Korean Circ J.
2019 May;49(5):437-445. 10.4070/kcj.2018.0323.
Assessing Accuracy of Wrist-Worn Wearable Devices in Measurement of Paroxysmal Supraventricular Tachycardia Heart Rate
- Affiliations
-
- 1Division of Cardiology, Department of Internal Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. mdjunkim@gmail.com
- 2Division of Cardiology, Department of Internal Medicine, Keimyung University Dongsan Medical Center, Daegu, Korea.
- KMID: 2444963
- DOI: http://doi.org/10.4070/kcj.2018.0323
Abstract
- BACKGROUND AND OBJECTIVES
Wrist-worn wearable devices provide heart rate (HR) monitoring function via photoplethysmography technology. Recently, these devices have been used by patients to measure the HR when palpitation occurs, but few validation studies of these instruments have been conducted. We assessed the accuracy of these devices for measuring a HR.
METHODS
This study enrolled 51 consecutive patients with a history of paroxysmal supraventricular tachyarrhythmia (SVT) or paroxysmal palpitations who were scheduled to undergo an electrophysiological study (EPS). Three devices were assessed: Apple Watch Series 2 (Apple), Samsung Galaxy Gear S3 (Galaxy), and Fitbit Charge 2 (Fitbit). Patients were randomly assigned to wear 2 different devices. The HR at baseline and induced SVT were measured during the EPS. After successful ablation of SVT, HR measurements was also done during atrial and ventricular pacing study.
RESULTS
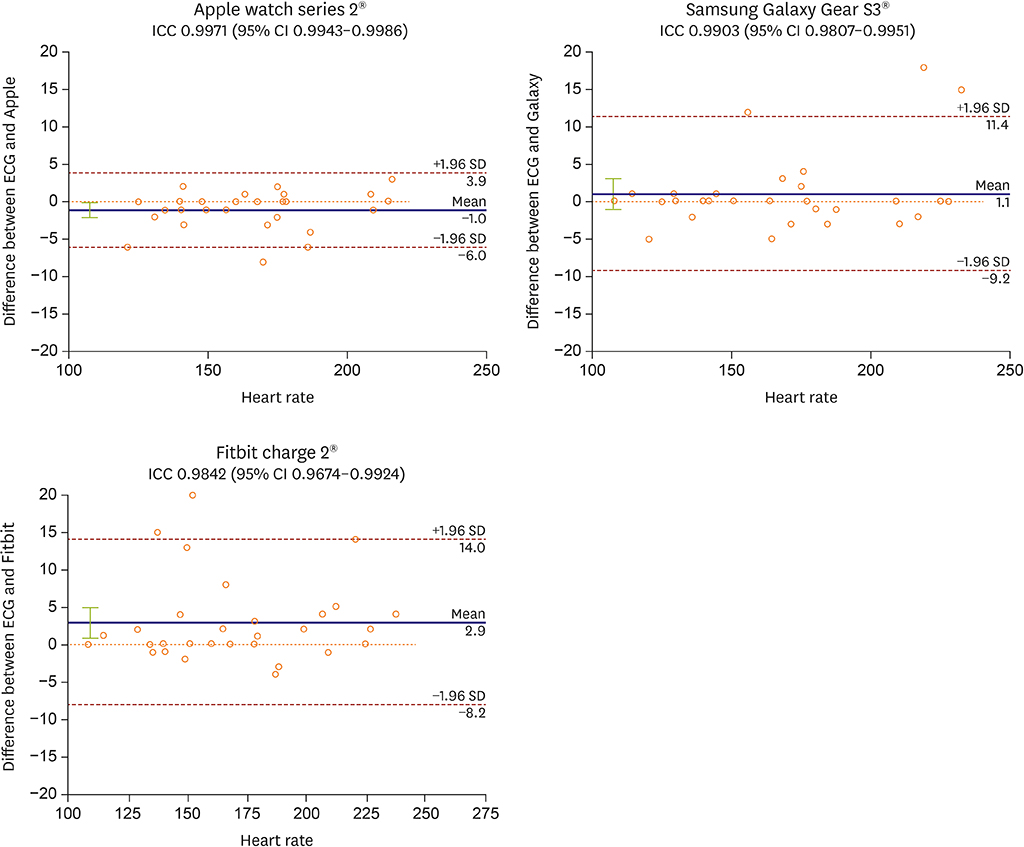
The mean patient age was 44.4±16.6 years and 27 patients were male (53%). The accuracy (within ±5 beats per minute [bpm] of an electrocardiogram [ECG] measurement) of the baseline HR measurements was 100%, 100%, and 94%, for Apple, Galaxy, and Fitbit, respectively. The HR during induced SVT ranged from 108 bpm to 228 bpm and the accuracy (within ±10 bpm of an ECG) was 100%, 90%, and 87% for the Apple, Galaxy, and Fitbit, respectively. During pacing study, accuracy of these devices was also acceptable but tended to decrease as the HR increased, and showed differences between the devices.
CONCLUSIONS
Wrist-worn wearable devices accurately measure baseline and induced SVT HR. TRIAL REGISTRATION: Clinical Research Information Service Identifier: KCT0002282
Keyword
MeSH Terms
Figure
Cited by 2 articles
-
Comparison of Continuous ECG Monitoring by Wearable Patch Device and Conventional Telemonitoring Device
Wonsuk Choi, Sun-Hwa Kim, Wonjae Lee, Si-Hyuck Kang, Chang-Hwan Yoon, Tae-Jin Youn, In-Ho Chae
J Korean Med Sci. 2020;35(44):e363. doi: 10.3346/jkms.2020.35.e363.Evolution of Smart Health Wearables: Novel Application for Detection of Arrhythmia
Jumsuk Ko, Nam-Ho Kim
Korean Circ J. 2019;49(5):446-447. doi: 10.4070/kcj.2019.0078.
Reference
-
1. Carpenter A, Frontera A. Smart-watches: a potential challenger to the implantable loop recorder? Europace. 2016; 18:791–793.
Article2. Scully CG, Lee J, Meyer J, et al. Physiological parameter monitoring from optical recordings with a mobile phone. IEEE Trans Biomed Eng. 2012; 59:303–306.
Article3. McManus DD, Lee J, Maitas O, et al. A novel application for the detection of an irregular pulse using an iPhone 4S in patients with atrial fibrillation. Heart Rhythm. 2013; 10:315–319.
Article4. Chan PH, Wong CK, Poh YC, et al. Diagnostic performance of a smartphone-based photoplethysmographic application for atrial fibrillation screening in a primary care setting. J Am Heart Assoc. 2016; 5:e003428.
Article5. McMANUS DD, Chong JW, Soni A, et al. PULSE-SMART: pulse-based arrhythmia discrimination using a novel smartphone application. J Cardiovasc Electrophysiol. 2016; 27:51–57.
Article6. Wang R, Blackburn G, Desai M, et al. Accuracy of wrist-worn heart rate monitors. JAMA Cardiol. 2017; 2:104–106.
Article7. Allen J. Photoplethysmography and its application in clinical physiological measurement. Physiol Meas. 2007; 28:R1–R39.
Article8. Halcox JP, Wareham K, Cardew A, et al. Assessment of remote heart rhythm sampling using the AliveCor heart monitor to screen for atrial fibrillation: the REHEARSE-AF study. Circulation. 2017; 136:1784–1794.9. Case MA, Burwick HA, Volpp KG, Patel MS. Accuracy of smartphone applications and wearable devices for tracking physical activity data. JAMA. 2015; 313:625–626.
Article10. Wackel P, Beerman L, West L, Arora G. Tachycardia detection using smartphone applications in pediatric patients. J Pediatr. 2014; 164:1133–1135.
Article11. Spierer DK, Rosen Z, Litman LL, Fujii K. Validation of photoplethysmography as a method to detect heart rate during rest and exercise. J Med Eng Technol. 2015; 39:264–271.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A case of broad QRS paroxysmall supraventricular tachycardia that is difficult to differentiate from ventricular tachycardia
- Paroxysmal Supraventriculer Tachycardia in Infants and Children
- Is the Analysis of ST Segment and T Wave during Tachycardia Useful in the Differential Diagnosis of Paroxysmal Supraventricular Tachycardia
- Differential Diagnosis of Supraventricular Tachycardia
- Emergency cesarean delivery in a parturient who had an intractable paroxysmal supraventricular tachycardia: A case report