Intest Res.
2019 Oct;17(4):516-526. 10.5217/ir.2018.00169.
Clinical outcomes of positive resection margin after endoscopic mucosal resection of early colon cancers
- Affiliations
-
- 1Department of Internal Medicine, Soonchunhyang University College of Medicine, Seoul, Korea. medgun@schmc.ac.kr
- 2Department of Pathology, Soonchunhyang University College of Medicine, Seoul, Korea.
- KMID: 2465817
- DOI: http://doi.org/10.5217/ir.2018.00169
Abstract
- BACKGROUND/AIMS
When determining the subsequent management after endoscopic resection of the early colon cancer (ECC), various factors including the margin status should be considered. This study assessed the subsequent management and outcomes of ECCs according to margin status.
METHODS
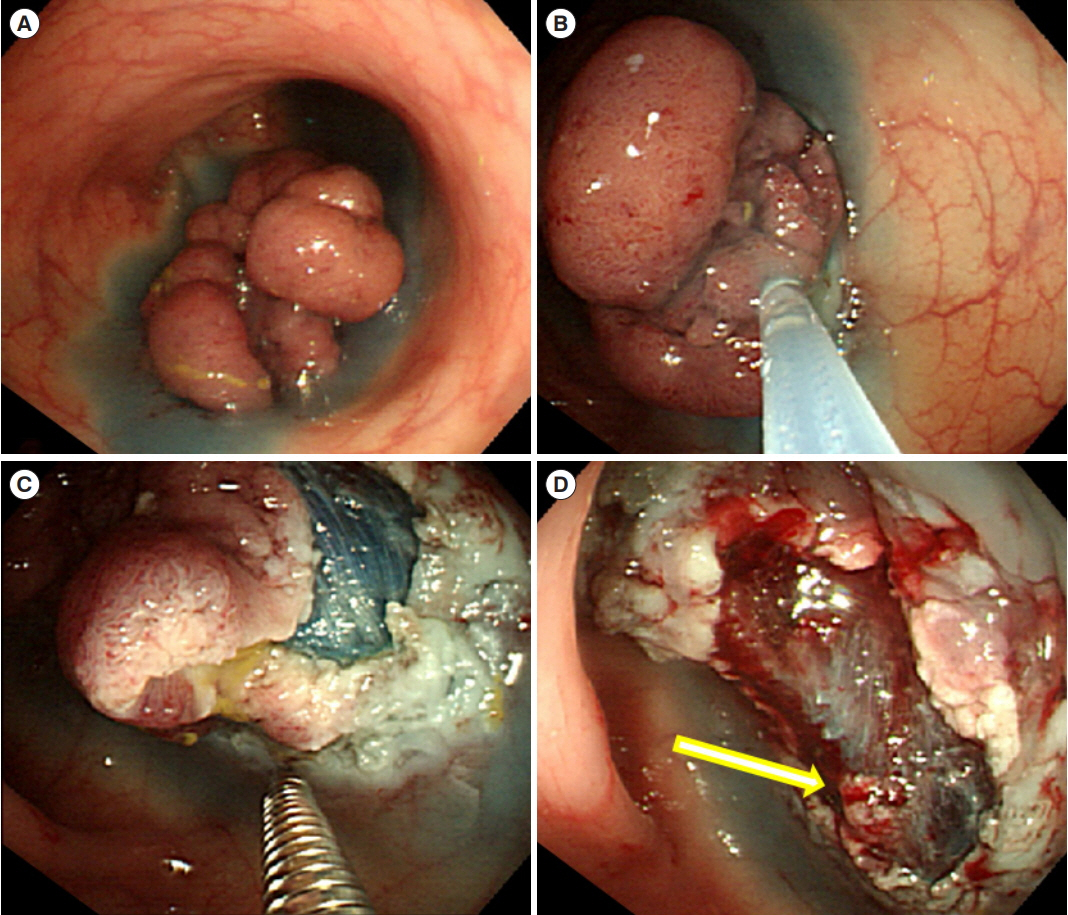
We examined the data of 223 ECCs treated by endoscopic mucosal resection (EMR) from 215 patients during 2004 to 2014, and all patients were followed-up at least for 2 years.
RESULTS
According to histological analyses, the margin statuses of all lesions after EMR were as follows: 138 cases (61.9%) were negative, 65 cases (29.1%) were positive for dysplastic cells on the resection margins, and 20 cases (8.9%) were uncertain. The decision regarding subsequent management was affected not only by pathologic outcomes but also by the endoscopist's opinion on whether complete resection was obtained. Surgery was preferred if the lesion extended to the submucosa (odds ratio [OR], 25.46; 95% confidence interval [CI], 7.09-91.42), the endoscopic resection was presumed incomplete (OR, 15.55; 95% CI, 4.28-56.56), or the lymph system was invaded (OR, 13.69; 95% CI, 1.76-106.57). Fourteen patients (6.2%) had residual or recurrent malignancies at the site of the previous ECC resection and were significantly associated with presumed incomplete endoscopic resection (OR, 4.59; 95% CI, 1.21-17.39) and submucosal invasion (OR, 5.14; 95% CI, 1.18-22.34).
CONCLUSIONS
Subsequent surgery was associated with submucosa invasion, lymphatic invasion, and cancer-positive margins. Presumed completeness of the resection may be helpful for guiding the subsequent management of patients who undergo endoscopic resection of ECC.
MeSH Terms
Figure
Cited by 1 articles
-
Clinical outcomes of submucosal colorectal cancer diagnosed after endoscopic resection: a focus on the need for surgery
Yun Sik Choi, Wan Soo Kim, Sung Wook Hwang, Sang Hyoung Park, Dong-Hoon Yang, Byong Duk Ye, Seung-Jae Myung, Suk-Kyun Yang, Jeong-Sik Byeon
Intest Res. 2020;18(1):96-106. doi: 10.5217/ir.2019.00092.
Reference
-
1. Okuyama T, Oya M, Ishikawa H. Budding as a risk factor for lymph node metastasis in pT1 or pT2 well-differentiated colorectal adenocarcinoma. Dis Colon Rectum. 2002; 45:628–634.2. Yim K, Won DD, Lee IK, Oh ST, Jung ES, Lee SH. Novel predictors for lymph node metastasis in submucosal invasive colorectal carcinoma. World J Gastroenterol. 2017; 23:5936–5944.3. Ueno H, Hase K, Hashiguchi Y, et al. Novel risk factors for lymph node metastasis in early invasive colorectal cancer: a multiinstitution pathology review. J Gastroenterol. 2014; 49:1314–1323.
Article4. Bosch SL, Teerenstra S, de Wilt JH, Cunningham C, Nagtegaal ID. Predicting lymph node metastasis in pT1 colorectal cancer: a systematic review of risk factors providing rationale for therapy decisions. Endoscopy. 2013; 45:827–834.
Article5. Suh JH, Han KS, Kim BC, et al. Predictors for lymph node metastasis in T1 colorectal cancer. Endoscopy. 2012; 44:590–595.6. Beaton C, Twine CP, Williams GL, Radcliffe AG. Systematic review and meta-analysis of histopathological factors influencing the risk of lymph node metastasis in early colorectal cancer. Colorectal Dis. 2013; 15:788–797.7. Kitajima K, Fujimori T, Fujii S, et al. Correlations between lymph node metastasis and depth of submucosal invasion in submucosal invasive colorectal carcinoma: a Japanese collaborative study. J Gastroenterol. 2004; 39:534–543.8. Cooper HS, Deppisch LM, Gourley WK, et al. Endoscopically removed malignant colorectal polyps: clinicopathologic correlations. Gastroenterology. 1995; 108:1657–1665.9. Doornebosch PG, Tollenaar RA, De Graaf EJ. Is the increasing role of Transanal Endoscopic Microsurgery in curation for T1 rectal cancer justified? A systematic review. Acta Oncol. 2009; 48:343–353.10. Cunningham KN, Mills LR, Schuman BM, Mwakyusa DH. Long-term prognosis of well-differentiated adenocarcinoma in endoscopically removed colorectal adenomas. Dig Dis Sci. 1994; 39:2034–2037.11. Volk EE, Goldblum JR, Petras RE, Carey WD, Fazio VW. Management and outcome of patients with invasive carcinoma arising in colorectal polyps. Gastroenterology. 1995; 109:1801–1807.12. Gill MD, Rutter MD, Holtham SJ. Management and short-term outcome of malignant colorectal polyps in the north of England(1). Colorectal Dis. 2013; 15:169–176.13. Borschitz T, Gockel I, Kiesslich R, Junginger T. Oncological outcome after local excision of rectal carcinomas. Ann Surg Oncol. 2008; 15:3101–3108.
Article14. Bach SP, Hill J, Monson JR, et al. A predictive model for local recurrence after transanal endoscopic microsurgery for rectal cancer. Br J Surg. 2009; 96:280–290.
Article15. Shin JW, Han KS, Hyun JH, et al. Risk of recurrence after endoscopic resection of early colorectal cancer with positive margins. Endoscopy. 2018; 50:241–247.16. Kim KM, Eo SJ, Shim SG, et al. Risk factors for residual cancer and lymph node metastasis after noncurative endoscopic resection of early colorectal cancer. Dis Colon Rectum. 2013; 56:35–42.17. Schlemper RJ, Hirata I, Dixon MF. The macroscopic classification of early neoplasia of the digestive tract. Endoscopy. 2002; 34:163–168.
Article18. Zauber AG, Winawer SJ, O’Brien MJ, et al. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med. 2012; 366:687–696.19. Lambert R, Kudo SE, Vieth M, et al. Pragmatic classification of superficial neoplastic colorectal lesions. Gastrointest Endosc. 2009; 70:1182–1199.
Article20. ASGE Technology Committee, Kantsevoy SV, Adler DG, et al. Endoscopic mucosal resection and endoscopic submucosal dissection. Gastrointest Endosc. 2008; 68:11–18.
Article21. Watanabe T, Itabashi M, Shimada Y, et al. Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2014 for treatment of colorectal cancer. Int J Clin Oncol. 2015; 20:207–239.22. Bujanda L, Cosme A, Gil I, Arenas-Mirave JI. Malignant colorectal polyps. World J Gastroenterol. 2010; 16:3103–3111.
Article23. Butte JM, Tang P, Gonen M, et al. Rate of residual disease after complete endoscopic resection of malignant colonic polyp. Dis Colon Rectum. 2012; 55:122–127.
Article24. ASGE Standards of Practice Committee, Fisher DA, Shergill AK, et al. Role of endoscopy in the staging and management of colorectal cancer. Gastrointest Endosc. 2013; 78:8–12.
Article25. Goldstein NS, Watts JC, Neill JS, et al. The effect of electrothermal cautery-assisted resection of diminutive colonic polyps on histopathologic diagnosis. Am J Clin Pathol. 2001; 115:356–361.26. Groisman GM, Amar M, Meir A. Utility of MIB-1 (Ki-67) in evaluating diminutive colorectal polyps with cautery artifact. Arch Pathol Lab Med. 2007; 131:1089–1093.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Tips and Tricks for Better Endoscopic Treatment of Colorectal Tumors: Usefulness of Cap and Band in Colorectal Endoscopic Mucosal Resection
- Treatment Strategy after Incomplete Endoscopic Resection of Early Gastric Cancer
- Therapeutic approach to non-curative resection after endoscopic treatment in early gastric cancer
- A Case of Early Esophageal Cancer Treated by Endoscopic Mucosal Resection Using a EEMR Tube
- Endoscopic Resection of Early Gastric Cancer