J Clin Neurol.
2019 Oct;15(4):537-544. 10.3988/jcn.2019.15.4.537.
Tissue-Clearing Technique and Cutaneous Nerve Biopsies: Quantification of the Intraepidermal Nerve-Fiber Density Using Active Clarity Technique-Pressure Related Efficient and Stable Transfer of Macromolecules Into Organs
- Affiliations
-
- 1Department of Anatomy, Korea University College of Medicine, Seoul, Korea. irhyu@korea.ac.kr
- 2Department of Dermatology, Korea University College of Medicine, Seoul, Korea.
- 3Department of Neurology, Korea University College of Medicine, Seoul, Korea.
- 4Division of Brain Korea 21 Plus Program for Biomedical Science, Korea University College of Medicine, Seoul, Korea.
- KMID: 2467764
- DOI: http://doi.org/10.3988/jcn.2019.15.4.537
Abstract
- BACKGROUND AND PURPOSE
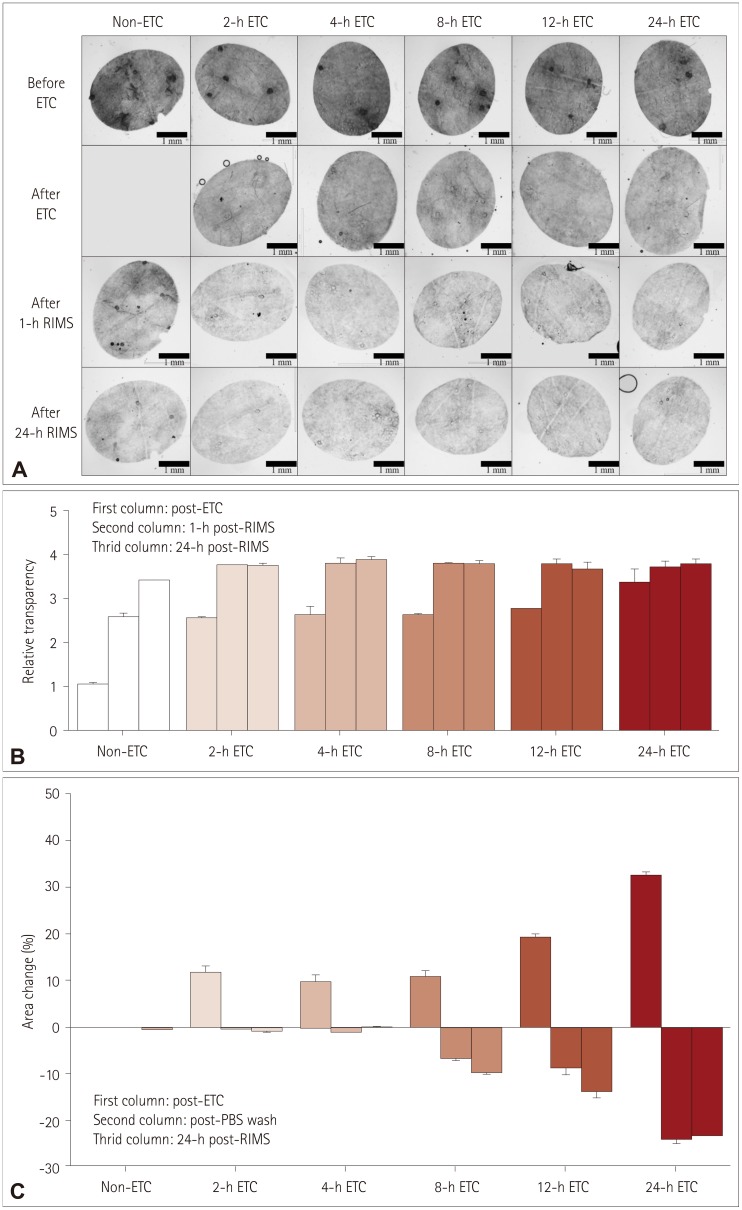
Cutaneous nerve biopsies based on two-dimensional analysis have been regarded as a creditable assessment tool for diagnosing peripheral neuropathies. However, advancements in methodological imaging are required for the analysis of intact structures of peripheral nerve fibers. A tissue-clearing and labeling technique facilitates three-dimensional imaging of internal structures in unsectioned, whole biological tissues without excessive time or labor costs. We sought to establish whether a tissue-clearing and labeling technique could be used for the diagnostic evaluation of peripheral neuropathies.
METHODS
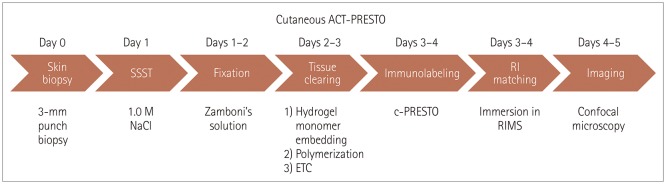
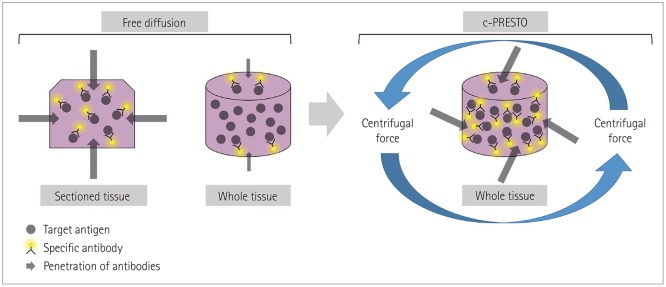
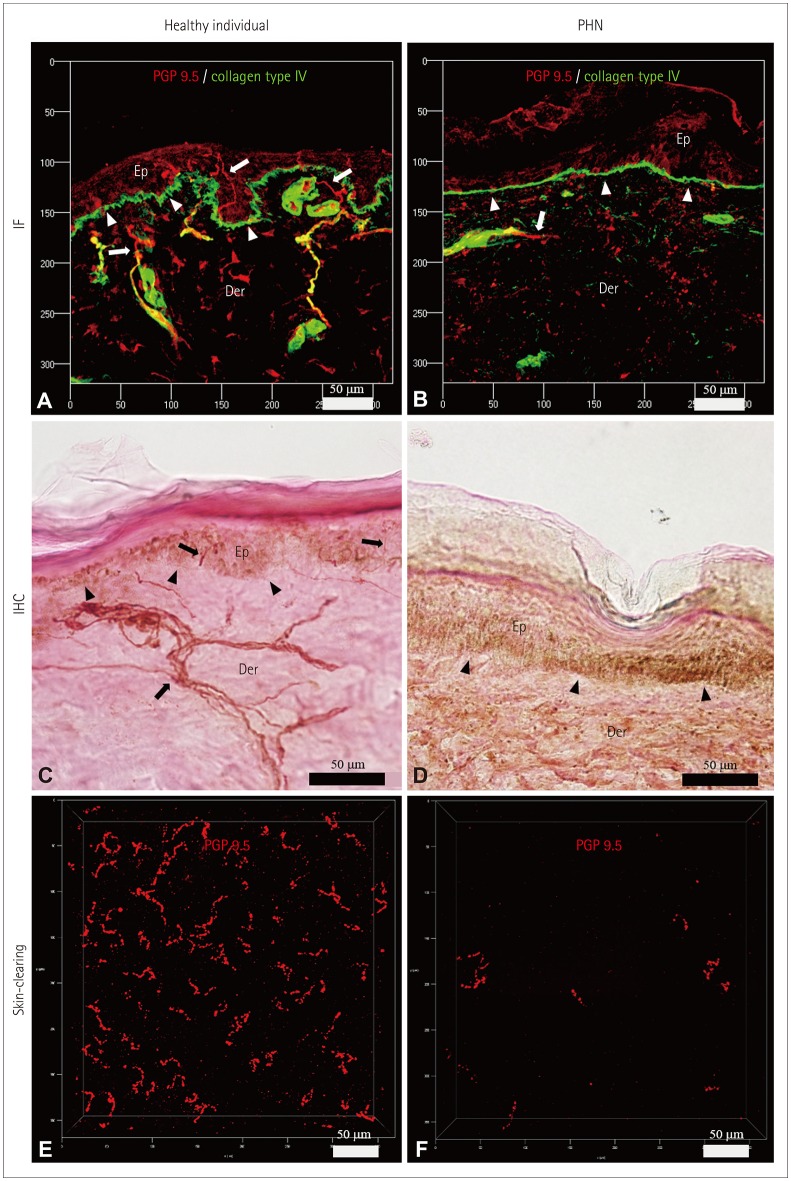
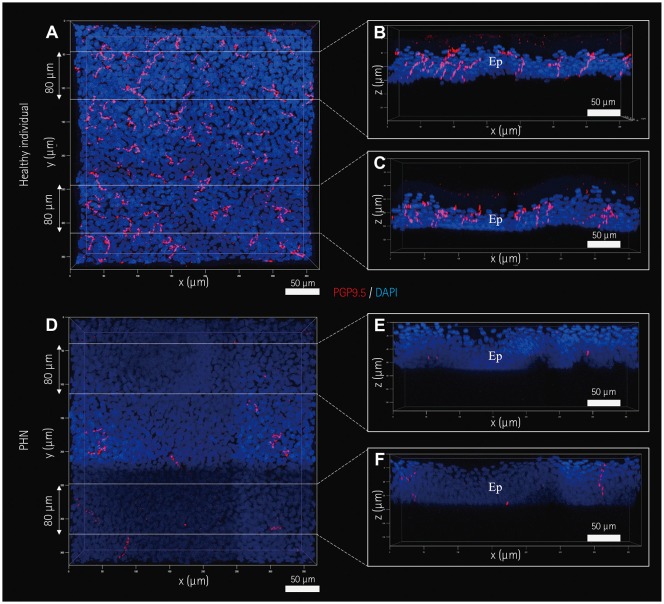
Five healthy individuals and four patients with small-fiber neuropathy (SFN) and postherpetic neuralgia (PHN) were prospectively enrolled. The conventional methods of indirect immunofluorescence (IF) and bright-field immunohistochemistry (IHC) were adopted in addition to the tissue-clearing and labeling method called active clarity technique-pressure related efficient and stable transfer of macromolecules into organs (ACT-PRESTO) to quantify the intraepidermal nerve-fiber density (IENFD).
RESULTS
The mean IENFD values obtained by IF, bright-field IHC, and ACT-PRESTO in the healthy control group were 6.54, 6.44, and 90.19 fibers/mm², respectively; the corresponding values in the patients with SFN were 1.99, 2.32, and 48.12 fibers/mm², respectively, and 3.06, 2.87, and 47.21 fibers/mm², respectively, in the patients with PHN.
CONCLUSIONS
This study has shown that a tissue-clearing method provided not only rapid and highly reproducible three-dimensional images of cutaneous nerve fibers but also yielded reliable quantitative IENFD data. Quantification of the IENFD using a tissue-clearing and labeling technique is a promising way to improve conventional cutaneous nerve biopsies.
Keyword
MeSH Terms
Figure
Reference
-
1. Lauria G, Cornblath DR, Johansson O, McArthur JC, Mellgren SI, Nolano M, et al. EFNS guidelines on the use of skin biopsy in the diagnosis of peripheral neuropathy. Eur J Neurol. 2005; 12:747–758. PMID: 16190912.
Article2. Joint Task Force of the EFNS and the PNS. European Federation of Neurological Societies/Peripheral Nerve Society Guideline on the use of skin biopsy in the diagnosis of small fiber neuropathy. Report of a joint task force of the European Federation of Neurological Societies and the Peripheral Nerve Society. J Peripher Nerv Syst. 2010; 15:79–92. PMID: 20626771.3. Devigili G, Tugnoli V, Penza P, Camozzi F, Lombardi R, Melli G, et al. The diagnostic criteria for small fibre neuropathy: from symptoms to neuropathology. Brain. 2008; 131:1912–1925. PMID: 18524793.
Article4. Mellgren SI, Nolano M, Sommer C. The cutaneous nerve biopsy: technical aspects, indications, and contribution. Handb Clin Neurol. 2013; 115:171–188. PMID: 23931780.5. Lauria G. Small fiber neuropathies. Curr Opin Neurol. 2005; 18:591–597. PMID: 16155446.6. Chung K, Wallace J, Kim SY, Kalyanasundaram S, Andalman AS, Davidson TJ, et al. Structural and molecular interrogation of intact biological systems. Nature. 2013; 497:332–337. PMID: 23575631.
Article7. Lee E, Choi J, Jo Y, Kim JY, Jang YJ, Lee HM, et al. ACT-PRESTO: rapid and consistent tissue clearing and labeling method for 3-dimensional (3D) imaging. Sci Rep. 2016; 6:18631. PMID: 26750588.
Article8. Gong H, Zeng S, Yan C, Lv X, Yang Z, Xu T, et al. Continuously tracing brain-wide long-distance axonal projections in mice at a one-micron voxel resolution. Neuroimage. 2013; 74:87–98. PMID: 23416252.
Article9. Li A, Gong H, Zhang B, Wang Q, Yan C, Wu J, et al. Micro-optical sectioning tomography to obtain a high-resolution atlas of the mouse brain. Science. 2010; 330:1404–1408. PMID: 21051596.
Article10. Kennedy WR, Wendelschafer-Crabb G. The innervation of human epidermis. J Neurol Sci. 1993; 115:184–190. PMID: 8482979.
Article11. McCarthy BG, Hsieh ST, Stocks A, Hauer P, Macko C, Cornblath DR, et al. Cutaneous innervation in sensory neuropathies: evaluation by skin biopsy. Neurology. 1995; 45:1848–1855. PMID: 7477980.
Article12. Gammon WR, Briggaman RA, Inman AO 3rd, Queen LL, Wheeler CE. Differentiating anti-lamina lucida and anti-sublamina densa anti-BMZ antibodies by indirect immunofluorescence on 1.0 M sodium chloride-separated skin. J Invest Dermatol. 1984; 82:139–144. PMID: 6363567.13. Rowbotham MC, Yosipovitch G, Connolly MK, Finlay D, Forde G, Fields HL. Cutaneous innervation density in the allodynic form of postherpetic neuralgia. Neurobiol Dis. 1996; 3:205–214. PMID: 8980021.
Article14. Oaklander AL, Romans K, Horasek S, Stocks A, Hauer P, Meyer RA. Unilateral postherpetic neuralgia is associated with bilateral sensory neuron damage. Ann Neurol. 1998; 44:789–795. PMID: 9818935.
Article15. Boucek P, Havrdova T, Voska L, Lodererova A, He L, Saudek F, et al. Epidermal innervation in type 1 diabetic patients: a 2.5-year prospective study after simultaneous pancreas/kidney transplantation. Diabetes Care. 2008; 31:1611–1612. PMID: 18443196.16. Schifitto G, Yiannoutsos C, Simpson DM, Adornato BT, Singer EJ, Hollander H, et al. Long-term treatment with recombinant nerve growth factor for HIV-associated sensory neuropathy. Neurology. 2001; 57:1313–1316. PMID: 11591856.
Article17. Helmchen F, Denk W. Deep tissue two-photon microscopy. Nat Methods. 2005; 12:932–940.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Skin Biopsy: Emerging Method for Small Nerve Fiber Evaluation
- Skin biopsy: an emerging method for small nerve fiber evaluation
- Intercostal nerve transfer for the treatment of brachial plexus injury
- Assessment of Intraepidermal Nerve Fiber using Skin Biopsy in Diabetic Polyneuropathy
- Quantification of the Nerve Fiber of the Terminal Branches of the Typical Brachial Plexus