J Breast Cancer.
2018 Mar;21(1):37-44. 10.4048/jbc.2018.21.1.37.
Endocrine Treatment-Related Symptoms and Patient Outcomes in Breast Cancer: A Meta-Analysis
- Affiliations
-
- 1Department of Surgery, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- 2Medical Research Collaborating Center, Seoul National University Hospital, Seoul, Korea.
- 3Department of Surgery, Seoul National University College of Medicine, Seoul, Korea. hanw@snu.ac.kr
- 4Laboratory of Breast Cancer Biology, Cancer Research Institute, Seoul National University College of Medicine, Seoul, Korea.
- KMID: 2441866
- DOI: http://doi.org/10.4048/jbc.2018.21.1.37
Abstract
- PURPOSE
An association between endocrine treatment-related symptoms and breast cancer recurrence has been suggested previously; however, conflicting results have been reported. We performed a meta-analysis of published studies to clarify this relationship.
METHODS
We systematically searched PubMed, Embase, Scopus, and the Cochrane database for studies investigating the association between endocrine treatment-related symptoms and patient survival. Random-effects meta-analysis was conducted with recurrence rate as the primary outcome.
RESULTS
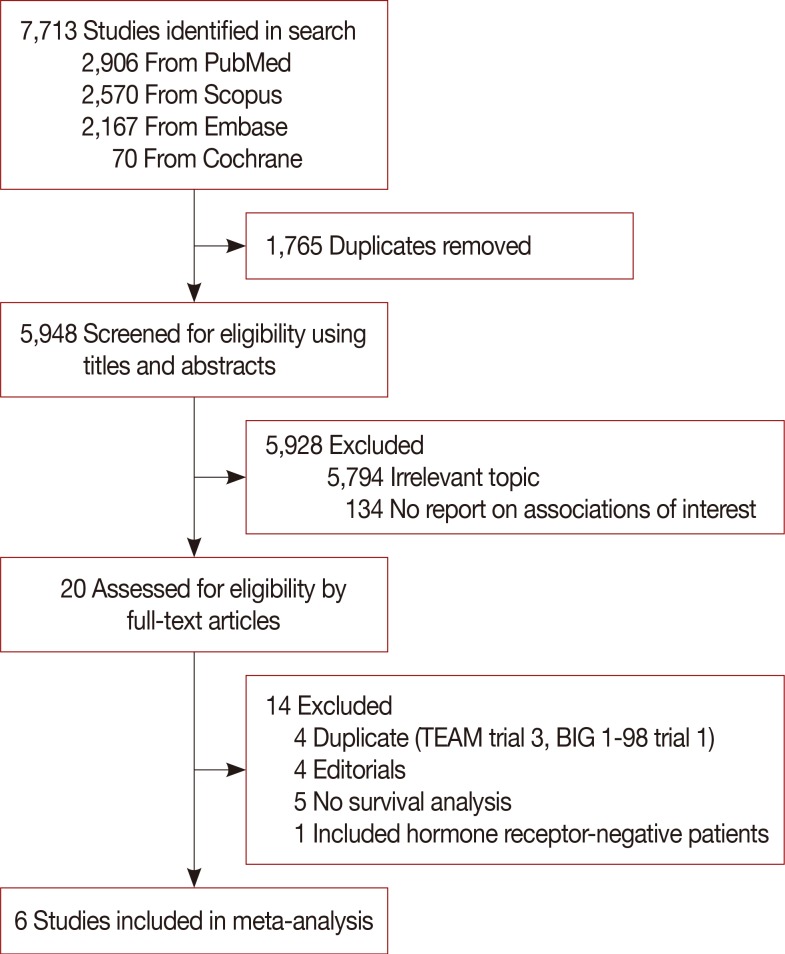
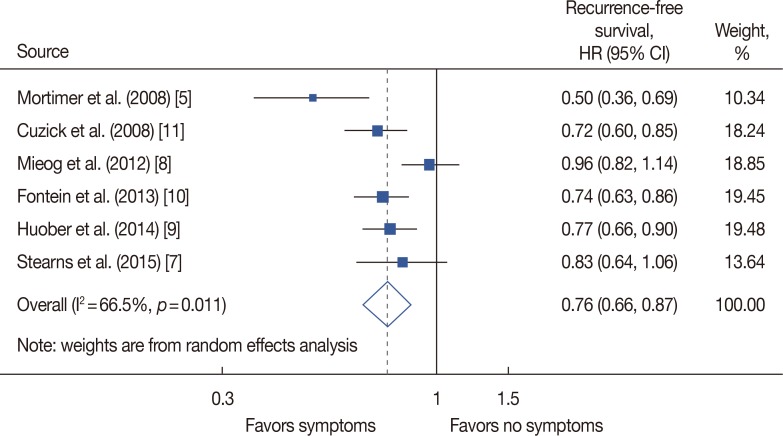
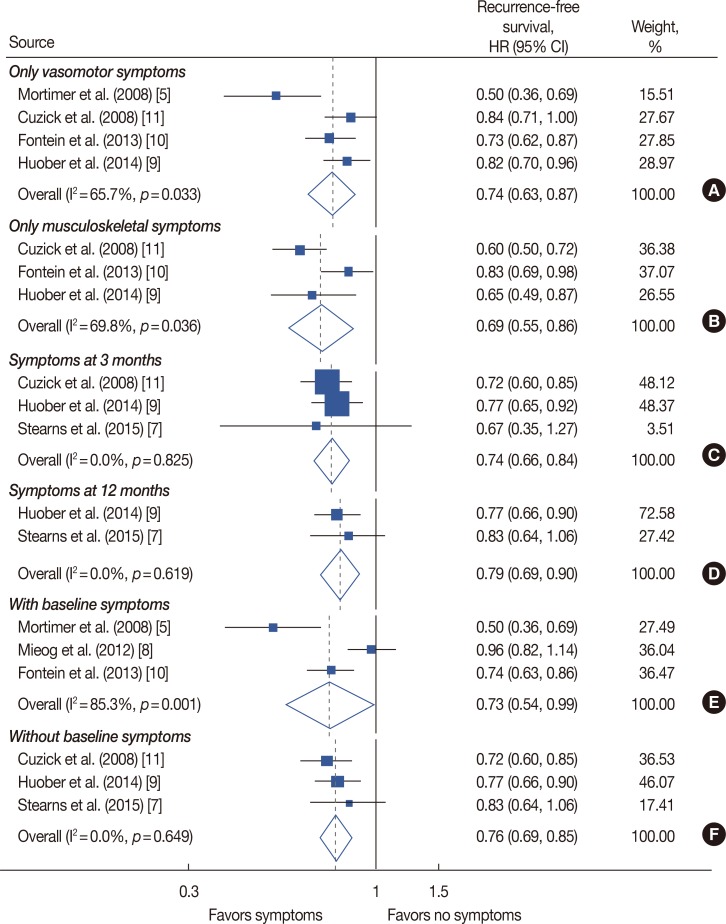
Out of 7,713 retrieved articles, six studies were included. In patients who received endocrine treatment, the presence of any endocrine treatment-related symptom was found to be associated with a lower recurrence rate in comparison to an absence of any symptoms (hazard ratio [HR], 0.76; 95% confidence interval [CI], 0.66-0.87). This relationship persisted in patients presenting with only vasomotor or only musculoskeletal symptoms (HR, 0.74, 95% CI, 0.63-0.87; HR, 0.69, 95% CI, 0.55-0.86, respectively). At both time-points of symptom evaluation (3 months and 12 months), patients with endocrine treatment-related symptoms had a lower recurrence rate (HR, 0.74, 95% CI, 0.66-0.84; HR, 0.79, 95% CI, 0.69-0.90, respectively). This association was also significant in pooled studies including patients with and without baseline symptoms (HR, 0.73, 95% CI, 0.54-0.99; HR, 0.76, 95% CI, 0.69-0.85, respectively).
CONCLUSION
Endocrine treatment-related symptoms are significantly predictive of lower recurrence rate in breast cancer patients, regardless of the type of symptoms, time-point of evaluation, or inclusion of baseline symptoms. These symptoms could be biomarkers for the prediction of long-term responses to endocrine treatment in patients with breast cancer.
Keyword
MeSH Terms
Figure
Reference
-
1. Ganz PA, Cecchini RS, Julian TB, Margolese RG, Costantino JP, Vallow LA, et al. Patient-reported outcomes with anastrozole versus tamoxifen for postmenopausal patients with ductal carcinoma in situ treated with lumpectomy plus radiotherapy (NSABP B-35): a randomised, doubleblind, phase 3 clinical trial. Lancet. 2016; 387:857–865. PMID: 26686960.
Article2. Grunfeld EA, Hunter MS, Sikka P, Mittal S. Adherence beliefs among breast cancer patients taking tamoxifen. Patient Educ Couns. 2005; 59:97–102. PMID: 16198223.
Article3. Ding K, Pater J, Whitehead M, Seymour L, Shepherd FA. Validation of treatment induced specific adverse effect as a predictor of treatment benefit: a case study of NCIC CTG BR21. Contemp Clin Trials. 2008; 29:527–536. PMID: 18280222.
Article4. Zielinski C, Lang I, Beslija S, Kahan Z, Inbar MJ, Stemmer SM, et al. Predictive role of hand-foot syndrome in patients receiving first-line capecitabine plus bevacizumab for HER2-negative metastatic breast cancer. Br J Cancer. 2016; 114:163–170. PMID: 26657657.
Article5. Mortimer JE, Flatt SW, Parker BA, Gold EB, Wasserman L, Natarajan L, et al. Tamoxifen, hot flashes and recurrence in breast cancer. Breast Cancer Res Treat. 2008; 108:421–426. PMID: 17541741.
Article6. Goetz MP, Rae JM, Suman VJ, Safgren SL, Ames MM, Visscher DW, et al. Pharmacogenetics of tamoxifen biotransformation is associated with clinical outcomes of efficacy and hot flashes. J Clin Oncol. 2005; 23:9312–9318. PMID: 16361630.
Article7. Stearns V, Chapman JA, Ma CX, Ellis MJ, Ingle JN, Pritchard KI, et al. Treatment-associated musculoskeletal and vasomotor symptoms and relapse-free survival in the NCIC CTG MA.27 adjuvant breast cancer aromatase inhibitor trial. J Clin Oncol. 2015; 33:265–271. PMID: 25512454.
Article8. Mieog JS, Morden JP, Bliss JM, Coombes RC, van de Velde CJ. IES Steering Committee. Carpal tunnel syndrome and musculoskeletal symptoms in postmenopausal women with early breast cancer treated with exemestane or tamoxifen after 2–3 years of tamoxifen: a retrospective analysis of the Intergroup Exemestane Study. Lancet Oncol. 2012; 13:420–432. PMID: 22265698.
Article9. Huober J, Cole BF, Rabaglio M, Giobbie-Hurder A, Wu J, Ejlertsen B, et al. Symptoms of endocrine treatment and outcome in the BIG 1-98 study. Breast Cancer Res Treat. 2014; 143:159–169. PMID: 24305979.
Article10. Fontein DB, Seynaeve C, Hadji P, Hille ET, van de Water W, Putter H, et al. Specific adverse events predict survival benefit in patients treated with tamoxifen or aromatase inhibitors: an international tamoxifen exemestane adjuvant multinational trial analysis. J Clin Oncol. 2013; 31:2257–2264. PMID: 23610112.
Article11. Cuzick J, Sestak I, Cella D, Fallowfield L. ATAC Trialists' Group. Treatment-emergent endocrine symptoms and the risk of breast cancer recurrence: a retrospective analysis of the ATAC trial. Lancet Oncol. 2008; 9:1143–1148. PMID: 18976959.
Article12. Moher D, Liberati A, Tetzlaff J, Altman DG. PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009; 6:e1000097. PMID: 19621072.
Article13. Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016; 355:i4919. PMID: 27733354.
Article14. Chirgwin JH, Giobbie-Hurder A, Coates AS, Price KN, Ejlertsen B, Debled M, et al. Treatment adherence and its impact on disease-free survival in the Breast International Group 1-98 Trial of tamoxifen and letrozole, alone and in sequence. J Clin Oncol. 2016; 34:2452–2459. PMID: 27217455.
Article15. van de Water W, Bastiaannet E, Hille ET, Meershoek-Klein Kranenbarg EM, Putter H, Seynaeve CM, et al. Age-specific nonpersistence of endocrine therapy in postmenopausal patients diagnosed with hormone receptor-positive breast cancer: a TEAM study analysis. Oncologist. 2012; 17:55–63. PMID: 22210087.16. Coombes RC, Hall E, Gibson LJ, Paridaens R, Jassem J, Delozier T, et al. A randomized trial of exemestane after two to three years of tamoxifen therapy in postmenopausal women with primary breast cancer. N Engl J Med. 2004; 350:1081–1092. PMID: 15014181.
Article17. Hershman DL, Kushi LH, Shao T, Buono D, Kershenbaum A, Tsai WY, et al. Early discontinuation and nonadherence to adjuvant hormonal therapy in a cohort of 8,769 early-stage breast cancer patients. J Clin Oncol. 2010; 28:4120–4128. PMID: 20585090.
Article18. Nekhlyudov L, Li L, Ross-Degnan D, Wagner AK. Five-year patterns of adjuvant hormonal therapy use, persistence, and adherence among insured women with early-stage breast cancer. Breast Cancer Res Treat. 2011; 130:681–689. PMID: 21842245.
Article19. Henry NL, Azzouz F, Desta Z, Li L, Nguyen AT, Lemler S, et al. Predictors of aromatase inhibitor discontinuation as a result of treatment-emergent symptoms in early-stage breast cancer. J Clin Oncol. 2012; 30:936–942. PMID: 22331951.
Article20. Fink AK, Gurwitz J, Rakowski W, Guadagnoli E, Silliman RA. Patient beliefs and tamoxifen discontinuance in older women with estrogen receptor-positive breast cancer. J Clin Oncol. 2004; 22:3309–3315. PMID: 15310774.
Article21. Oberguggenberger A, Hubalek M, Sztankay M, Meraner V, Beer B, Oberacher H, et al. Is the toxicity of adjuvant aromatase inhibitor therapy underestimated? Complementary information from patient-reported outcomes (PROs). Breast Cancer Res Treat. 2011; 128:553–561. PMID: 21311968.
Article22. Ruhstaller T, von Moos R, Rufibach K, Ribi K, Glaus A, Spaeti B, et al. Breast cancer patients on endocrine therapy reveal more symptoms when self-reporting than in pivotal trials: an outcome research study. Oncology. 2009; 76:142–148. PMID: 19158446.
Article23. Rosenberg SM, Stanton AL, Petrie KJ, Partridge AH. Symptoms and symptom attribution among women on endocrine therapy for breast cancer. Oncologist. 2015; 20:598–604. PMID: 25933930.
Article24. Stearns V, Ullmer L, López JF, Smith Y, Isaacs C, Hayes D. Hot flushes. Lancet. 2002; 360:1851–1861. PMID: 12480376.
Article25. Jager NG, Koornstra RH, Vincent AD, van Schaik RH, Huitema AD, Korse TM, et al. Hot flashes are not predictive for serum concentrations of tamoxifen and its metabolites. BMC Cancer. 2013; 13:612. PMID: 24373320.
Article26. Honda J, Kanematsu M, Nakagawa M, Takahashi M, Nagao T, Tangoku A, et al. Joint symptoms, aromatase inhibitor-related adverse reactions, are indirectly associated with decreased serum estradiol. Int J Surg Oncol. 2011; 2011:951260. PMID: 22312534.
Article27. Gaillard S, Stearns V. Aromatase inhibitor-associated bone and musculoskeletal effects: new evidence defining etiology and strategies for management. Breast Cancer Res. 2011; 13:205. PMID: 21457526.
Article28. Henry NL, Giles JT, Stearns V. Aromatase inhibitor-associated musculoskeletal symptoms: etiology and strategies for management. Oncology (Williston Park). 2008; 22:1401–1408. PMID: 19086600.29. Brett J, Fenlon D, Boulton M, Hulbert-Williams NJ, Walter FM, Donnelly P, et al. Factors associated with intentional and unintentional non-adherence to adjuvant endocrine therapy following breast cancer. Eur J Cancer Care (Engl). 2016; 11. 30. Epub. DOI: 10.1111/ecc.12601.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Pregnancy After Breast Cancer – Prognostic Safety and Pregnancy Outcomes According to Oestrogen Receptor Status: A Systematic Review
- Highlights of 10th St. Gallen Breast Cancer Conference: Systemic Adjuvant Treatment
- Stacking Ensemble Technique for Classifying Breast Cancer
- Treatment Outcomes of Weakly Positive Hormone Receptor Breast Cancer and Triple-Negative Breast Cancer
- Influence of Androgen Receptor Expression on the Survival Outcomes in Breast Cancer: A Meta-Analysis