J Clin Neurol.
2018 Oct;14(4):444-453. 10.3988/jcn.2018.14.4.444.
Neuromonitoring for Spinal Cord Stimulation Lead Placement Under General Anesthesia
- Affiliations
-
- 1Department of Anesthesiology, Rush University Medical Center, Chicago, IL, USA. Jay_l_shils@rush.edu
- 2Department of Neurosurgery, Beth Israel Deaconess Medical Center, Boston, MA, USA.
- 3Department of Neurosurgery, Harvard Medical School, Boston, MA, USA.
- KMID: 2424171
- DOI: http://doi.org/10.3988/jcn.2018.14.4.444
Abstract
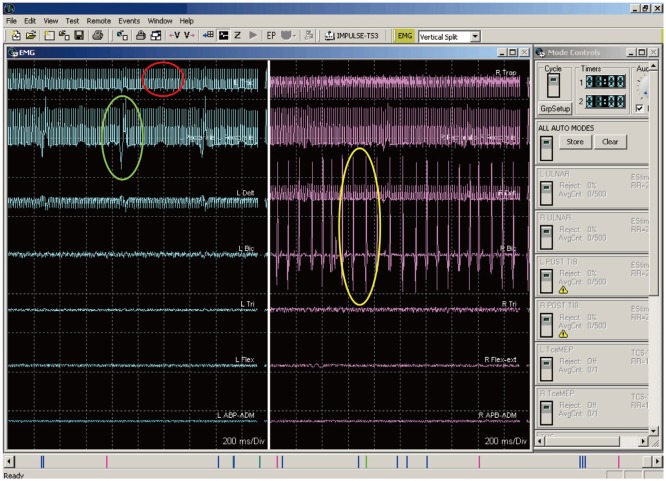
- Spinal cord stimulation (SCS) is a common therapeutic technique for treating medically refractory neuropathic back and other limb pain syndromes. SCS has historically been performed using a sedative anesthetic technique where the patient is awakened at various times during a surgical procedure to evaluate the location of the stimulator lead. This technique has potential complications, and thus other methods that allow the use of a general anesthetic have been developed. There are two primary methods for placing leads under general anesthesia, based on 1) compound muscle action potentials and 2) collisions between somatosensory evoked potentials. Both techniques are discussed, and the literature on SCS lead placement under general anesthesia using intraoperative neurophysiological mapping is comprehensively reviewed.
Keyword
MeSH Terms
Figure
Reference
-
1. Zhang K, Bhatia S, Oh M, Whiting D. Epidural anesthesia for placement of spinal cord stimulators with paddle-type electrodes. Stereotact Funct Neurosurg. 2009; 87:292–296. PMID: 19590261.
Article2. Vangeneugden J. Implantation of surgical electrodes for spinal cord stimulation: classical midline laminotomy technique versus minimal invasive unilateral technique combined with spinal anaesthesia. Acta Neurochir Suppl. 2007; 97:111–114.
Article3. Skipsey IG, Colvin JR, Mackenzie N, Kenny GN. Sedation with propofol during surgery under local blockade. Assessment of a target-controlled infusion system. Anaesthesia. 1993; 48:210–213. PMID: 8460797.4. Mingus ML, Monk TG, Gold MI, Jenkins W, Roland C. Remifentanil versus propofol as adjuncts to regional anesthesia. Remifentanil 3010 Study Group. J Clin Anesth. 1998; 10:46–53. PMID: 9526938.5. Holsheimer J, Barolat G, Struijk JJ, He J. Significance of the spinal cord position in spinal cord stimulation. Acta Neurochir Suppl. 1995; 64:119–124. PMID: 8748598.
Article6. Holsheimef J, Barolat G. Spinal geometry and paresthesia coverage in spinal cord stimulation. Neuromodulation. 1998; 1:129–136. PMID: 22150980.
Article7. Bhananker SM, Posner KL, Cheney FW, Caplan RA, Lee LA, Domino KB. Injury and liability associated with monitored anesthesia care: a closed claims analysis. Anesthesiology. 2006; 104:228–234. PMID: 16436839.8. Shils JL, Arle JE. Intraoperative neurophysiologic methods for spinal cord stimulator placement under general anesthesia. Neuromodulation. 2012; 15:560–571. PMID: 22672099.
Article9. Mammis A, Mogilner AY. The use of intraoperative electrophysiology for the placement of spinal cord stimulator paddle leads under general anesthesia. Neurosurgery. 2012; 70:230–236. PMID: 21869720.
Article10. Falowski SM, Celii A, Sestokas AK, Schwartz DM, Matsumoto C, Sharan A. Awake vs. asleep placement of spinal cord stimulators: a cohort analysis of complications associated with placement. Neuromodulation. 2011; 14:130–134. PMID: 21992199.
Article11. Schoen N, Chieng LO, Madhavan K, Jermakowicz WJ, Vanni S. The use of intraoperative electromyogram during spinal cord stimulator placement surgery: a case series. World Neurosurg. 2017; 100:74–84. PMID: 28034811.
Article12. Tamkus AA, Scott AF, Khan FR. Neurophysiological monitoring during spinal cord stimulator placement surgery. Neuromodulation. 2015; 18:460–464. PMID: 25677059.
Article13. Roth SG, Lange S, Haller J, De La Cruz P, Kumar V, Wilock M, et al. A prospective study of the intra- and postoperative efficacy of intraoperative neuromonitoring in spinal cord stimulation. Stereotact Funct Neurosurg. 2015; 93:348–354. PMID: 26444517.
Article14. Air EL, Toczyl GR, Mandybur GT. Electrophysiologic monitoring for placement of laminectomy leads for spinal cord stimulation under general anesthesia. Neuromodulation. 2012; 15:573–579. PMID: 23205816.
Article15. Collison C, Prusik J, Paniccioli S, Briotte M, Grey R, Feustel P, et al. Prospective study of the use of intraoperative neuromonitoring in determining post-operative energy requirements and physiologic midline in spinal cord stimulation. Neuromodulation. 2017; 20:575–581. PMID: 28370852.16. Davidoff RA. Handbook of the Spinal Cord: Anatomy and Physiology (Vols 2 and 3). New York, NY: Dekker Publishing;1984. p. 318.17. Balzer JR, Tomycz ND, Crammond DJ, Habeych M, Thirumala PD, Urgo L, et al. Localization of cervical and cervicomedullary stimulation leads for pain treatment using median nerve somatosensory evoked potential collision testing. J Neurosurg. 2011; 114:200–205. PMID: 20509731.
Article18. Hunter JP, Ashby P. Segmental effects of epidural spinal cord stimulation in humans. J Physiol. 1994; 474:407–419. PMID: 8014902.
Article19. Schirmer CM, Shils JL, Arle JE, Cosgrove GR, Dempsey PK, Tarlov E, et al. Heuristic map of myotomal innervation in humans using direct intraoperative nerve root stimulation. J Neurosurg Spine. 2011; 15:64–70. PMID: 21476796.
Article20. Arle JE, Mei LZ, Carlson KE, Shils JL. Theoretical effect of DBS on axonal fibers of passage: firing rates, entropy, and information content. Stereotact Funct Neurosurg. 2018; 96:1–12. PMID: 29393267.
Article21. Yingling CD, Hosobuchi Y. Use of antidromic evoked potentials in placement of dorsal cord disc electrodes. Appl Neurophysiol. 1986; 49:36–41. PMID: 3490219.
Article22. Deletis V, Bueno De. Interventional neurophysiological mapping during spinal cord procedures. Stereotact Funct Neurosurg. 2001; 77:25–28. PMID: 12378052.
Article23. Falowski SM, Sharan A, McInerney J, Jacobs D, Venkatesan L, Agnesi F. Nonawake vs Awake Placement of Spinal Cord Stimulators: A Prospective, Multicenter Study Comparing Safety and Efficacy. Neurosurgery. 2018; 3. 14. [Epub] available from: . DOI: 10.1093/neuros/nyy062.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Spinal anesthesia for implantation of a spinal cord stimulator in a patient with failed back surgery syndrome: A case report
- Epidural anesthesia for permanent spinal cord stimulation with a cylindrical type lead: a case series
- Cervical Spinal Cord Stimulation Using an 8 Electrode Lead in a Patient with Complex Regional Pain Syndrome Type I: A case report
- Spontaneous Lead Breakage in Implanted Spinal Cord Stimulation Systems
- An Analysis of Paresthesia Areas Evoked by Spinal Cord Stimulation in Relation to the Position of Electrode Tip