J Clin Neurol.
2018 Oct;14(4):537-541. 10.3988/jcn.2018.14.4.537.
Characteristics of South Korean Patients with Hereditary Transthyretin Amyloidosis
- Affiliations
-
- 1Department of Neurology, Konkuk University Medical Center, Konkuk University School of Medicine, Seoul, Korea. serein@kuh.ac.kr
- 2Department of Neurology, Soonchunhyang University Cheonan Hospital, Soonchunhyang University College of Medicine, Cheonan, Korea.
- 3Department of Neurology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. juhongm@gmail.com
- 4Department of Neurology, Gangnam Severance Hospital, Yonsei University College of Medicine, Seoul, Korea.
- 5Department of Neurology, Yonsei University College of Medicine, Seoul, Korea.
- 6Sunwoo and Cho Neurology Clinic, Seoul, Korea.
- 7Department of Neurology, Pusan National University Yangsan Hospital, Pusan National University College of Medicine, Yangsan, Korea.
- 8Department of Neurology, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea.
- 9Division of Cardiology, Department of Medicine, Cardiac and Vascular Center, Samsung Medical Center, Seoul, Korea.
- 10Department of Neurology, Dongguk University Ilsan Hospital, Goyang, Korea.
- KMID: 2424183
- DOI: http://doi.org/10.3988/jcn.2018.14.4.537
Abstract
- BACKGROUND AND PURPOSE
This retrospective cross-sectional study included 18 patients from unrelated families harboring mutations of the transthyretin gene (TTR), and analyzed their characteristics and geographical distribution in South Korea.
METHODS
The included patients had a diagnosis of systemic amyloidosis, clinical symptoms, such as amyloid neuropathy or cardiomyopathy, and confirmation of a TTR gene mutation using genetic analysis recorded between April 1995 and November 2014.
RESULTS
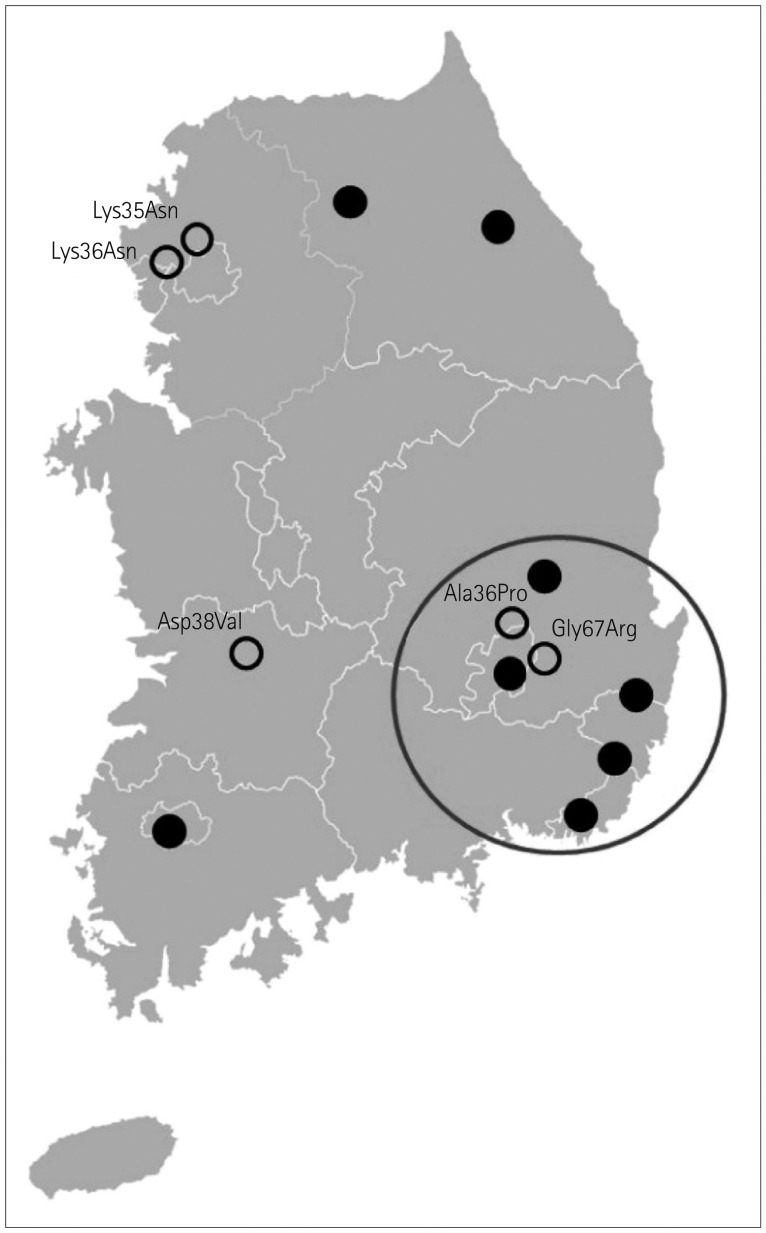
The mean age at disease onset was 49.6 years, and the mean disease duration from symptom onset to diagnosis was 3.67 years. Fifteen of the 18 patients were classified as mixed phenotype, 2 as the neurological phenotype, and only 1 patient as the cardiac phenotype. The most-common mutation pattern in South Korea was Asp38Ala, which was detected in eight patients. Thirteen patients reported their family hometowns, and five of the eight harboring the Asp38Ala mutation were from the Gyeongsang province in southeast Korea. The other eight patients exhibited a widespread geographical distribution. A particularly noteworthy finding was that the valine at position 30 (Val30Met) mutation, which was previously reported as the most-common TTR mutation worldwide and also the most common in the Japanese population, was not detected in the present South Korean patients.
CONCLUSIONS
South Korean patients with hereditary TTR amyloidosis exhibited heterogeneous TTR genotypes and clinical phenotypes. The findings of this study suggest that the distribution of TTR amyloidosis in South Korea is due to de novo mutations and/or related to the other countries in East Asia.
Keyword
MeSH Terms
Figure
Reference
-
1. Saraiva MJ. Transthyretin mutations in hyperthyroxinemia and amyloid diseases. Hum Mutat. 2001; 17:493–503. PMID: 11385707.
Article2. Ikeda S, Nakazato M, Ando Y, Sobue G. Familial transthyretin-type amyloid polyneuropathy in Japan: clinical and genetic heterogeneity. Neurology. 2002; 58:1001–1007. PMID: 11940682.
Article3. Sousa A, Coelho T, Barros J, Sequeiros J. Genetic epidemiology of familial amyloidotic polyneuropathy (FAP)-type I in Póvoa do Varzim and Vila do Conde (north of Portugal). Am J Med Genet. 1995; 60:512–521. PMID: 8825887.
Article4. Holmgren G, Holmberg E, Lindström A, Lindström E, Nordenson I, Sandgren O, et al. Diagnosis of familial amyloidotic polyneuropathy in Sweden by RFLP analysis. Clin Genet. 1988; 33:176–180. PMID: 2896079.
Article5. Coelho T, Maurer MS, Suhr OB. THAOS - The Transthyretin Amyloidosis Outcomes Survey: initial report on clinical manifestations in patients with hereditary and wild-type transthyretin amyloidosis. Curr Med Res Opin. 2013; 29:63–76. PMID: 23193944.
Article6. Maurer MS, Hanna M, Grogan M, Dispenzieri A, Witteles R, Drachman B, et al. Genotype and phenotype of transthyretin cardiac amyloidosis: THAOS (Transthyretin Amyloid Outcome Survey). J Am Coll Cardiol. 2016; 68:161–172. PMID: 27386769.7. Kim HS, Kim SM, Kang SW, Jung SC, Lee KS, Kim TS, et al. An aggressive form of familial amyloid polyneuropathy caused by a Glu54Gly mutation in the transthyretin gene. Eur J Neurol. 2005; 12:657–659. PMID: 16053476.
Article8. Kim YJ, Lee J, Park J, Kim S, Jung I, Lim HJ, et al. Familial amyloidotic polyneuropathy with transthyretin gene mutation. J Korean Neurol Assoc. 2011; 29:220–223.9. Ryu JK, Baik HW, Bae JS, Hwang TJ, Paik SY, Yu HJ, et al. Familial amyloid polyneuropathy in Korea: the first case report with a proven ATTR Lys35Asn gene. Amyloid. 2005; 12:62–64. PMID: 16076613.
Article10. Jang MA, Lee GY, Kim K, Kim SJ, Kim JS, Lee SY, et al. Asp58Ala is the predominant mutation of the TTR gene in Korean patients with hereditary transthyretin-related amyloidosis. Ann Hum Genet. 2015; 79:99–107. PMID: 25644864.11. Ando Y, Coelho T, Berk JL, Cruz MW, Ericzon BG, Ikeda S, et al. Guideline of transthyretin-related hereditary amyloidosis for clinicians. Orphanet J Rare Dis. 2013; 8:31. PMID: 23425518.
Article12. Nuvolone M, Obici L, Merlini G. Transthyretin-associated familial amyloid polyneuropathy-current and emerging therapies. US Neurology. 2012; 8:24–32.
Article13. Hsieh ST. Amyloid neuropathy with transthyretin mutations: overview and unique Ala97Ser in Taiwan. Acta Neurol Taiwan. 2011; 20:155–160. PMID: 21739396.14. Yazak M, Take YI, Katoh M, Ikeda SI. Postmortem findings in two familial amyloidosis patients with transthyretin variant Asp38Ala. Amyloid. 2000; 7:270–277. PMID: 11132096.
Article15. Reinés JB, Vera TR, Martín MU, Serra HA, Campins MM, Millán JM, et al. Epidemiology of transthyretin-associated familial amyloid polyneuropathy in the Majorcan area: Son Llàtzer Hospital descriptive study. Orphanet J Rare Dis. 2014; 9:29. PMID: 24572009.
Article16. Dardiotis E, Koutsou P, Papanicolaou EZ, Vonta I, Kladi A, Vassilopoulos D, et al. Epidemiological, clinical and genetic study of familial amyloidotic polyneuropathy in Cyprus. Amyloid. 2009; 16:32–37. PMID: 19291512.
Article17. Kato-Motozaki Y, Ono K, Shima K, Morinaga A, Machiya T, Nozaki I, et al. Epidemiology of familial amyloid polyneuropathy in Japan: identification of a novel endemic focus. J Neurol Sci. 2008; 270:133–140. PMID: 18410945.
Article18. Sekijima Y, Yoshida K, Tokuda T, Ikeda S. Familial transthyretin amyloidosis. In : Adam MP, Ardinger HH, Pagon RA, Wallace SE, Bean LJH, Stephens K, editors. GeneReviews® [Internet]. Seattle, WA: University of Washington, Seattle;2012. p. 1–32.19. Mascalchi M, Salvi F, Pirini MG, D'Errico A, Ferlini A, Lolli F, et al. Transthyretin amyloidosis and superficial siderosis of the CNS. Neurology. 1999; 53:1498–1503. PMID: 10534258.
Article20. Jones LA, Skare JC, Harding JA, Cohen AS, Milunsky A, Skinner M. Proline at position 36: a new transthyretin mutation associated with familial amyloidotic polyneuropathy. Am J Hum Genet. 1991; 48:979–982. PMID: 1850191.21. Long D, Zeng J, Wu LQ, Tang LS, Wang HL, Wang H. Vitreous amyloidosis in two large mainland Chinese kindreds resulting from transthyretin variant Lys35Thr and Leu55Arg. Ophthalmic Genet. 2012; 33:28–33. PMID: 21843040.
Article22. Liu G, Ni W, Wang H, Li H, Zhang Y, Wang N, et al. Clinical features of familial amyloid polyneuropathy carrying transthyretin mutations in four Chinese kindreds. J Peripher Nerv Syst. 2017; 22:19–26. PMID: 27859927.
Article23. Jacobson DR, Pastore RD, Yaghoubian R, Kane I, Gallo G, Buck FS, et al. Variant-sequence transthyretin (isoleucine 122) in late-onset cardiac amyloidosis in black Americans. N Engl J Med. 1997; 336:466–473. PMID: 9017939.
Article24. Swiecicki PL, Zhen DB, Mauermann ML, Kyle RA, Zeldenrust SR, Grogan M, et al. Hereditary ATTR amyloidosis: a single-institution experience with 266 patients. Amyloid. 2015; 22:123–131. PMID: 26017327.
Article25. Takahashi K, Yi S, Kimura Y, Araki S. Familial amyloidotic polyneuropathy type 1 in Kumamoto, Japan: a clinicopathologic, histochemical, immunohistochemical, and ultrastructural study. Hum Pathol. 1991; 22:519–527. PMID: 1864584.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Hereditary Transthyretin Amyloidosis Misdiagnosed as Demyelinating Neuropathy: A Report of Three Cases
- A rare pathogenic variant identified in a heart transplant recipient with hereditary transthyretin amyloidosis: a case report
- Familial Amyloidotic Polyneuropathy With Transthyretin Gene Mutation
- Transthyretin Cardiac Amyloidosis: A Case Report
- Tafamidis for Cardiac Transthyretin Amyloidosis