J Korean Assoc Oral Maxillofac Surg.
2019 Apr;45(2):83-90. 10.5125/jkaoms.2019.45.2.83.
Overall and disease-specific survival outcomes following primary surgery for oral squamous cell carcinoma: analysis of consecutive 67 patients
- Affiliations
-
- 1School of Medicine, Ewha Womans University, Seoul, Korea.
- 2Department of Oral and Maxillofacial Surgery, University of Ulsan College of Medicine, Asan Medical Center, Seoul, Korea. ahnkangmin@hanmail.net
- KMID: 2467151
- DOI: http://doi.org/10.5125/jkaoms.2019.45.2.83
Abstract
OBJECTIVES
This study evaluated the predictive factors for survival of patients with oral squamous cell carcinoma (OSCC) and investigated the overall and disease-specific survival (DSS) outcomes.
MATERIALS AND METHODS
A total of 67 consecutive patients who underwent surgery for OSCC from January 2006 to November 2014 were included in this study. Patients were classified according to age, sex, pTNM stages, primary sites, smoking and alcohol drinking habits, depth of invasion, perineural and lymphovascular invasion, cell differentiation and postoperative radiotherapy. Kaplan-Meier methods were used to estimate the survival categorized by patient groups. Cox regression methods were used to investigate the main independent predictors of survival.
RESULTS
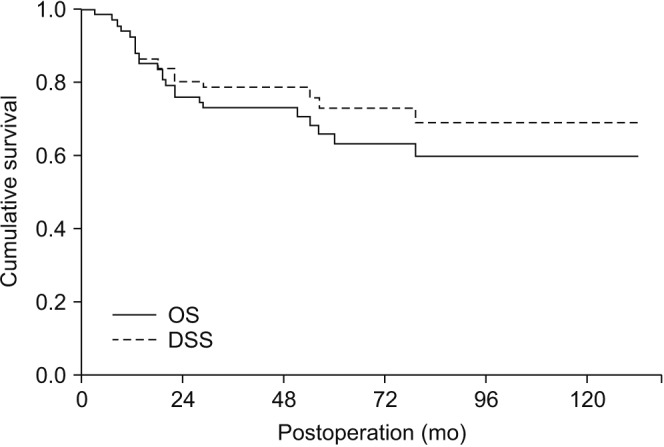
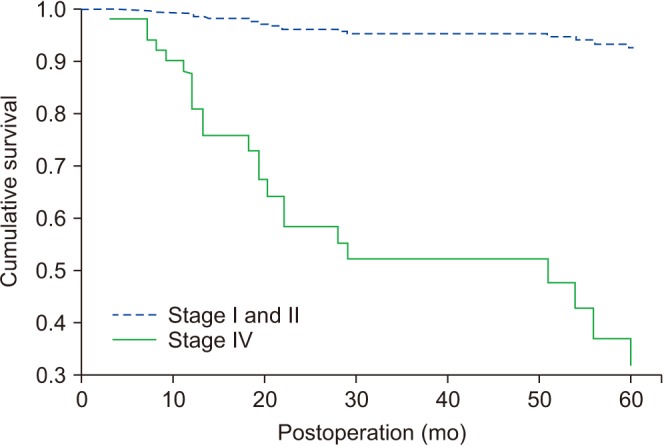
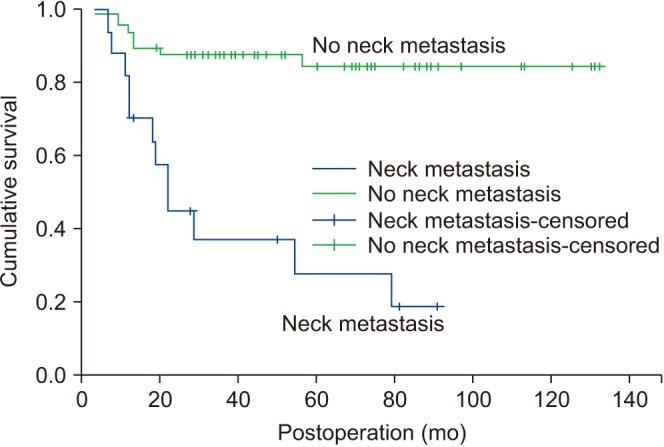
Nineteen patients died of OSCC during follow-up periods. Another five patients died of other diseases including lung adenocarcinoma (n=1), cerebral infarction (n=1), general weakness (n=2), and pneumonia (n=1). The tongue (n=16) was the most common site for primary origin, followed by buccal mucosa (n=15), mandibular gingiva (n=15), maxillary gingiva (n=9), floor of mouth (n=9), retromolar trigone (n=2), and palate (n=1). Eleven patients had pTNM stage I disease, followed by stage II (n=22) and stage IV (n=34). No patients had pTNM stage III disease in this study. The overall survival of all patients was 64.2% and the DSS was 71.6%. DSS of patients with stage I and II disease was 100%. Stepwise Cox regression showed the two predictors for DSS were pTNM stage (P<0.0001, odds ratio=19.633) and presence of metastatic lymph nodes (P=0.0004, odds ratio=0.1039).
CONCLUSION
OSCC has been associated with poor prognosis; however, there were improved survival outcomes compared with past studies. Advanced-stage disease and presence of metastatic lymph nodes were associated with poorer survival compared with early-stage OSCC and absence of neck node metastasis. Stage I and II OSCC were associated with excellent survival results in this study.
MeSH Terms
Figure
Cited by 1 articles
-
Analysis of prognostic factors through survival rate analysis of oral squamous cell carcinoma patients treated at the National Cancer Center: 20 years of experience
Yong-Seok Choi, Min Gyeong Kim, Jong-Ho Lee, Joo-Yong Park, Sung-Weon Choi
J Korean Assoc Oral Maxillofac Surg. 2022;48(5):284-291. doi: 10.5125/jkaoms.2022.48.5.284.
Reference
-
1. Moore SR, Johnson NW, Pierce AM, Wilson DF. The epidemiology of tongue cancer: a review of global incidence. Oral Dis. 2000; 6:75–84. PMID: 10702783.
Article2. Johnson NW. Orofacial neoplasms: global epidemiology, risk factors and recommendations for research. Int Dent J. 1991; 41:365–375. PMID: 1800387.3. Kim BG, Kim JH, Kim MI, Han JJ, Jung S, Kook MS, et al. Retrospective study on factors affecting the prognosis in oral cancer patients who underwent surgical treatment only. Maxillofac Plast Reconstr Surg. 2016; 38:3. PMID: 26807400.
Article4. Madani AH, Dikshit M, Bhaduri D, Aghamolaei T, Moosavy SH, Azarpaykan A. Interaction of alcohol use and specific types of smoking on the development of oral cancer. Int J High Risk Behav Addict. 2014; 3:e12120. PMID: 24971295.
Article5. Shiboski CH, Schmidt BL, Jordan RC. Tongue and tonsil carcinoma: increasing trends in the U.S. population ages 20–44 years. Cancer. 2005; 103:1843–1849. PMID: 15772957.6. Kademani D, Bell RB, Bagheri S, Holmgren E, Dierks E, Potter B, et al. Prognostic factors in intraoral squamous cell carcinoma: the influence of histologic grade. J Oral Maxillofac Surg. 2005; 63:1599–1605. PMID: 16243176.
Article7. Rogers SN, Brown JS, Woolgar JA, Lowe D, Magennis P, Shaw RJ, et al. Survival following primary surgery for oral cancer. Oral Oncol. 2009; 45:201–211. PMID: 18674959.
Article8. Geum DH, Roh YC, Yoon SY, Kim HG, Lee JH, Song JM, et al. The impact factors on 5-year survival rate in patients operated with oral cancer. J Korean Assoc Oral Maxillofac Surg. 2013; 39:207–216. PMID: 24471047.
Article9. Hwang DS, Park J, Kim UK, Park HR, Kim GC, Ryu MH. Reconstruction of cheek mucosal defect with a buccal fat pad flap in a squamous cell carcinoma patient: a case report and literature review. Maxillofac Plast Reconstr Surg. 2018; 40:11. PMID: 29872648.
Article10. Holtmann H, Spalthoff S, Gellrich NC, Handschel J, Lommen J, Kübler NR, et al. Determinants for further wishes for cosmetic and reconstructive interventions in 1652 patients with surgical treated carcinomas of the oral cavity. Maxillofac Plast Reconstr Surg. 2017; 39:26. PMID: 28944220.
Article11. Shah JP, Medina JE, Shaha AR, Schantz SP, Marti JR. Cervical lymph node metastasis. Curr Probl Surg. 1993; 30:1–335.
Article12. Bloom ND, Spiro RH. Carcinoma of the cheek mucosa. A retrospective analysis. Am J Surg. 1980; 140:556–559. PMID: 7425239.13. Kang S, Leem DH. Verrucous carcinoma arising from a previous cystic lesion: a case report. Maxillofac Plast Reconstr Surg. 2018; 40:31. PMID: 30397593.
Article14. Shaha AR, Spiro RH, Shah JP, Strong EW. Squamous carcinoma of the floor of the mouth. Am J Surg. 1984; 148:455–459. PMID: 6486312.
Article15. Shah JP, Candela FC, Poddar AK. The patterns of cervical lymph node metastases from squamous carcinoma of the oral cavity. Cancer. 1990; 66:109–113. PMID: 2354399.
Article16. Shah JP. Patterns of cervical lymph node metastasis from squamous carcinomas of the upper aerodigestive tract. Am J Surg. 1990; 160:405–409. PMID: 2221244.
Article17. Lindberg R. Distribution of cervical lymph node metastases from squamous cell carcinoma of the upper respiratory and digestive tracts. Cancer. 1972; 29:1446–1449. PMID: 5031238.
Article18. Candela FC, Kothari K, Shah JP. Patterns of cervical node metastases from squamous carcinoma of the oropharynx and hypopharynx. Head Neck. 1990; 12:197–203. PMID: 2358329.
Article19. De Vicente JC, Recio OR, Pendás SL, López-Arranz JS. Oral squamous cell carcinoma of the mandibular region: a survival study. Head Neck. 2001; 23:536–543. PMID: 11400241.
Article20. Park TJ, Kim HJ, Ahn KM. Double-layered collagen graft to the radial forearm free flap donor sites without skin graft. Maxillofac Plast Reconstr Surg. 2015; 37:45. PMID: 26693164.
Article21. Warnakulasuriya S, Mak V, Möller H. Oral cancer survival in young people in South East England. Oral Oncol. 2007; 43:982–986. PMID: 17350878.
Article22. Sessions DG, Spector GJ, Lenox J, Parriott S, Haughey B, Chao C, et al. Analysis of treatment results for floor-of-mouth cancer. Laryngoscope. 2000; 110:1764–1772. PMID: 11037841.
Article23. Horner MJ, Ries LAG, Krapcho M, Neyman N, Aminou R, Howlader N, et al. SEER cancer statistics review, 1975-2006 [Internet]. Bethesda (MD): National Cancer Institute;cited 2018 Jan 25. Available from: https://seer.cancer.gov/archive/csr/1975_2006.24. Warnakulasuriya S. Global epidemiology of oral and oropharyngeal cancer. Oral Oncol. 2009; 45:309–316. PMID: 18804401.
Article25. Redaniel MT, Laudico A, Mirasol-Lumague MR, Gondos A, Pulte D, Mapua C, et al. Cancer survival discrepancies in developed and developing countries: comparisons between the Philippines and the United States. Br J Cancer. 2009; 100:858–862. PMID: 19240723.
Article26. Listl S, Jansen L, Stenzinger A, Freier K, Emrich K, Holleczek B, et al. Survival of patients with oral cavity cancer in Germany. PLoS One. 2013; 8:e53415. PMID: 23349710.
Article27. Sklenicka S, Gardiner S, Dierks EJ, Potter BE, Bell RB. Survival analysis and risk factors for recurrence in oral squamous cell carcinoma: does surgical salvage affect outcome? J Oral Maxillofac Surg. 2010; 68:1270–1275. PMID: 20347201.
Article28. Süslü N, Hoşal AŞ, Aslan T, Sözeri B, Dolgun A. Carcinoma of the oral tongue: a case series analysis of prognostic factors and surgical outcomes. J Oral Maxillofac Surg. 2013; 71:1283–1290. PMID: 23522769.
Article29. Grandi C, Alloisio M, Moglia D, Podrecca S, Sala L, Salvatori P, et al. Prognostic significance of lymphatic spread in head and neck carcinomas: therapeutic implications. Head Neck Surg. 1985; 8:67–73. PMID: 4077553.
Article30. Johnson NW, Jayasekara P, Amarasinghe AA. Squamous cell carcinoma and precursor lesions of the oral cavity: epidemiology and aetiology. Periodontol 2000. 2011; 57:19–37. PMID: 21781177.
Article31. Phusingha P, Ekalaksananan T, Vatanasapt P, Loyha K, Promthet S, Kongyingyoes B, et al. Human papillomavirus (HPV) infection in a case-control study of oral squamous cell carcinoma and its increasing trend in northeastern Thailand. J Med Virol. 2017; 89:1096–1101. PMID: 27935063.
Article32. Zelefsky MJ, Harrison LB, Fass DE, Armstrong J, Spiro RH, Shah JP, et al. Postoperative radiotherapy for oral cavity cancers: impact of anatomic subsite on treatment outcome. Head Neck. 1990; 12:470–475. PMID: 2258285.
Article33. Kim HJ, Park TJ, Ahn KM. Bisphosphonate-related osteonecrosis of the jaw in metastatic breast cancer patients: a review of 25 cases. Maxillofac Plast Reconstr Surg. 2016; 38:6. PMID: 26870717.
Article34. Kurokawa H, Zhang M, Matsumoto S, Yamashita Y, Tomoyose T, Tanaka T, et al. The high prognostic value of the histologic grade at the deep invasive front of tongue squamous cell carcinoma. J Oral Pathol Med. 2005; 34:329–333. PMID: 15946179.
Article35. Kim BG, Kim JH, Kim MI, Han JJ, Jung S, Kook MS, et al. Retrospective study on factors affecting the prognosis in oral cancer patients who underwent surgical treatment only. Maxillofac Plast Reconstr Surg. 2016; 38:3. PMID: 26807400.
Article36. Mohit-Tabatabai MA, Sobel HJ, Rush BF, Mashberg A. Relation of thickness of floor of mouth stage I and II cancers to regional metastasis. Am J Surg. 1986; 152:351–353. PMID: 3766862.
Article37. Jones KR, Lodge-Rigal RD, Reddick RL, Tudor GE, Shockley WW. Prognostic factors in the recurrence of stage I and II squamous cell cancer of the oral cavity. Arch Otolaryngol Head Neck Surg. 1992; 118:483–485. PMID: 1571116.
Article38. Fukano H, Matsuura H, Hasegawa Y, Nakamura S. Depth of invasion as a predictive factor for cervical lymph node metastasis in tongue carcinoma. Head Neck. 1997; 19:205–210. PMID: 9142520.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Secondary Squamous Cell Carcinoma of the Oral Cavity after Nasopharyngeal Carcinoma
- Overall Five-Year Survival Rate in Squamous Cell Carcinoma of Oral Cavity
- Diagnostic problem of squamous papilloma and oral mucosa malignancy
- Synchronous thyroid carcinoma and squamous cell carcinoma: A case report
- Squamous Cell Carcinoma of the Maxilla Originated in Odontogenic Cyst: A Case Report