif C, Piotin M, Saleme S, Barreau X, Sedat J, Chau Y, et al. TRAIL Investigators. Safety and effectiveness of the Low Profile Visualized Intraluminal Support (LVIS and LVIS Jr) devices in the endovascular treatment of intracranial aneurysms: results of the TRAIL multicenter observational study. J Neurointerv Surg. 2018; 10:675–681. PMID: 29175829.
Article9. Fiorella D, Boulos A, Turk AS, Siddiqui AH, Arthur AS, Diaz O, et al. LVIS investigators. The safety and effectiveness of the LVIS stent system for the treatment of wide-necked cerebral aneurysms: final results of the pivotal US LVIS trial. J Neurointerv Surg. 2019; 11:357–361. PMID: 30297543.
Article10. Shankar JJS, Quateen A, Weill A, Tampieri D, Del Pilar Cortes M, Fahed R, et al. Canadian Registry of LVIS Jr for Treatment of Intracranial Aneurysms (CaRLA). J Neurointerv Surg. 2017; 9:849–853. PMID: 27543629.
Article11. Wakhloo AK, Linfante I, Silva CF, Samaniego EA, Dabus G, Etezadi V, et al. Closed-cell stent for coil embolization of intracranial aneurysms: clinical and angiographic results. AJNR Am J Neuroradiol. 2012; 33:1651–1656. PMID: 22492570.
Article12. Zhang X, Zhong J, Gao H, Xu F, Bambakidis NC. Endovascular treatment of intracranial aneurysms with the LVIS device: a systematic review. J Neurointerv Surg. 2017; 9:553–557. PMID: 27206450.
Article13. Ge H, Lv X, Yang X, He H, Jin H, Li Y. LVIS stent versus enterprise stent for the treatment of unruptured intracranial aneurysms. World Neurosurg. 2016; 91:365–370. PMID: 27113398.
Article14. Cho YD, Kim KM, Lee WJ, Sohn CH, Kang HS, Kim JE, et al. Time-of-flight magnetic resonance angiography for follow-up of coil embolization with enterprise stent for intracranial aneurysm: usefulness of source images. Korean J Radiol. 2014; 15:161–168. PMID: 24497808.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Preliminary Experience of Lvis Blue in the Internal Carotid Artery for The Treatment Of Wide-Necked Intracranial Aneurysms
- Double-Balloon-Assisted Coiling for Wide-Necked Posterior Communicating Artery Aneurysms with a Fetal-Type Variant of the Posterior Cerebral Artery: A Case Series
- Clinical and Angiographic Outcomes of Wide-necked Aneurysms Treated with the Solitaire AB Stent
- Outcomes of Stent-Assisted Coiling Using the Neuroform Atlas Stent in Unruptured Wide-Necked Intracranial Aneurysms
- Physiologic Flow Diversion Coiling Technique for Wide-Necked Aneurysms with an Asymmetric Bidirectional Flow at the Aneurysm Neck