J Vet Sci.
2018 Sep;19(5):699-707. 10.4142/jvs.2018.19.5.699.
Cranial cruciate ligament structure in relation to the tibial plateau slope and intercondylar notch width in dogs
- Affiliations
-
- 1Department of Anatomy, Histology and Embryology, Faculty of Veterinary Medicine, University of Veterinary and Pharmaceutical Sciences Brno, 612 42 Brno, Czech Republic. kyllarm@vfu.cz
- 2Companion Care, Broadstairs CT10 2RQ, United Kingdom.
- KMID: 2420940
- DOI: http://doi.org/10.4142/jvs.2018.19.5.699
Abstract
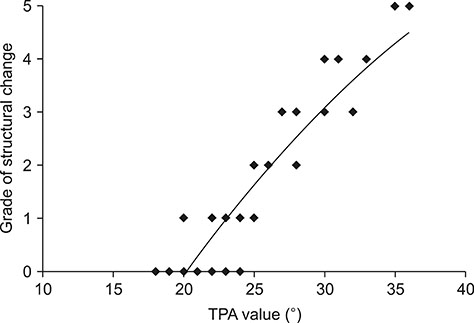
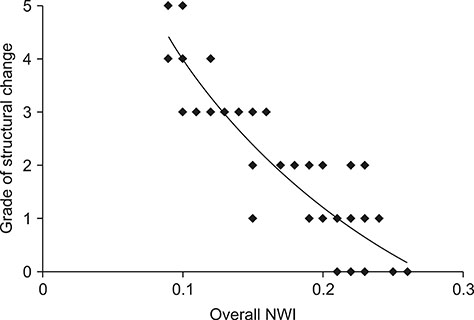
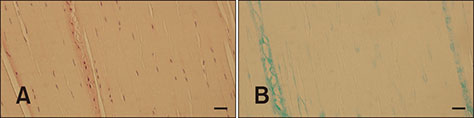
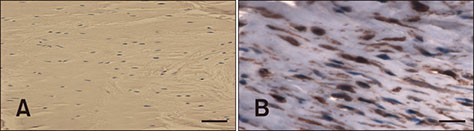
- Cranial cruciate ligament (CCL) rupture is one of the most common orthopedic conditions in dogs. The pathogenesis of CCL rupture is not fully described and remains to be elucidated fully. Several hypotheses have been proposed to explain the etiology of these changes. The objective of this study was to investigate structural changes in the CCL in relation to the tibial plateau angle (TPA) and the intercondylar notch (ICN) width in dogs. Fifty-five skeletally mature dogs were included in this study. ICN width and TPA measurements were obtained from intact CCL stifles. Samples of the CCL, caudal cruciate ligament (CaCL), and femoral head ligament (FHL) were harvested and stained for routine histological and immunohistochemical analysis. Microscopic changes in the ligaments were observed and were found to correlate with the TPA and ICN width values. The degree of structural changes within the CCL was observed to correlate with an increasing TPA and a narrowing ICN width. Changes in the CCL are likely to be caused by excessive forces acting through the ligament in stifles with a high TPA. Chondroid metaplasia of the CCL is an adaptation to abnormal mechanics within the stifle joint caused by altered bone morphology.
Keyword
MeSH Terms
Figure
Reference
-
1. Anderst WJ, Tashman S. The association between velocity of the center of closest proximity on subchondral bones and osteoarthritis progression. J Orthop Res. 2009; 27:71–77.
Article2. Barrett JG, Hao Z, Graf BK, Kaplan LD, Heiner JP, Muir P. Inflammatory changes in ruptured canine cranial and human anterior cruciate ligaments. Am J Vet Res. 2005; 66:2073–2080.
Article3. Bossard MJ, Tomaszek TA, Thompson SK, Amegadzie BY, Hanning CR, Jones C, Kurdyla JT, McNulty DE, Drake FH, Gowen M, Levy MA. Proteolytic activity of human osteoclast cathepsin K. Expression, purification, activation, and substrate identification. J Biol Chem. 1996; 271:12517–12524.4. Comerford EJ, Innes JF, Tarlton JF, Bailey AJ. Investigation of the composition, turnover, and thermal properties of ruptured cranial cruciate ligaments of dogs. Am J Vet Res. 2004; 65:1136–1141.
Article5. Comerford EJ, Tarlton JF, Avery NC, Bailey AJ, Innes JF. Distal femoral intercondylar notch dimensions and their relationship to composition and metabolism of the canine anterior cruciate ligament. Osteoarthritis Cartilage. 2006; 14:273–278.
Article6. Comerford EJ, Tarlton JF, Innes JF, Johnson KA, Amis AA, Bailey AJ. Metabolism and composition of the canine anterior cruciate ligament relate to differences in knee joint mechanics and predisposition to ligament rupture. J Orthop Res. 2005; 23:61–66.
Article7. Comerford EJ, Tarlton JF, Wales A, Bailey AJ, Innes JF. Ultrastructural differences in cranial cruciate ligaments from dogs of two breeds with a differing predisposition to ligament degeneration and rupture. J Comp Pathol. 2006; 134:8–16.
Article8. Cook JL. Cranial cruciate ligament disease in dogs: biology versus biomechanics. Vet Surg. 2010; 39:270–277.
Article9. de Rooster H, de Bruin T, van Bree H. Morphologic and functional features of the canine cruciate ligaments. Vet Surg. 2006; 35:769–780.10. Dennler R, Kipfer NM, Tepic S, Hassig M, Montavon PM. Inclination of the patellar ligament in relation to flexion angle in stifle joints of dogs without degenerative joint disease. Am J Vet Res. 2006; 67:1849–1854.
Article11. Duval JM, Budsberg SC, Flo GL, Sammarco JL. Breed, sex, and body weight as risk factors for rupture of the cranial cruciate ligament in young dogs. J Am Vet Med Assoc. 1999; 215:811–814.12. Fitch RB, Montgomery RD, Milton JL, Garrett PD, Kincaid SA, Wright JC, Terry GC. The intercondylar fossa of the normal canine stifle an anatomic and radiographic study. Vet Surg. 1995; 24:148–155.
Article13. Fujita Y, Hara Y, Nezu Y, Schulz KS, Tagawa M. Proinflammatory cytokine activities, matrix metalloproteinase-3 activity, and sulfated glycosaminoglycan content in synovial fluid of dogs with naturally acquired cranial cruciate ligament rupture. Vet Surg. 2006; 35:369–376.
Article14. Galloway RH, Lester SJ. Histopathological evaluation of canine stifle joint synovial membrane collected at the time of repair of cranial cruciate ligament rupture. J Am Anim Hosp Assoc. 1995; 31:289–294.
Article15. Gambardella PC, Wallace LJ, Cassidy F. Lateral suture technique for management of anterior cruciate ligament rupture in dogs: a retrospective study. J Am Anim Hosp Assoc. 1981; 17:33–38.16. Guerrero TG, Geyer H, Hässig M, Montavon PM. Effect of conformation of the distal portion of the femur and proximal portion of the tibia on the pathogenesis of cranial cruciate ligament disease in dogs. Am J Vet Res. 2007; 68:1332–1337.
Article17. Halleen JM, Räisänen S, Salo JJ, Reddy SV, Roodman GD, Hentunen TA, Lehenkari PP, Kaija H, Vihko P, Väänänen HK. Intracellular fragmentation of bone resorption products by reactive oxygen species generated by osteoclastic tartrate-resistant acid phosphatase. J Biol Chem. 1999; 274:22907–22910.
Article18. Hayashi K, Kim SY, Lansdowne JL, Kapatkin A, Déjardin LM. Evaluation of a collagenase generated osteoarthritis biomarker in naturally occurring canine cruciate disease. Vet Surg. 2009; 38:117–121.
Article19. Hayashi K, Manley PA, Muir P. Cranial cruciate ligament pathophysiology in dogs with cruciate disease: a review. J Am Anim Hosp Assoc. 2004; 40:385–390.
Article20. Innes JF, Bacon D, Lynch C, Pollard A. Long-term outcome of surgery for dogs with cranial cruciate ligament deficiency. Vet Rec. 2000; 147:325–328.
Article21. Kafienah W, Brömme D, Buttle DJ, Croucher LJ, Hollander AP. Human cathepsin K cleaves native type I and II collagens at the N-terminal end of the triple helix. Biochem J. 1998; 331:727–732.
Article22. Kim SE, Pozzi A, Kowaleski MP, Lewis DD. Tibial osteotomies for cranial cruciate ligament insufficiency in dogs. Vet Surg. 2008; 37:111–125.
Article23. Kipfer NM, Tepic S, Damur DM, Guerrero T, Hässig M, Montavon PM. Effect of tibial tuberosity advancement on femorotibial shear in cranial cruciate-deficient stifles. An in vitro study. Vet Comp Orthop Traumatol. 2008; 21:385–390.
Article24. Kirkby KA, Lewis DD. Canine hip dysplasia: reviewing the evidence for nonsurgical management. Vet Surg. 2012; 41:2–9.
Article25. Kowaleski MP, Apelt D, Mattoon JS, Litsky AS. The effect of tibial plateau leveling osteotomy position on cranial tibial subluxation: an in vitro study. Vet Surg. 2005; 34:332–336.
Article26. Krayer M, Rytz U, Oevermann A, Doherr MG, Forterre F, Zurbriggen A, Spreng DE. Apoptosis of ligamentous cells of the cranial cruciate ligament from stable stifle joints of dogs with partial cranial cruciate ligament rupture. Am J Vet Res. 2008; 69:625–630.
Article27. Leunig M, Beck M, Stauffer E, Hertel R, Ganz R. Free nerve endings in the ligamentum capitis femoris. Acta Orthop Scand. 2000; 71:452–454.
Article28. Morris E, Lipowitz AJ. Comparison of tibial plateau angles in dogs with and without cranial cruciate ligament injuries. J Am Vet Med Assoc. 2001; 218:363–366.
Article29. Muir P, Hayashi K, Manley PA, Colopy SA, Hao Z. Evaluation of tartrate-resistant acid phosphatase and cathepsin K in ruptured cranial cruciate ligaments in dogs. Am J Vet Res. 2002; 63:1279–1284.
Article30. Muir P, Schamberger GM, Manley PA, Hao Z. Localization of cathepsin K and tartrate-resistant acid phosphatase in synovium and cranial cruciate ligament in dogs with cruciate disease. Vet Surg. 2005; 34:239–246.
Article31. Pozzi A, Kowaleski MP, Apelt D, Meadows C, Andrews CM, Johnson KA. Effect of medial meniscal release on tibial translation after tibial plateau leveling osteotomy. Vet Surg. 2006; 35:486–494.
Article32. Reif U, Hulse DA, Hauptman JG. Effect of tibial plateau leveling on stability of the canine cranial cruciate-deficient stifle joint: an in vitro study. Vet Surg. 2002; 31:147–154.
Article33. Riitano MC, Pfister H, Engelhardt P, Neumann U, Reist M, Zurbriggen A, Stoffel M, Peel J, Jungi T, Schawalder P, Spreng DE. Effects of stimulus with proinflammatory mediators on nitric oxide production and matrix metalloproteinase activity in explants of cranial cruciate ligaments obtained from dogs. Am J Vet Res. 2002; 63:1423–1428.
Article34. Shahar R, Milgram J. Biomechanics of tibial plateau leveling of the canine cruciate-deficient stifle joint: a theoretical model. Vet Surg. 2006; 35:144–149.
Article35. Trumble TN, Billinghurst RC, McIlwraith CW. Correlation of prostaglandin E2 concentrations in synovial fluid with ground reaction forces and clinical variables for pain or inflammation in dogs with osteoarthritis induced by transection of the cranial cruciate ligament. Am J Vet Res. 2004; 65:1269–1275.
Article36. Vasseur PB, Pool RR, Arnoczky SP, Lau RE. Correlative biomechanical and histologic study of the cranial cruciate ligament in dogs. Am J Vet Res. 1985; 46:1842–1854.37. Warzee CC, Dejardin LM, Arnoczky SP, Perry RL. Effect of tibial plateau leveling on cranial and caudal tibial thrusts in canine cranial cruciate-deficient stifles: an in vitro experimental study. Vet Surg. 2001; 30:278–286.
Article38. Whitehair JG, Vasseur PB, Willits NH. Epidemiology of cranial cruciate ligament rupture in dogs. J Am Vet Med Assoc. 1993; 203:1016–1019.39. Wingfield C, Amis AA, Stead AC, Law HT. Comparison of the biomechanical properties of rottweiler and racing greyhound cranial cruciate ligaments. J Small Anim Pract. 2000; 41:303–307.
Article40. Zachos TA, Arnoczky SP, Lavagnino M, Tashman S. The effect of cranial cruciate ligament insufficiency on caudal cruciate ligament morphology: An experimental study in dogs. Vet Surg. 2002; 31:596–603.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Measurement of the Morphologic Changes of Intercondylar Notch by Magnetic Resonance Imaging
- Clinical Relevance of the Femoral Intercondylar Notch Width and the Posterior Tibial Slope for ACL Rupture
- Correlation of Notch Configuration between Subjects with and Subjects without Anterior Cruciate Ligament Injury
- MRI Measurement of the Intercondylar Notch and Correlation to Anterior Cruciate Ligament Injuries
- Do the Clinical and Radiological Features of Knees with Mucoid Degeneration of the Anterior Cruciate Ligament Differ According to Knee Osteoarthritis Status?