Cancer Res Treat.
2018 Oct;50(4):1462-1466. 10.4143/crt.2017.473.
Delayed Terminal Ileal Perforation in a Relapsed/Refractory B-Cell Lymphoma Patient with Rapid Remission Following Chimeric Antigen Receptor T-Cell Therapy
- Affiliations
-
- 1Bone Marrow Transplantation Center, The First Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou, China. huanghe@zju.edu.cn
- 2Innovative Cellular Therapeutics Co., Ltd., Shanghai, China. wuzhao@sidansai.com
- 3PET/CT Center, The First Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou, China.
- 4Department of Hematology, Beijing Tiantan Hospital, Capital Medical University, Beijing, China.
- KMID: 2424817
- DOI: http://doi.org/10.4143/crt.2017.473
Abstract
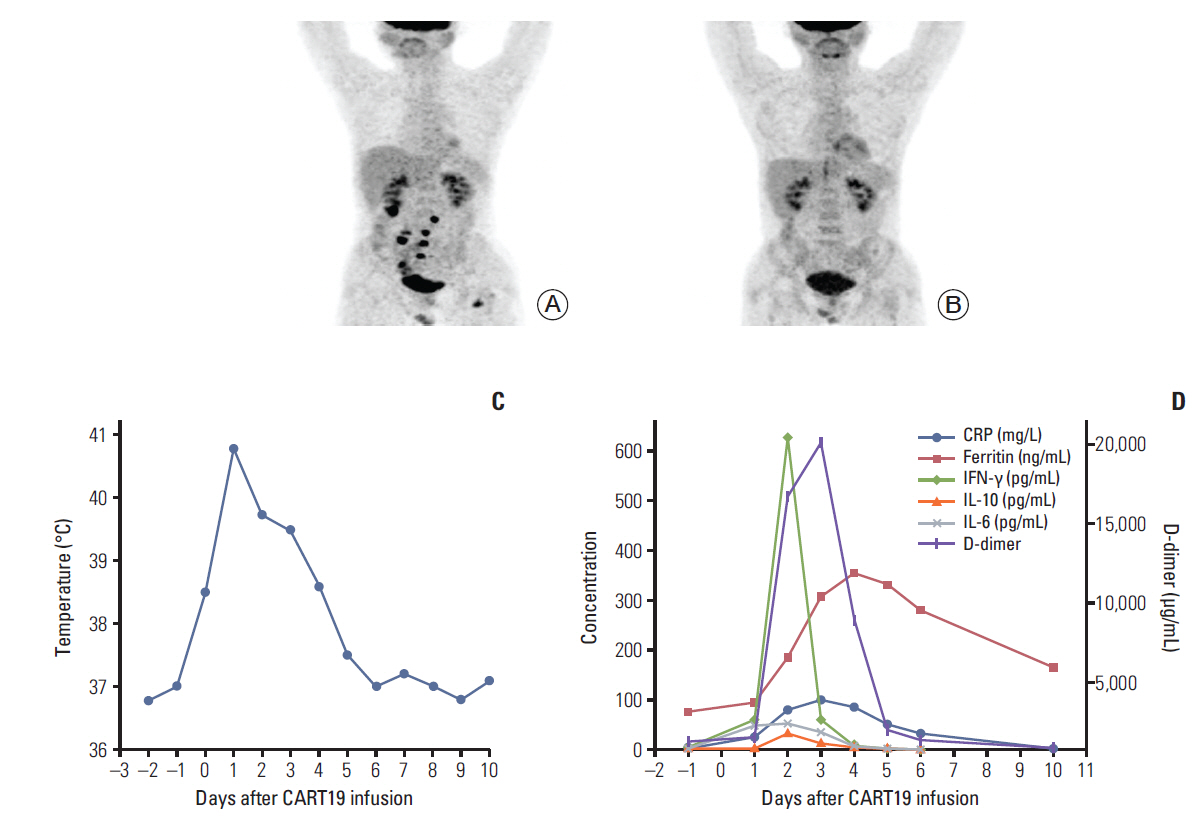
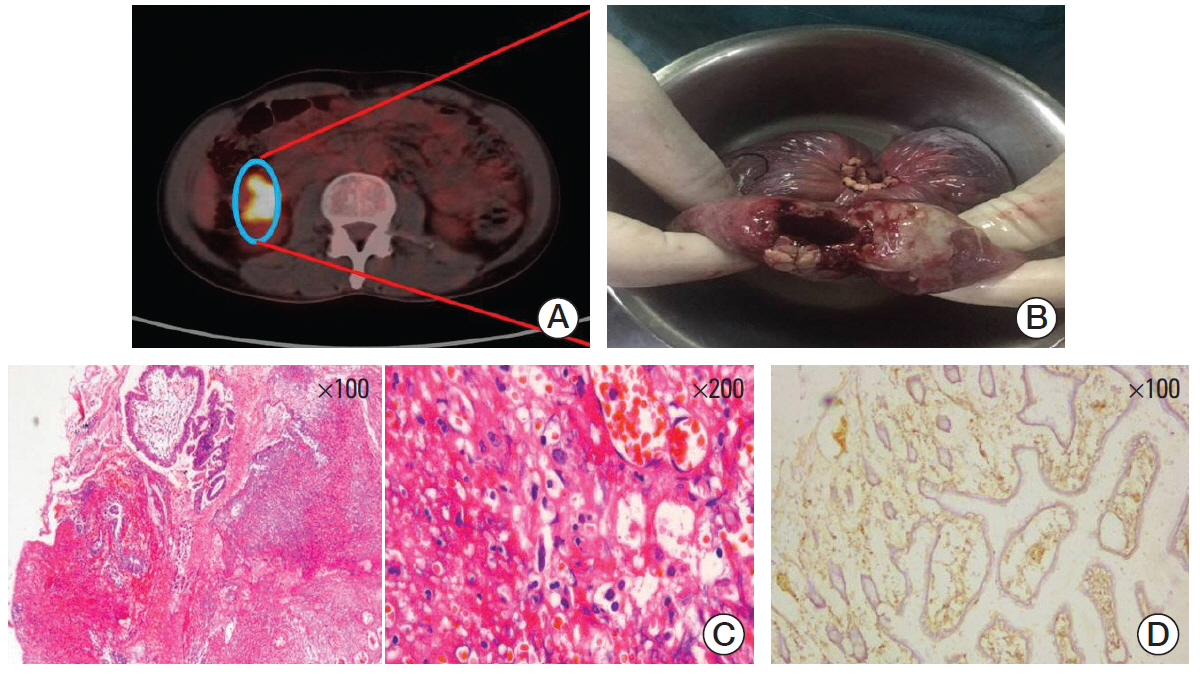
- Chimeric antigen receptor T-cell strategy targeting CD19 (CART19) has prominent anti-tumor effect for relapsed/refractory B-cell lymphomas. CART19-associated complications have been gradually recognized, however, late-onset complications have not been extensively studied. Herein, for the first time we report a diffuse large B-cell lymphoma patient with terminal ileum involvement obtained rapid remission and developed spontaneous terminal ileal perforation 38 days following CART19 infusion. The late-onset perforation reminds us that, for the safety of CART treatment, more cautions are warranted for the management of delayed GI complications.
Keyword
MeSH Terms
Figure
Reference
-
References
1. Kochenderfer JN, Somerville RP, Lu T, Shi V, Bot A, Rossi J, et al. Lymphoma remissions caused by anti-CD19 chimeric antigen receptor T cells are associated with high serum interleukin-15 levels. J Clin Oncol. 2017; 35:1803–13.
Article2. Turtle CJ, Hanafi LA, Berger C, Gooley TA, Cherian S, Hudecek M, et al. CD19 CAR-T cells of defined CD4+:CD8+ composition in adult B cell ALL patients. J Clin Invest. 2016; 126:2123–38.
Article3. Locke FL, Neelapu SS, Bartlett NL, Siddiqi T, Chavez JC, Hosing CM, et al. Phase 1 results of ZUMA-1: a multicenter study of KTE-C19 anti-CD19 CAR T cell therapy in refractory aggressive lymphoma. Mol Ther. 2017; 25:285–95.
Article4. Brudno JN, Kochenderfer JN. Toxicities of chimeric antigen receptor T cells: recognition and management. Blood. 2016; 127:3321–30.
Article5. Hu Y, Sun J, Wu Z, Yu J, Cui Q, Pu C, et al. Predominant cerebral cytokine release syndrome in CD19-directed chimeric antigen receptor-modified T cell therapy. J Hematol Oncol. 2016; 9:70.
Article6. Hu Y, Wu Z, Luo Y, Shi J, Yu J, Pu C, et al. Potent antileukemia activities of chimeric antigen receptor-modified T cells against CD19 in Chinese patients with relapsed/refractory acute lymphocytic leukemia. Clin Cancer Res. 2017; 23:3297–306.
Article7. Vaidya R, Habermann TM, Donohue JH, Ristow KM, Maurer MJ, Macon WR, et al. Bowel perforation in intestinal lymphoma: incidence and clinical features. Ann Oncol. 2013; 24:2439–43.
Article8. Vaidya R, Witzig TE. Incidence of bowel perforation in gastrointestinal lymphomas by location and histology. Ann Oncol. 2014; 25:1249–50.
Article9. Ara C, Coban S, Kayaalp C, Yilmaz S, Kirimlioglu V. Spontaneous intestinal perforation due to non-Hodgkin's lymphoma: evaluation of eight cases. Dig Dis Sci. 2007; 52:1752–6.
Article10. Wada M, Onda M, Tokunaga A, Kiyama T, Yoshiyuki T, Matsukura N, et al. Spontaneous gastrointestinal perforation in patients with lymphoma receiving chemotherapy and steroids: report of three cases. Nihon Ika Daigaku Zasshi. 1999; 66:37–40.
Article11. Zhai L, Zhao Y, Lin L, Tian Y, Chen X, Huang H, et al. Non-Hodgkin's lymphoma involving the ileocecal region: a singleinstitution analysis of 46 cases in a Chinese population. J Clin Gastroenterol. 2012; 46:509–14.12. Maisey N, Norman A, Prior Y, Cunningham D. Chemotherapy for primary gastric lymphoma: does in-patient observation prevent complications? Clin Oncol (R Coll Radiol). 2004; 16:48–52.
Article13. Porter DL, Hwang WT, Frey NV, Lacey SF, Shaw PA, Loren AW, et al. Chimeric antigen receptor T cells persist and induce sustained remissions in relapsed refractory chronic lymphocytic leukemia. Sci Transl Med. 2015; 7:303ra139.
Article14. Kebriaei P, Singh H, Huls MH, Figliola MJ, Bassett R, Olivares S, et al. Phase I trials using Sleeping Beauty to generate CD19-specific CAR T cells. J Clin Invest. 2016; 126:3363–76.
Article15. Wang Y, Zhang WY, Han QW, Liu Y, Dai HR, Guo YL, et al. Effective response and delayed toxicities of refractory advanced diffuse large B-cell lymphoma treated by CD20-directed chimeric antigen receptor-modified T cells. Clin Immunol. 2014; 155:160–75.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Chimeric Antigen Receptor T-Cell Therapy for Diffuse Large B-Cell Lymphoma
- Current Challenges in Chimeric Antigen Receptor T-cell Therapy in Patients With B-cell Lymphoid Malignancies
- Management of adverse events in young adults and children with acute B-cell lymphoblastic leukemia receiving anti-CD19 chimeric antigen receptor (CAR) T-cell therapy
- Chimeric Antigen Receptor-T Cell Therapy
- Engineered T Cell Receptor for Cancer Immunotherapy