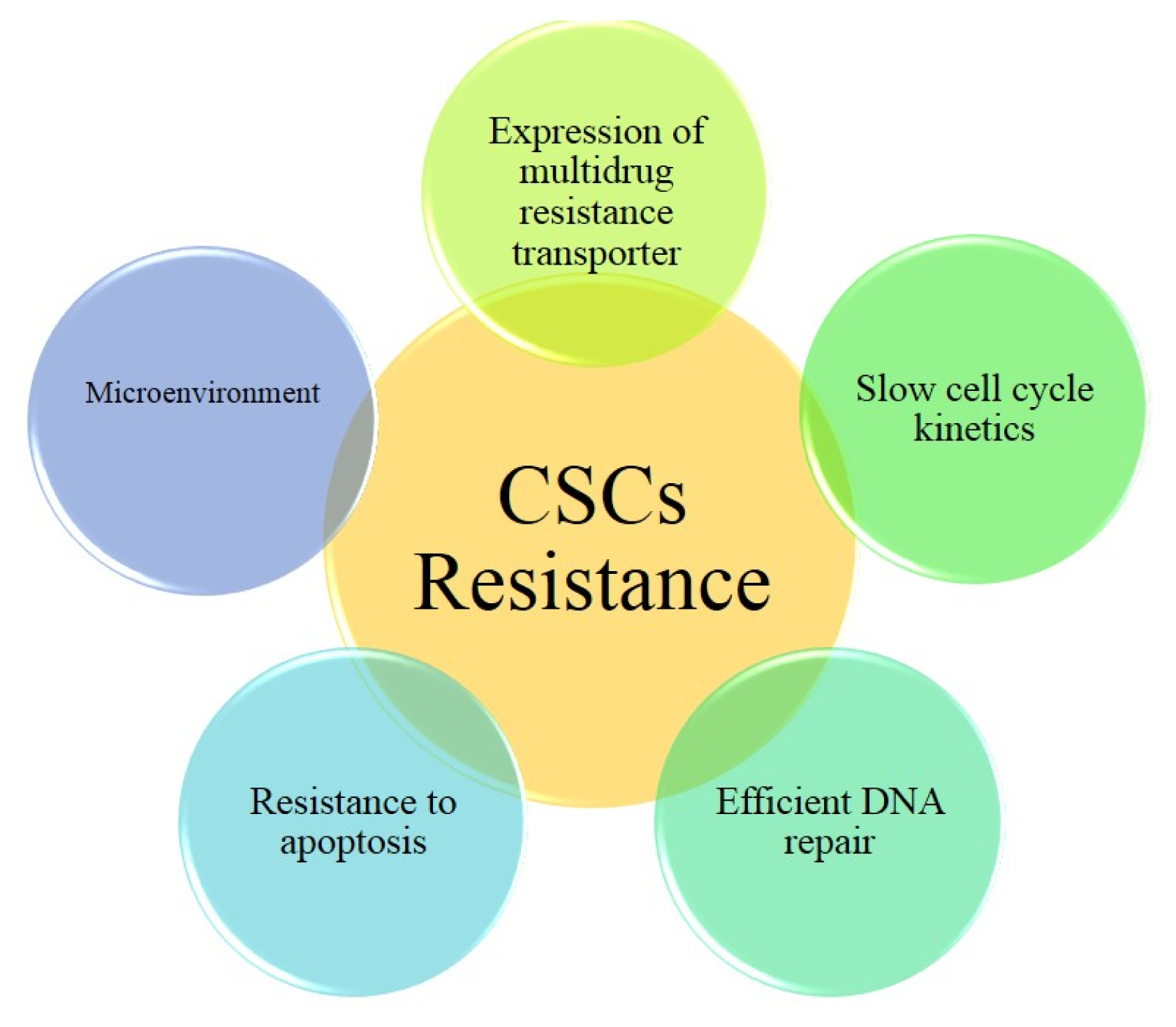
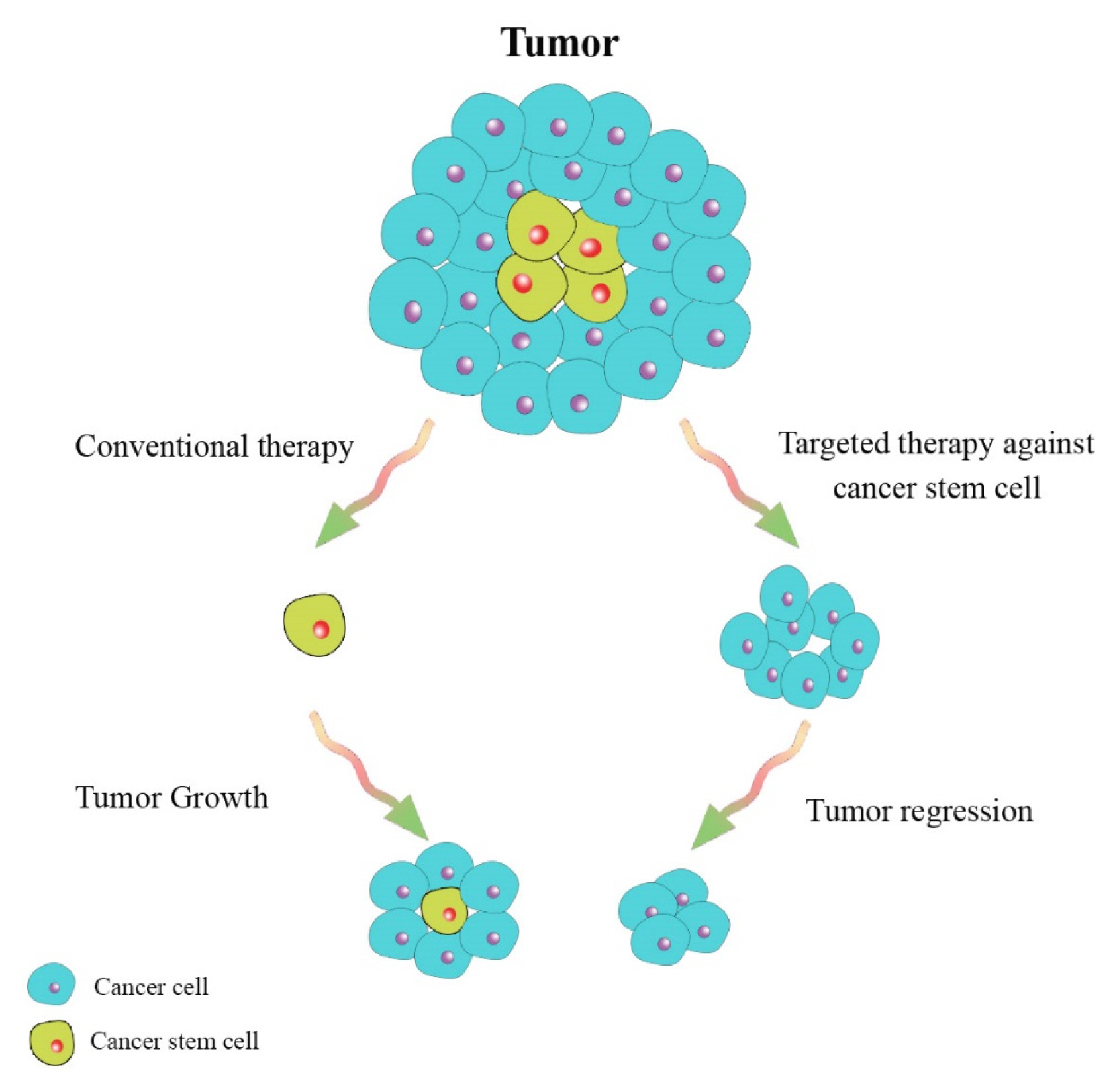
Int J Stem Cells.
2019 Jul;12(2):227-239. 10.15283/ijsc19006.
Application of Nanotechnology in Targeting of Cancer Stem Cells: A Review
- Affiliations
-
- 1Department of Medical Nanotechnology, School of Advanced Technologies in Medicine, Tehran University of Medical Sciences, Tehran, Iran. refaridi@tums.ac.ir
- 2International affairs, School of Advanced Technologies in Medicine, Tehran University of Medical Sciences, Tehran, Iran.
- 3Department of Tissue Engineering and Applied Cell Sciences, School of Advanced Technologies in Medicine, Tehran University of Medical Sciences, Tehran, Iran.
- 4Department of Medical Biotechnology, School of Advanced Technologies in Medicine, Tehran University of Medical Sciences, Tehran, Iran.
- KMID: 2465894
- DOI: http://doi.org/10.15283/ijsc19006
Abstract
- Cancer is increasingly apparent as a systems-level, network happening. The central tendency of malignant alteration can be described as a two-phase procedure, where an initial increase of network plasticity is followed by reducing plasticity at late stages of tumor improvement. Cancer stem cells (CSCs) are cancer cells that take characteristics associated with normal stem cells. Cancer therapy has been based on the concept that most of the cancer cells have a similar ability to separate metastasise and kill the host. In this review, we addressed the use of nanotechnology in the treatment of cancer stem cells.
Keyword
MeSH Terms
Figure
Reference
-
References
1. Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013; 63:11–30. DOI: 10.3322/caac.21166. PMID: 23335087.
Article2. Sahai E. Mechanisms of cancer cell invasion. Curr Opin Genet Dev. 2005; 15:87–96. DOI: 10.1016/j.gde.2004.12.002. PMID: 15661538.
Article3. Dalerba P, Cho RW, Clarke MF. Cancer stem cells: models and concepts. Annu Rev Med. 2007; 58:267–284. DOI: 10.1146/annurev.med.58.062105.204854. PMID: 17002552.
Article4. Jordan CT, Guzman ML, Noble M. Cancer stem cells. N Engl J Med. 2006; 355:1253–1261. DOI: 10.1056/NEJMra061808. PMID: 16990388.
Article5. World Health Organization. WHO handbook for reporting results of cancer treatment. Geneva: WHO;1979.6. Li C, Heidt DG, Dalerba P, Burant CF, Zhang L, Adsay V, Wicha M, Clarke MF, Simeone DM. Identification of pancreatic cancer stem cells. Cancer Res. 2007; 67:1030–1037. DOI: 10.1158/0008-5472.CAN-06-2030. PMID: 17283135.
Article7. Bonnet D, Dick JE. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med. 1997; 3:730–737. DOI: 10.1038/nm0797-730. PMID: 9212098.
Article8. Ciurea ME, Georgescu AM, Purcaru SO, Artene SA, Emami GH, Boldeanu MV, Tache DE, Dricu A. Cancer stem cells: biological functions and therapeutically targeting. Int J Mol Sci. 2014; 15:8169–8185. DOI: 10.3390/ijms15058169. PMID: 24821540. PMCID: PMC4057726.9. Castelo-Branco P, Zhang C, Lipman T, Fujitani M, Hansford L, Clarke I, Harley CB, Tressler R, Malkin D, Walker E, Kaplan DR, Dirks P, Tabori U. Neural tumor-initiating cells have distinct telomere maintenance and can be safely targeted for telomerase inhibition. Clin Cancer Res. 2011; 17:111–121. DOI: 10.1158/1078-0432.CCR-10-2075. PMID: 21208905.
Article10. Li C, Hynes MJ, Jing J. Pancreatic cancer stem cells: new direction for pancreatic cancer treatment. Trends Bio/Pharm Ind. 2010; 6:34–40.11. Miki J, Furusato B, Li H, Gu Y, Takahashi H, Egawa S, Sesterhenn IA, McLeod DG, Srivastava S, Rhim JS. Identification of putative stem cell markers, CD133 and CXCR4, in hTERT-immortalized primary nonmalignant and malignant tumor-derived human prostate epithelial cell lines and in prostate cancer specimens. Cancer Res. 2007; 67:3153–3161. DOI: 10.1158/0008-5472.CAN-06-4429. PMID: 17409422.
Article12. Ricardo S, Vieira AF, Gerhard R, Leitão D, Pinto R, Cameselle-Teijeiro JF, Milanezi F, Schmitt F, Paredes J. Breast cancer stem cell markers CD44, CD24 and ALDH1: expression distribution within intrinsic molecular subtype. J Clin Pathol. 2011; 64:937–946. DOI: 10.1136/jcp.2011.090456. PMID: 21680574.
Article13. Takaishi S, Okumura T, Tu S, Wang SS, Shibata W, Vigneshwaran R, Gordon SA, Shimada Y, Wang TC. Identification of gastric cancer stem cells using the cell surface marker CD44. Stem Cells. 2009; 27:1006–1020. DOI: 10.1002/stem.30. PMID: 19415765. PMCID: PMC2746367.
Article14. Burnett J. The natural product sulforaphane inhibits breast cancer stem cell targets in triple negative and trastuzumab-resistant breast cancers [PhD dissertation]. Michigan: University of Michigan;2015.15. Wang JC, Dick JE. Cancer stem cells: lessons from leukemia. Trends Cell Biol. 2005; 15:494–501. DOI: 10.1016/j.tcb.2005.07.004. PMID: 16084092.
Article16. Hosen N, Park CY, Tatsumi N, Oji Y, Sugiyama H, Gramatzki M, Krensky AM, Weissman IL. CD96 is a leukemic stem cell-specific marker in human acute myeloid leukemia. Proc Natl Acad Sci U S A. 2007; 104:11008–11013. DOI: 10.1073/pnas.0704271104. PMID: 17576927. PMCID: PMC1904175.
Article17. Croker AK, Goodale D, Chu J, Postenka C, Hedley BD, Hess DA, Allan AL. High aldehyde dehydrogenase and expression of cancer stem cell markers selects for breast cancer cells with enhanced malignant and metastatic ability. J Cell Mol Med. 2009; 13:2236–2252. DOI: 10.1111/j.1582-4934.2008.00455.x. PMID: 18681906. PMCID: PMC6512388.
Article18. Beier D, Hau P, Proescholdt M, Lohmeier A, Wischhusen J, Oefner PJ, Aigner L, Brawanski A, Bogdahn U, Beier CP. CD133(+) and CD133(−) glioblastoma-derived cancer stem cells show differential growth characteristics and molecular profiles. Cancer Res. 2007; 67:4010–4015. DOI: 10.1158/0008-5472.CAN-06-4180. PMID: 17483311.
Article19. Singh SK, Clarke ID, Terasaki M, Bonn VE, Hawkins C, Squire J, Dirks PB. Identification of a cancer stem cell in human brain tumors. Cancer Res. 2003; 63:5821–5828. PMID: 14522905.20. Peacock CD, Wang Q, Gesell GS, Corcoran-Schwartz IM, Jones E, Kim J, Devereux WL, Rhodes JT, Huff CA, Beachy PA, Watkins DN, Matsui W. Hedgehog signaling maintains a tumor stem cell compartment in multiple myeloma. Proc Natl Acad Sci U S A. 2007; 104:4048–4053. DOI: 10.1073/pnas.0611682104. PMID: 17360475. PMCID: PMC1805487.
Article21. Dammacco F, Leone P, Silvestris F, Racanelli V, Vacca A. Cancer stem cells in multiple myeloma and the development of novel therapeutic strategies. Dammacco F, editor. Oncogenomics. London: Elsevier;2019. p. 121–137. DOI: 10.1016/B978-0-12-811785-9.00009-0.
Article22. Hermann PC, Huber SL, Herrler T, Aicher A, Ellwart JW, Guba M, Bruns CJ, Heeschen C. Distinct populations of cancer stem cells determine tumor growth and metastatic activity in human pancreatic cancer. Cell Stem Cell. 2007; 1:313–323. DOI: 10.1016/j.stem.2007.06.002. PMID: 18371365.
Article23. Vermeulen L, De Sousa E, Melo F, van der Heijden M, Cameron K, de Jong JH, Borovski T, Tuynman JB, Todaro M, Merz C, Rodermond H, Sprick MR, Kemper K, Richel DJ, Stassi G, Medema JP. Wnt activity defines colon cancer stem cells and is regulated by the microenvironment. Nat Cell Biol. 2010; 12:468–476. DOI: 10.1038/ncb2048. PMID: 20418870.
Article24. Ma S, Chan KW, Hu L, Lee TK, Wo JY, Ng IO, Zheng BJ, Guan XY. Identification and characterization of tumorigenic liver cancer stem/progenitor cells. Gastroenterology. 2007; 132:2542–2556. DOI: 10.1053/j.gastro.2007.04.025. PMID: 17570225.
Article25. Yang ZF, Ho DW, Ng MN, Lau CK, Yu WC, Ngai P, Chu PW, Lam CT, Poon RT, Fan ST. Significance of CD90+ cancer stem cells in human liver cancer. Cancer Cell. 2008; 13:153–166. DOI: 10.1016/j.ccr.2008.01.013. PMID: 18242515.
Article26. Collins AT, Berry PA, Hyde C, Stower MJ, Maitland NJ. Prospective identification of tumorigenic prostate cancer stem cells. Cancer Res. 2005; 65:10946–10951. DOI: 10.1158/0008-5472.CAN-05-2018. PMID: 16322242.
Article27. Liu C, Kelnar K, Liu B, Chen X, Calhoun-Davis T, Li H, Patrawala L, Yan H, Jeter C, Honorio S, Wiggins JF, Bader AG, Fagin R, Brown D, Tang DG. The microRNA miR-34a inhibits prostate cancer stem cells and metastasis by directly repressing CD44. Nat Med. 2011; 17:211–215. DOI: 10.1038/nm.2284. PMID: 21240262. PMCID: PMC3076220.
Article28. Eramo A, Lotti F, Sette G, Pilozzi E, Biffoni M, Di Virgilio A, Conticello C, Ruco L, Peschle C, De Maria R. Identification and expansion of the tumorigenic lung cancer stem cell population. Cell Death Differ. 2008; 15:504–514. DOI: 10.1038/sj.cdd.4402283. PMID: 18049477.
Article29. Gao MQ, Choi YP, Kang S, Youn JH, Cho NH. CD24+ cells from hierarchically organized ovarian cancer are enriched in cancer stem cells. Oncogene. 2010; 29:2672–2680. DOI: 10.1038/onc.2010.35. PMID: 20190812.
Article30. Silva IA, Bai S, McLean K, Yang K, Griffith K, Thomas D, Ginestier C, Johnston C, Kueck A, Reynolds RK, Wicha MS, Buckanovich RJ. Aldehyde dehydrogenase in combination with CD133 defines angiogenic ovarian cancer stem cells that portend poor patient survival. Cancer Res. 2011; 71:3991–4001. DOI: 10.1158/0008-5472.CAN-10-3175. PMID: 21498635. PMCID: PMC3107359.
Article31. Zhang S, Balch C, Chan MW, Lai HC, Matei D, Schilder JM, Yan PS, Huang TH, Nephew KP. Identification and characterization of ovarian cancer-initiating cells from primary human tumors. Cancer Res. 2008; 68:4311–4320. DOI: 10.1158/0008-5472.CAN-08-0364. PMID: 18519691. PMCID: PMC2553722.
Article32. Csermely P, Hódsági J, Korcsmáros T, Módos D, Perez-Lopez ÁR, Szalay K, Veres DV, Lenti K, Wu LY, Zhang XS. Cancer stem cells display extremely large evolvability: alternating plastic and rigid networks as a potential mechanism: network models, novel therapeutic target strategies, and the contributions of hypoxia, inflammation and cellular senescence. Semin Cancer Biol. 2015; 30:42–51. DOI: 10.1016/j.semcancer.2013.12.004. PMID: 24412105.
Article33. Liu L, Rando TA. Manifestations and mechanisms of stem cell aging. J Cell Biol. 2011; 193:257–266. DOI: 10.1083/jcb.201010131. PMID: 21502357. PMCID: PMC3080271.
Article34. Hoek KS, Goding CR. Cancer stem cells versus phenotype-switching in melanoma. Pigment Cell Melanoma Res. 2010; 23:746–759. DOI: 10.1111/j.1755-148X.2010.00757.x. PMID: 20726948.
Article35. Visvader JE, Lindeman GJ. Cancer stem cells in solid tumours: accumulating evidence and unresolved questions. Nat Rev Cancer. 2008; 8:755–768. DOI: 10.1038/nrc2499. PMID: 18784658.
Article36. Singh A, Settleman J. EMT, cancer stem cells and drug resistance: an emerging axis of evil in the war on cancer. Oncogene. 2010; 29:4741–4751. DOI: 10.1038/onc.2010.215. PMID: 20531305. PMCID: PMC3176718.
Article37. Fletcher JI, Haber M, Henderson MJ, Norris MD. ABC transporters in cancer: more than just drug efflux pumps. Nat Rev Cancer. 2010; 10:147–156. DOI: 10.1038/nrc2789. PMID: 20075923.
Article38. Gottesman MM. Mechanisms of cancer drug resistance. Annu Rev Med. 2002; 53:615–627. DOI: 10.1146/annurev.med.53.082901.103929. PMID: 11818492.
Article39. Quintana E, Shackleton M, Sabel MS, Fullen DR, Johnson TM, Morrison SJ. Efficient tumour formation by single human melanoma cells. Nature. 2008; 456:593–598. DOI: 10.1038/nature07567. PMID: 19052619. PMCID: PMC2597380.
Article40. Cho RW, Clarke MF. Recent advances in cancer stem cells. Curr Opin Genet Dev. 2008; 18:48–53. DOI: 10.1016/j.gde.2008.01.017. PMID: 18356041.
Article41. Bloushtain-Qimron N, Yao J, Snyder EL, Shipitsin M, Campbell LL, Mani SA, Hu M, Chen H, Ustyansky V, Antosiewicz JE, Argani P, Halushka MK, Thomson JA, Pharoah P, Porgador A, Sukumar S, Parsons R, Richardson AL, Stampfer MR, Gelman RS, Nikolskaya T, Nikolsky Y, Polyak K. Cell type-specific DNA methylation patterns in the human breast. Proc Natl Acad Sci U S A. 2008; 105:14076–14081. DOI: 10.1073/pnas.0805206105. PMID: 18780791. PMCID: PMC2532972.
Article42. Shipitsin M, Campbell LL, Argani P, Weremowicz S, Bloushtain-Qimron N, Yao J, Nikolskaya T, Serebryiskaya T, Beroukhim R, Hu M, Halushka MK, Sukumar S, Parker LM, Anderson KS, Harris LN, Garber JE, Richardson AL, Schnitt SJ, Nikolsky Y, Gelman RS, Polyak K. Molecular definition of breast tumor heterogeneity. Cancer Cell. 2007; 11:259–273. DOI: 10.1016/j.ccr.2007.01.013. PMID: 17349583.
Article43. Dick JE. Looking ahead in cancer stem cell research. Nat Biotechnol. 2009; 27:44–46. DOI: 10.1038/nbt0109-44. PMID: 19131997.
Article44. Moitra K, Lou H, Dean M. Multidrug efflux pumps and cancer stem cells: insights into multidrug resistance and therapeutic development. Clin Pharmacol Ther. 2011; 89:491–502. DOI: 10.1038/clpt.2011.14. PMID: 21368752.
Article45. Vermeulen L, de Sousa e Melo F, Richel DJ, Medema JP. The developing cancer stem-cell model: clinical challenges and opportunities. Lancet Oncol. 2012; 13:e83–e89. DOI: 10.1016/S1470-2045(11)70257-1. PMID: 22300863.
Article46. Zhou BB, Zhang H, Damelin M, Geles KG, Grindley JC, Dirks PB. Tumour-initiating cells: challenges and opportunities for anticancer drug discovery. Nat Rev Drug Discov. 2009; 8:806–823. DOI: 10.1038/nrd2137. PMID: 19794444.
Article47. Naujokat C, Steinhart R. Salinomycin as a drug for targeting human cancer stem cells. J Biomed Biotechnol. 2012; 2012:950658. DOI: 10.1155/2012/950658. PMID: 23251084. PMCID: PMC3516046.
Article48. Domingo-Domenech J, Vidal SJ, Rodriguez-Bravo V, Castillo-Martin M, Quinn SA, Rodriguez-Barrueco R, Bonal DM, Charytonowicz E, Gladoun N, de la Iglesia-Vicente J, Petrylak DP, Benson MC, Silva JM, Cordon-Cardo C. Suppression of acquired docetaxel resistance in prostate cancer through depletion of notch- and hedgehog-dependent tumor-initiating cells. Cancer Cell. 2012; 22:373–388. DOI: 10.1016/j.ccr.2012.07.016. PMID: 22975379. PMCID: PMC5989708.
Article49. Hu Y, Fu L. Targeting cancer stem cells: a new therapy to cure cancer patients. Am J Cancer Res. 2012; 2:340–356. PMID: 22679565. PMCID: PMC3365812.50. Zhao C, Blum J, Chen A, Kwon HY, Jung SH, Cook JM, Lagoo A, Reya T. Loss of beta-catenin impairs the renewal of normal and CML stem cells in vivo. Cancer Cell. 2007; 12:528–541. DOI: 10.1016/j.ccr.2007.11.003. PMID: 18068630. PMCID: PMC2262869.
Article51. Yin L, Velazquez OC, Liu ZJ. Notch signaling: emerging molecular targets for cancer therapy. Biochem Pharmacol. 2010; 80:690–701. DOI: 10.1016/j.bcp.2010.03.026. PMID: 20361945.
Article52. Guo S, Liu M, Gonzalez-Perez RR. Role of Notch and its oncogenic signaling crosstalk in breast cancer. Biochim Biophys Acta. 2011; 1815:197–213. PMID: 21193018. PMCID: PMC3060666.
Article53. Hovinga KE, Shimizu F, Wang R, Panagiotakos G, Van Der Heijden M, Moayedpardazi H, Correia AS, Soulet D, Major T, Menon J, Tabar V. Inhibition of notch signaling in glioblastoma targets cancer stem cells via an endothelial cell intermediate. Stem Cells. 2010; 28:1019–1029. DOI: 10.1002/stem.429. PMID: 20506127. PMCID: PMC5532884.
Article54. Peukert S, Miller-Moslin K. Small-molecule inhibitors of the hedgehog signaling pathway as cancer therapeutics. ChemMedChem. 2010; 5:500–512. DOI: 10.1002/cmdc.201000011. PMID: 20229564.
Article55. Chen JK, Taipale J, Cooper MK, Beachy PA. Inhibition of Hedgehog signaling by direct binding of cyclopamine to Smoothened. Genes Dev. 2002; 16:2743–2748. DOI: 10.1101/gad.1025302. PMID: 12414725. PMCID: PMC187469.
Article56. Alexis F, Rhee JW, Richie JP, Radovic-Moreno AF, Langer R, Farokhzad OC. New frontiers in nanotechnology for cancer treatment. Urol Oncol. 2008; 26:74–85. DOI: 10.1016/j.urolonc.2007.03.017. PMID: 18190835.
Article57. Guler B, Demir B, Guler E, Gulec K, Yesiltepe O, Demirkol DO, Timur S. Targeting and imaging of cancer cells using nanomaterials. Grumezescu AM, editor. Nanobiomaterials in Medical Imaging. Oxford: Elsevier;2016. p. 209–251. DOI: 10.1016/B978-0-323-41736-5.00007-8.
Article58. Bae KH, Chung HJ, Park TG. Nanomaterials for cancer therapy and imaging. Mol Cells. 2011; 31:295–302. DOI: 10.1007/s10059-011-0051-5. PMID: 21360197. PMCID: PMC3933969.
Article59. Liu Q, Jin C, Wang Y, Fang X, Zhang X, Chen Z, Tan W. Aptamer-conjugated nanomaterials for specific cancer cell recognition and targeted cancer therapy. NPG Asia Mater. 2014; 6:pii: e95. DOI: 10.1038/am.2014.12. PMID: 29619132. PMCID: PMC5880215.
Article60. Tan WB, Jiang S, Zhang Y. Quantum-dot based nanoparticles for targeted silencing of HER2/neu gene via RNA interference. Biomaterials. 2007; 28:1565–1571. DOI: 10.1016/j.biomaterials.2006.11.018. PMID: 17161865.
Article61. Savla R, Taratula O, Garbuzenko O, Minko T. Tumor targeted quantum dot-mucin 1 aptamer-doxorubicin conjugate for imaging and treatment of cancer. J Control Release. 2011; 153:16–22. DOI: 10.1016/j.jconrel.2011.02.015. PMID: 21342659.
Article62. Ruan J, Song H, Qian Q, Li C, Wang K, Bao C, Cui D. HER2 monoclonal antibody conjugated RNase-A-associated CdTe quantum dots for targeted imaging and therapy of gastric cancer. Biomaterials. 2012; 33:7093–7102. DOI: 10.1016/j.biomaterials.2012.06.053. PMID: 22796163.
Article63. Li Y, He H, Jia X, Lu WL, Lou J, Wei Y. A dual-targeting nanocarrier based on poly(amidoamine) dendrimers conjugated with transferrin and tamoxifen for treating brain gliomas. Biomaterials. 2012; 33:3899–3908. DOI: 10.1016/j.biomaterials.2012.02.004. PMID: 22364698.
Article64. Yang W, Cheng Y, Xu T, Wang X, Wen LP. Targeting cancer cells with biotin-dendrimer conjugates. Eur J Med Chem. 2009; 44:862–868. DOI: 10.1016/j.ejmech.2008.04.021. PMID: 18550227.
Article65. Montet X, Funovics M, Montet-Abou K, Weissleder R, Josephson L. Multivalent effects of RGD peptides obtained by nanoparticle display. J Med Chem. 2006; 49:6087–6093. DOI: 10.1021/jm060515m. PMID: 17004722.
Article66. Toma A, Otsuji E, Kuriu Y, Okamoto K, Ichikawa D, Hagiwara A, Ito H, Nishimura T, Yamagishi H. Monoclonal antibody A7-superparamagnetic iron oxide as contrast agent of MR imaging of rectal carcinoma. Br J Cancer. 2005; 93:131–136. DOI: 10.1038/sj.bjc.6602668. PMID: 15970924. PMCID: PMC2361484.
Article67. Chanda N, Kattumuri V, Shukla R, Zambre A, Katti K, Upendran A, Kulkarni RR, Kan P, Fent GM, Casteel SW, Smith CJ, Boote E, Robertson JD, Cutler C, Lever JR, Katti KV, Kannan R. Bombesin functionalized gold nanoparticles show in vitro and in vivo cancer receptor specificity. Proc Natl Acad Sci U S A. 2010; 107:8760–8765. DOI: 10.1073/pnas.1002143107. PMID: 20410458. PMCID: PMC2889350.
Article68. Patra CR, Bhattacharya R, Mukherjee P. Fabrication and functional characterization of goldnanoconjugates for potential application in ovarian cancer. J Mater Chem. 2010; 20:547–554. DOI: 10.1039/B913224D. PMID: 20436942. PMCID: PMC2860188.
Article69. Li JL, Wang L, Liu XY, Zhang ZP, Guo HC, Liu WM, Tang SH. In vitro cancer cell imaging and therapy using transferrin-conjugated gold nanoparticles. Cancer Lett. 2009; 274:319–326. DOI: 10.1016/j.canlet.2008.09.024. PMID: 18977071.
Article70. Li R, Wu R, Zhao L, Hu Z, Guo S, Pan X, Zou H. Folate and iron difunctionalized multiwall carbon nanotubes as dual-targeted drug nanocarrier to cancer cells. Carbon. 2011; 49:1797–1805. DOI: 10.1016/j.carbon.2011.01.003.
Article71. Yao HJ, Zhang YG, Sun L, Liu Y. The effect of hyaluronic acid functionalized carbon nanotubes loaded with salinomycin on gastric cancer stem cells. Biomaterials. 2014; 35:9208–9223. DOI: 10.1016/j.biomaterials.2014.07.033. PMID: 25115788.
Article72. Sun X, Liu Z, Welsher K, Robinson JT, Goodwin A, Zaric S, Dai H. Nano-graphene oxide for cellular imaging and drug delivery. Nano Res. 2008; 1:203–212. DOI: 10.1007/s12274-008-8021-8. PMID: 20216934. PMCID: PMC2834318.
Article73. Laginha KM, Moase EH, Yu N, Huang A, Allen TM. Bioavailability and therapeutic efficacy of HER2 scFv-targeted liposomal doxorubicin in a murine model of HER2-overexpressing breast cancer. J Drug Target. 2008; 16:605–610. DOI: 10.1080/10611860802229978. PMID: 18686132.
Article74. Chen Y, Liang H. Applications of quantum dots with up-converting luminescence in bioimaging. J Photochem Photobiol B. 2014; 135:23–32. DOI: 10.1016/j.jphotobiol.2014.04.003. PMID: 24792570.
Article75. Zhao Y, Liu S, Li Y, Jiang W, Chang Y, Pan S, Fang X, Wang YA, Wang J. Synthesis and grafting of folate-PEG-PAMAM conjugates onto quantum dots for selective targeting of folate-receptor-positive tumor cells. J Colloid Interface Sci. 2010; 350:44–50. DOI: 10.1016/j.jcis.2010.05.035. PMID: 20624622.
Article76. Pathakoti K, Hwang HM, Xu H, Aguilar ZP, Wang A. In vitro cytotoxicity of CdSe/ZnS quantum dots with different surface coatings to human keratinocytes HaCaT cells. J Environ Sci (China). 2013; 25:163–171. DOI: 10.1016/S1001-0742(12)60015-1. PMID: 23586311.
Article77. Hughes GA. Nanostructure-mediated drug delivery. Balogh LP, editor. Nanomedicine. Singapore: Pan Stanford Publishing;2017. p. 47–72.
Article78. Aulenta F, Hayes W, Rannard S. Dendrimers: a new class of nanoscopic containers and delivery devices. Eur Polym J. 2003; 39:1741–1771. DOI: 10.1016/S0014-3057(03)00100-9.
Article79. Najlah M, D’Emanuele A. Crossing cellular barriers using dendrimer nanotechnologies. Curr Opin Pharmacol. 2006; 6:522–527. DOI: 10.1016/j.coph.2006.05.004. PMID: 16890022.
Article80. Oliveira JM, Salgado AJ, Sousa N, Mano JF, Reis RL. Dendrimers and derivatives as a potential therapeutic tool in regenerative medicine strategies--a review. Prog Polym Sci. 2010; 35:1163–1194. DOI: 10.1016/j.progpolymsci.2010.04.006.
Article81. Pillai O, Panchagnula R. Polymers in drug delivery. Curr Opin Chem Biol. 2001; 5:447–451. DOI: 10.1016/S1367-5931(00)00227-1. PMID: 11470609.
Article82. Pissuwan D, Valenzuela SM, Cortie MB. Therapeutic possibilities of plasmonically heated gold nanoparticles. Trends Biotechnol. 2006; 24:62–67. DOI: 10.1016/j.tibtech.2005.12.004. PMID: 16380179.
Article83. Mahmoudi M, Sant S, Wang B, Laurent S, Sen T. Superparamagnetic iron oxide nanoparticles (SPIONs): development, surface modification and applications in chemotherapy. Adv Drug Deliv Rev. 2011; 63:24–46. DOI: 10.1016/j.addr.2010.05.006. PMID: 20685224.
Article84. Kodama R. Magnetic nanoparticles. J Magn Magn Mater. 1999; 200:359–372. DOI: 10.1016/S0304-8853(99)00347-9.
Article85. Neuberger T, Schöpf B, Hofmann H, Hofmann M, von Rechenberg B. Superparamagnetic nanoparticles for biomedical applications: possibilities and limitations of a new drug delivery system. J Magn Mater. 2005; 293:483–496. DOI: 10.1016/j.jmmm.2005.01.064.
Article86. Syková E, Jendelová P. Migration, fate and in vivo imaging of adult stem cells in the CNS. Cell Death Differ. 2007; 14:1336–1342. DOI: 10.1038/sj.cdd.4402140. PMID: 17396130.
Article87. Magrez A, Kasas S, Salicio V, Pasquier N, Seo JW, Celio M, Catsicas S, Schwaller B, Forró L. Cellular toxicity of carbon-based nanomaterials. Nano Lett. 2006; 6:1121–1125. DOI: 10.1021/nl060162e. PMID: 16771565.
Article88. Zhang BT, Zheng X, Li HF, Lin JM. Application of carbon-based nanomaterials in sample preparation: a review. Anal Chim Acta. 2013; 784:1–17. DOI: 10.1016/j.aca.2013.03.054. PMID: 23746402.
Article89. Scida K, Stege PW, Haby G, Messina GA, García CD. Recent applications of carbon-based nanomaterials in analytical chemistry: critical review. Anal Chim Acta. 2011; 691:6–17. DOI: 10.1016/j.aca.2011.02.025. PMID: 21458626. PMCID: PMC3088727.
Article90. Cha C, Shin SR, Annabi N, Dokmeci MR, Khademhosseini A. Carbon-based nanomaterials: multifunctional materials for biomedical engineering. ACS Nano. 2013; 7:2891–2897. DOI: 10.1021/nn401196a. PMID: 23560817. PMCID: PMC3648999.
Article91. Lasic DD. Novel applications of liposomes. Trends Biotechnol. 1998; 16:307–321. DOI: 10.1016/S0167-7799(98)01220-7. PMID: 9675915.
Article92. Fenske DB, Chonn A, Cullis PR. Liposomal nanomedicines: an emerging field. Toxicol Pathol. 2008; 36:21–29. DOI: 10.1177/0192623307310960. PMID: 18337218.
Article93. Lyakhovich A, Lleonart ME. Bypassing mechanisms of mitochondria-mediated cancer stem cells resistance to chemo-and radiotherapy. Oxid Med Cell Longev. 2016; 2016:1716341. DOI: 10.1155/2016/1716341. PMID: 26697128. PMCID: PMC4677234.94. Hong IS, Jang GB, Lee HY, Nam JS. Targeting cancer stem cells by using the nanoparticles. Int J Nanomedicine. 2015; 10:251–260. PMID: 26425092. PMCID: PMC4583536.95. Abetov D, Mustapova Z, Saliev T, Bulanin D, Batyrbekov K, Gilman CP. Novel small molecule inhibitors of cancer stem cell signaling pathways. Stem Cell Rev. 2015; 11:909–918. DOI: 10.1007/s12015-015-9612-x. PMID: 26210995.
Article96. He L, Gu J, Lim LY, Yuan ZX, Mo J. Nanomedicine-mediated therapies to target breast cancer stem cells. Front Pharmacol. 2016; 7:313. DOI: 10.3389/fphar.2016.00313. PMID: 27679576. PMCID: PMC5020043.
Article97. Wang Z, Tan J, McConville C, Kannappan V, Tawari PE, Brown J, Ding J, Armesilla AL, Irache JM, Mei QB, Tan Y, Liu Y, Jiang W, Bian XW, Wang W. Poly lactic-co-glycolic acid controlled delivery of disulfiram to target liver cancer stem-like cells. Nanomedicine. 2017; 13:641–657. DOI: 10.1016/j.nano.2016.08.001. PMCID: PMC5364371.
Article98. Estey T, Piatigorsky J, Lassen N, Vasiliou V. ALDH3A1: a corneal crystallin with diverse functions. Exp Eye Res. 2007; 84:3–12. DOI: 10.1016/j.exer.2006.04.010. PMID: 16797007.
Article99. Ginestier C, Hur MH, Charafe-Jauffret E, Monville F, Dutcher J, Brown M, Jacquemier J, Viens P, Kleer CG, Liu S, Schott A, Hayes D, Birnbaum D, Wicha MS, Dontu G. ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell. 2007; 1:555–567. DOI: 10.1016/j.stem.2007.08.014. PMID: 18371393. PMCID: PMC2423808.
Article100. Lu B, Huang X, Mo J, Zhao W. Drug delivery using nanoparticles for cancer stem-like cell targeting. Front Pharmacol. 2016; 7:84. DOI: 10.3389/fphar.2016.00084. PMID: 27148051. PMCID: PMC4828437.
Article101. Li D, Zhang T, Gu W, Li P, Cheng X, Tong T, Wang W. The ALDH1+ subpopulation of the human NMFH-1 cell line exhibits cancer stem-like characteristics. Oncol Rep. 2015; 33:2291–2298. DOI: 10.3892/or.2015.3842. PMID: 25760144.
Article102. Liu SY, Zheng PS. High aldehyde dehydrogenase activity identifies cancer stem cells in human cervical cancer. Oncotarget. 2013; 4:2462–2475. DOI: 10.18632/oncotarget.1578. PMID: 24318570. PMCID: PMC3926841.
Article103. Shao C, Sullivan JP, Girard L, Augustyn A, Yenerall P, Rodriguez-Canales J, Liu H, Behrens C, Shay JW, Wistuba II, Minna JD. Essential role of aldehyde dehydrogenase 1A3 for the maintenance of non-small cell lung cancer stem cells is associated with the STAT3 pathway. Clin Cancer Res. 2014; 20:4154–4166. DOI: 10.1158/1078-0432.CCR-13-3292. PMID: 24907115. PMCID: PMC4438754.
Article104. Yue L, Huang ZM, Fong S, Leong S, Jakowatz JG, Charruyer-Reinwald A, Wei M, Ghadially R. Targeting ALDH1 to decrease tumorigenicity, growth and metastasis of human melanoma. Melanoma Res. 2015; 25:138–148. DOI: 10.1097/CMR.0000000000000144. PMID: 25643237.
Article105. Vira D, Basak SK, Veena MS, Wang MB, Batra RK, Srivatsan ES. Cancer stem cells, microRNAs, and therapeutic strategies including natural products. Cancer Metastasis Rev. 2012; 31:733–751. DOI: 10.1007/s10555-012-9382-8. PMID: 22752409.
Article106. Yip NC, Fombon IS, Liu P, Brown S, Kannappan V, Armesilla AL, Xu B, Cassidy J, Darling JL, Wang W. Disulfiram modulated ROS-MAPK and NFκB pathways and targeted breast cancer cells with cancer stem cell-like properties. Br J Cancer. 2011; 104:1564–1574. DOI: 10.1038/bjc.2011.126. PMID: 21487404. PMCID: PMC3101904.
Article107. Chen D, Cui QC, Yang H, Dou QP. Disulfiram, a clinically used anti-alcoholism drug and copper-binding agent, induces apoptotic cell death in breast cancer cultures and xenografts via inhibition of the proteasome activity. Cancer Res. 2006; 66:10425–10433. DOI: 10.1158/0008-5472.CAN-06-2126. PMID: 17079463.
Article108. Kast RE, Boockvar JA, Brüning A, Cappello F, Chang WW, Cvek B, Dou QP, Duenas-Gonzalez A, Efferth T, Focosi D, Ghaffari SH, Karpel-Massler G, Ketola K, Khoshnevisan A, Keizman D, Magné N, Marosi C, McDonald K, Muñoz M, Paranjpe A, Pourgholami MH, Sardi I, Sella A, Srivenugopal KS, Tuccori M, Wang W, Wirtz CR, Halatsch ME. A conceptually new treatment approach for relapsed glioblastoma: coordinated undermining of survival paths with nine repurposed drugs (CUSP9) by the International Initiative for Accelerated Improvement of Glioblastoma Care. Oncotarget. 2013; 4:502–530. DOI: 10.18632/oncotarget.969. PMID: 23594434. PMCID: PMC3720600.
Article109. Hothi P, Martins TJ, Chen L, Deleyrolle L, Yoon JG, Reynolds B, Foltz G. High-throughput chemical screens identify disulfiram as an inhibitor of human glioblastoma stem cells. Oncotarget. 2012; 3:1124–1136. DOI: 10.18632/oncotarget.707. PMID: 23165409. PMCID: PMC3717950.
Article110. Wang W, McLeod HL, Cassidy J. Disulfiram-mediated inhibition of NF-kappaB activity enhances cytotoxicity of 5-fluorouracil in human colorectal cancer cell lines. Int J Cancer. 2003; 104:504–511. DOI: 10.1002/ijc.10972. PMID: 12584750.
Article111. Guo X, Xu B, Pandey S, Goessl E, Brown J, Armesilla AL, Darling JL, Wang W. Disulfiram/copper complex inhibiting NFkappaB activity and potentiating cytotoxic effect of gemcitabine on colon and breast cancer cell lines. Cancer Lett. 2010; 290:104–113. DOI: 10.1016/j.canlet.2009.09.002. PMID: 19782464.
Article112. Liu P, Brown S, Goktug T, Channathodiyil P, Kannappan V, Hugnot JP, Guichet PO, Bian X, Armesilla AL, Darling JL, Wang W. Cytotoxic effect of disulfiram/copper on human glioblastoma cell lines and ALDH-positive cancer-stem-like cells. Br J Cancer. 2012; 107:1488–1497. DOI: 10.1038/bjc.2012.442. PMID: 23033007. PMCID: PMC3493777.
Article113. Zöller M. CD44: can a cancer-initiating cell profit from an abundantly expressed molecule? Nat Rev Cancer. 2011; 11:254–267. DOI: 10.1038/nrc3023. PMID: 21390059.
Article114. Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci U S A. 2003; 100:3983–3988. DOI: 10.1073/pnas.0530291100. PMID: 12629218. PMCID: PMC153034.
Article115. Dalerba P, Dylla SJ, Park IK, Liu R, Wang X, Cho RW, Hoey T, Gurney A, Huang EH, Simeone DM, Shelton AA, Parmiani G, Castelli C, Clarke MF. Phenotypic characterization of human colorectal cancer stem cells. Proc Natl Acad Sci U S A. 2007; 104:10158–10163. DOI: 10.1073/pnas.0703478104. PMID: 17548814. PMCID: PMC1891215.
Article116. Deonarain MP, Kousparou CA, Epenetos AA. Antibodies targeting cancer stem cells: a new paradigm in immunotherapy? MAbs. 2009; 1:12–25. DOI: 10.4161/mabs.1.1.7347. PMID: 20046569. PMCID: PMC2715180.
Article117. Gener P, Gouveia LP, Sabat GR, de Sousa Rafael DF, Fort NB, Arranja A, Fernández Y, Prieto RM, Ortega JS, Arango D, Abasolo I, Videira M, Schwartz S Jr. Fluorescent CSC models evidence that targeted nanomedicines improve treatment sensitivity of breast and colon cancer stem cells. Nanomedicine. 2015; 11:1883–1892. DOI: 10.1016/j.nano.2015.07.009. PMID: 26238079.
Article118. Arabi L, Badiee A, Mosaffa F, Jaafari MR. Targeting CD44 expressing cancer cells with anti-CD44 monoclonal antibody improves cellular uptake and antitumor efficacy of liposomal doxorubicin. J Control Release. 2015; 220:275–286. DOI: 10.1016/j.jconrel.2015.10.044. PMID: 26518722.
Article119. Aires A, Ocampo SM, Simões BM, Josefa Rodríguez M, Cadenas JF, Couleaud P, Spence K, Latorre A, Miranda R, Somoza Á, Clarke RB, Carrascosa JL, Cortajarena AL. Multifunctionalized iron oxide nanoparticles for selective drug delivery to CD44-positive cancer cells. Nanotechnology. 2016; 27:065103. DOI: 10.1088/0957-4484/27/6/065103. PMID: 26754042.
Article120. Ni M, Xiong M, Zhang X, Cai G, Chen H, Zeng Q, Yu Z. Poly(lactic-co-glycolic acid) nanoparticles conjugated with CD133 aptamers for targeted salinomycin delivery to CD133+ osteosarcoma cancer stem cells. Int J Nanomedicine. 2015; 10:2537–2554. PMID: 25848270. PMCID: PMC4386781.121. Qi X, Yu D, Jia B, Jin C, Liu X, Zhao X, Zhang G. Targeting CD133(+) laryngeal carcinoma cells with chemotherapeutic drugs and siRNA against ABCG2 mediated by thermo/pH-sensitive mesoporous silica nanoparticles. Tumour Biol. 2016; 37:2209–2217. DOI: 10.1007/s13277-015-4007-9. PMID: 26353857.
Article122. Li Y, Zhang T. Targeting cancer stem cells by curcumin and clinical applications. Cancer Lett. 2014; 346:197–205. DOI: 10.1016/j.canlet.2014.01.012. PMID: 24463298.
Article123. Lim KJ, Bisht S, Bar EE, Maitra A, Eberhart CG. A polymeric nanoparticle formulation of curcumin inhibits growth, clonogenicity and stem-like fraction in malignant brain tumors. Cancer Biol Ther. 2011; 11:464–473. DOI: 10.4161/cbt.11.5.14410. PMID: 21193839. PMCID: PMC3087900.
Article124. Mumm JS, Kopan R. Notch signaling: from the outside in. Dev Biol. 2000; 228:151–165. DOI: 10.1006/dbio.2000.9960. PMID: 11112321.
Article125. Tang SN, Fu J, Nall D, Rodova M, Shankar S, Srivastava RK. Inhibition of sonic hedgehog pathway and pluripotency maintaining factors regulate human pancreatic cancer stem cell characteristics. Int J Cancer. 2012; 131:30–40. DOI: 10.1002/ijc.26323. PMID: 21796625. PMCID: PMC3480310.
Article126. Fan X, Matsui W, Khaki L, Stearns D, Chun J, Li YM, Eberhart CG. Notch pathway inhibition depletes stem-like cells and blocks engraftment in embryonal brain tumors. Cancer Res. 2006; 66:7445–7452. DOI: 10.1158/0008-5472.CAN-06-0858. PMID: 16885340.
Article127. Mamaeva V, Rosenholm JM, Bate-Eya LT, Bergman L, Peuhu E, Duchanoy A, Fortelius LE, Landor S, Toivola DM, Lindén M, Sahlgren C. Mesoporous silica nanoparticles as drug delivery systems for targeted inhibition of Notch signaling in cancer. Mol Ther. 2011; 19:1538–1546. DOI: 10.1038/mt.2011.105. PMID: 21629222. PMCID: PMC3149161.
Article128. Mamaeva V, Niemi R, Beck M, Özliseli E, Desai D, Landor S, Gronroos T, Kronqvist P, Pettersen IK, McCormack E, Rosenholm JM, Linden M, Sahlgren C. Inhibiting notch activity in breast cancer stem cells by glucose functionalized nanoparticles carrying γ-secretase inhibitors. Mol Ther. 2016; 24:926–936. DOI: 10.1038/mt.2016.42. PMID: 26916284. PMCID: PMC4881775.
Article129. Yang ZF, Ngai P, Ho DW, Yu WC, Ng MN, Lau CK, Li ML, Tam KH, Lam CT, Poon RT, Fan ST. Identification of local and circulating cancer stem cells in human liver cancer. Hepatology. 2008; 47:919–928. DOI: 10.1002/hep.22082. PMID: 18275073.
Article130. Bakalova R, Ohba H, Zhelev Z, Ishikawa M, Baba Y. Quantum dots as photosensitizers? Nat Biotechnol. 2004; 22:1360–1361. DOI: 10.1038/nbt1104-1360. PMID: 15529155.
Article131. Ruiz i Altaba A, Sánchez P, Dahmane N. Gli and hedgehog in cancer: tumours, embryos and stem cells. Nat Rev Cancer. 2002; 2:361–372. DOI: 10.1038/nrc796. PMID: 12044012.
Article132. Rubin LL, de Sauvage FJ. Targeting the Hedgehog pathway in cancer. Nat Rev Drug Discov. 2006; 5:1026–1033. DOI: 10.1038/nrd2086. PMID: 17139287.
Article133. Kiesslich T, Berr F, Alinger B, Kemmerling R, Pichler M, Ocker M, Neureiter D. Current status of therapeutic targeting of developmental signalling pathways in oncology. Curr Pharm Biotechnol. 2012; 13:2184–2220. DOI: 10.2174/138920112802502114. PMID: 21605074.
Article134. Chenna V, Hu C, Pramanik D, Aftab BT, Karikari C, Campbell NR, Hong SM, Zhao M, Rudek MA, Khan SR, Rudin CM, Maitra A. A polymeric nanoparticle encapsulated small-molecule inhibitor of Hedgehog signaling (NanoHHI) bypasses secondary mutational resistance to Smoothened antagonists. Mol Cancer Ther. 2012; 11:165–173. DOI: 10.1158/1535-7163.MCT-11-0341. PMID: 22027695. PMCID: PMC3256300.
Article135. Xu Y, Chenna V, Hu C, Sun HX, Khan M, Bai H, Yang XR, Zhu QF, Sun YF, Maitra A, Fan J, Anders RA. Polymeric nanoparticle-encapsulated hedgehog pathway inhibitor HPI-1 (NanoHHI) inhibits systemic metastases in an orthotopic model of human hepatocellular carcinoma. Clin Cancer Res. 2012; 18:1291–1302. DOI: 10.1158/1078-0432.CCR-11-0950. PMID: 21868763. PMCID: PMC3233659.
Article136. Verma RK, Yu W, Singh SP, Shankar S, Srivastava RK. Anthothecol-encapsulated PLGA nanoparticles inhibit pancreatic cancer stem cell growth by modulating sonic hedgehog pathway. Nanomedicine. 2015; 11:2061–2070. DOI: 10.1016/j.nano.2015.07.001. PMID: 26199979.
Article137. Moses H, Barcellos-Hoff MH. TGF-beta biology in mammary development and breast cancer. Cold Spring Harb Perspect Biol. 2011; 3:a003277. DOI: 10.1101/cshperspect.a003277. PMID: 20810549. PMCID: PMC3003461.138. Bellam N, Pasche B. Tgf-beta signaling alterations and colon cancer. Cancer Treat Res. 2010; 155:85–103. DOI: 10.1007/978-1-4419-6033-7_5. PMID: 20517689.139. Wu K, Ding J, Chen C, Sun W, Ning BF, Wen W, Huang L, Han T, Yang W, Wang C, Li Z, Wu MC, Feng GS, Xie WF, Wang HY. Hepatic transforming growth factor beta gives rise to tumor-initiating cells and promotes liver cancer development. Hepatology. 2012; 56:2255–2267. DOI: 10.1002/hep.26007. PMID: 22898879.
Article140. Ischenko I, Liu J, Petrenko O, Hayman MJ. Transforming growth factor-beta signaling network regulates plasticity and lineage commitment of lung cancer cells. Cell Death Differ. 2014; 21:1218–1228. DOI: 10.1038/cdd.2014.38. PMID: 24682004. PMCID: PMC4085528.
Article141. Gao J, Zhu Y, Nilsson M, Sundfeldt K. TGF-β isoforms induce EMT independent migration of ovarian cancer cells. Cancer Cell Int. 2014; 14:72. DOI: 10.1186/s12935-014-0072-1. PMID: 25278811. PMCID: PMC4180856.
Article142. Liu Z, Bandyopadhyay A, Nichols RW, Wang L, Hinck AP, Wang S, Sun LZ. Blockade of autocrine TGF-β signaling inhibits stem cell phenotype, survival, and metastasis of murine breast cancer cells. J Stem Cell Res Ther. 2012; 2:1–8. DOI: 10.4172/2157-7633.1000116. PMID: 23482850. PMCID: PMC3593047.
Article143. Mishra L, Shetty K, Tang Y, Stuart A, Byers SW. The role of TGF-beta and Wnt signaling in gastrointestinal stem cells and cancer. Oncogene. 2005; 24:5775–5789. DOI: 10.1038/sj.onc.1208924. PMID: 16123810.
Article144. Amin R, Mishra L. Liver stem cells and tgf-Beta in hepatic carcinogenesis. Gastrointest Cancer Res. 2008; 2(4 Suppl):S27–S30. PMID: 19343145. PMCID: PMC2661545.145. Gomez-Casal R, Bhattacharya C, Ganesh N, Bailey L, Basse P, Gibson M, Epperly M, Levina V. Non-small cell lung cancer cells survived ionizing radiation treatment display cancer stem cell and epithelial-mesenchymal transition phenotypes. Mol Cancer. 2013; 12:94. DOI: 10.1186/1476-4598-12-94. PMID: 23947765. PMCID: PMC3751356.
Article146. Meng H, Zhao Y, Dong J, Xue M, Lin YS, Ji Z, Mai WX, Zhang H, Chang CH, Brinker CJ, Zink JI, Nel AE. Two-wave nanotherapy to target the stroma and optimize gemcitabine delivery to a human pancreatic cancer model in mice. ACS Nano. 2013; 7:10048–10065. DOI: 10.1021/nn404083m. PMID: 24143858. PMCID: PMC3878438.
Article147. Zuo ZQ, Chen KG, Yu XY, Zhao G, Shen S, Cao ZT, Luo YL, Wang YC, Wang J. Promoting tumor penetration of nanoparticles for cancer stem cell therapy by TGF-β signaling pathway inhibition. Biomaterials. 2016; 82:48–59. DOI: 10.1016/j.biomaterials.2015.12.014. PMID: 26751819.
Article148. Tsai YS, Chen YH, Cheng PC, Tsai HT, Shiau AL, Tzai TS, Wu CL. TGF-β1 conjugated to gold nanoparticles results in protein conformational changes and attenuates the biological function. Small. 2013; 9:2119–2128. DOI: 10.1002/smll.201202755. PMID: 23335450.
Article149. Singh VK, Saini A, Chandra R. The implications and future perspectives of nanomedicine for cancer stem cell targeted therapies. Front Mol Biosci. 2017; 4:52. DOI: 10.3389/fmolb.2017.00052. PMID: 28785557. PMCID: PMC5520001.
Article150. Zhao Y, Alakhova DY, Kabanov AV. Can nanomedicines kill cancer stem cells? Adv Drug Deliv Rev. 2013; 65:1763–1783. DOI: 10.1016/j.addr.2013.09.016. PMID: 24120657. PMCID: PMC4174448.
Article151. Youn YS, Bae YH. Perspectives on the past, present, and future of cancer nanomedicine. Adv Drug Deliv Rev. 2018; 130:3–11. DOI: 10.1016/j.addr.2018.05.008. PMID: 29778902.
Article152. Tabassum N, Verma V, Kumar M, Kumar A, Singh B. Nanomedicine in cancer stem cell therapy: from fringe to forefront. Cell Tissue Res. 2018; 374:427–438. DOI: 10.1007/s00441-018-2928-5. PMID: 30302547.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical Application of Nanotechnology
- Advances of Cancer Therapy by Nanotechnology
- Specific Protein Markers for Stem Cell Cross-Talk with Neighboring Cells in the Environment
- Gastric Cancer Stem Cells: Mechanisms and Therapeutic Approaches
- NIRF Heptamethine Cyanine Dye Nanocomplexes for Multi Modal Theranosis of Tumors