Korean J Radiol.
2018 Oct;19(5):978-991. 10.3348/kjr.2018.19.5.978.
Breast Lesions in Children and Adolescents: Diagnosis and Management
- Affiliations
-
- 1Department of Radiology, Soonchunhyang University Seoul Hospital, Seoul 04401, Korea. ywchang@schmc.ac.kr
- KMID: 2418562
- DOI: http://doi.org/10.3348/kjr.2018.19.5.978
Abstract
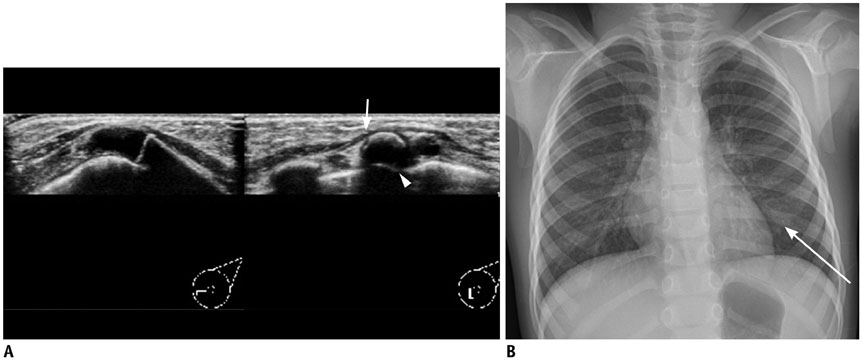
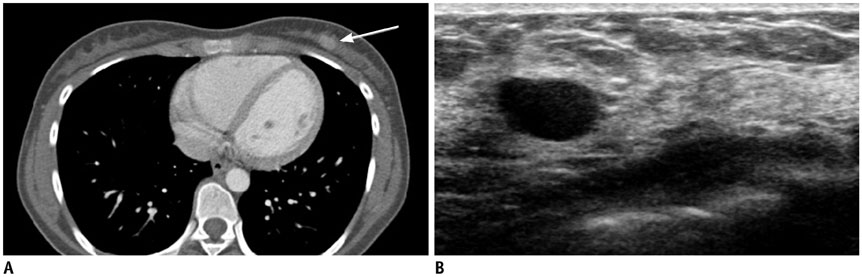
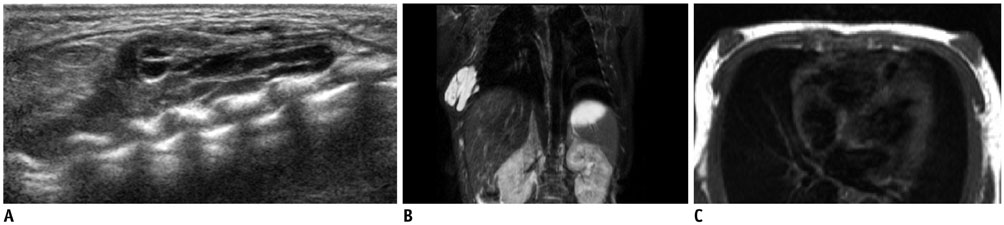
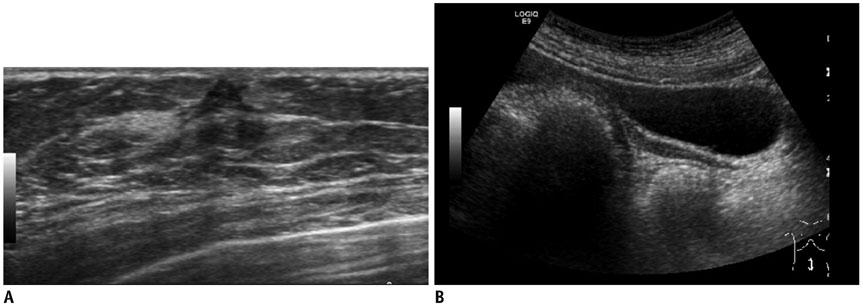
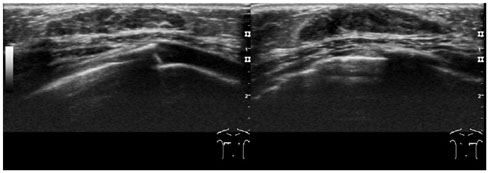
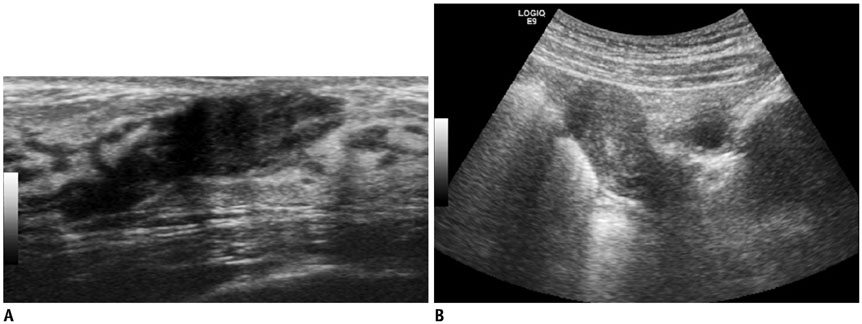
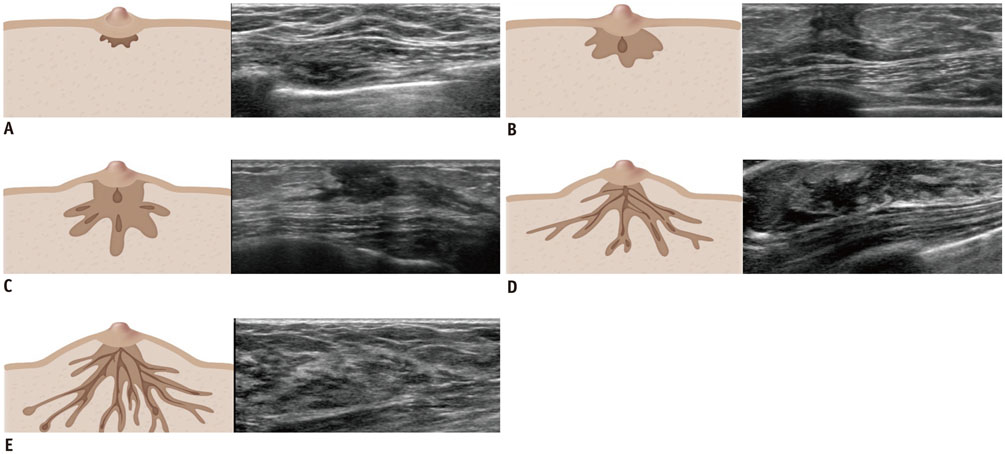
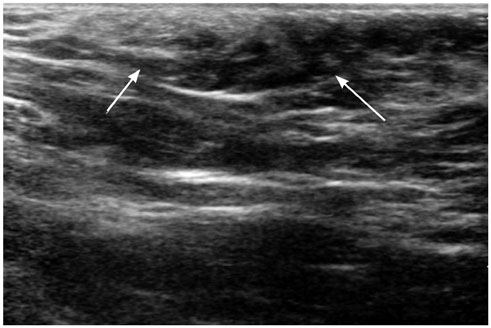
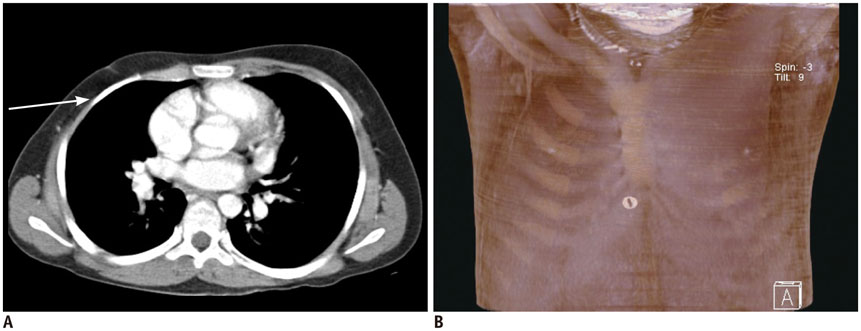
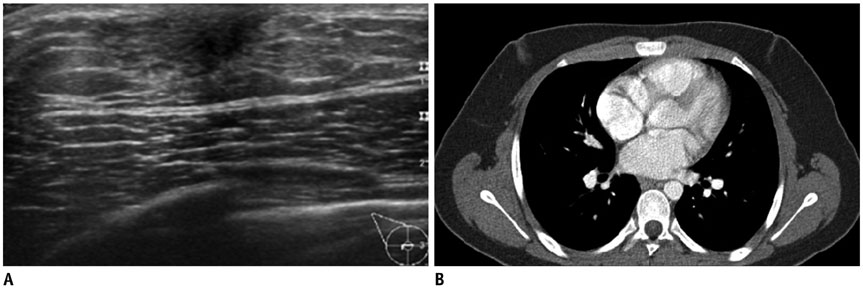
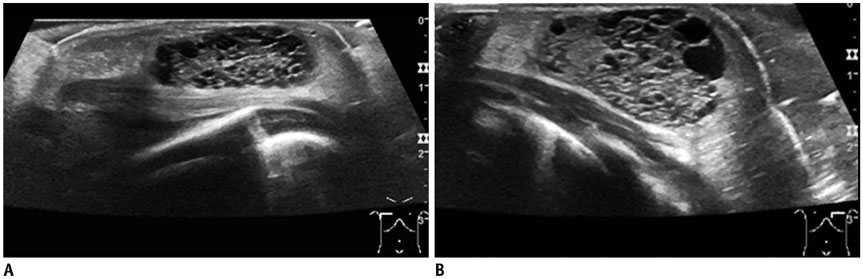
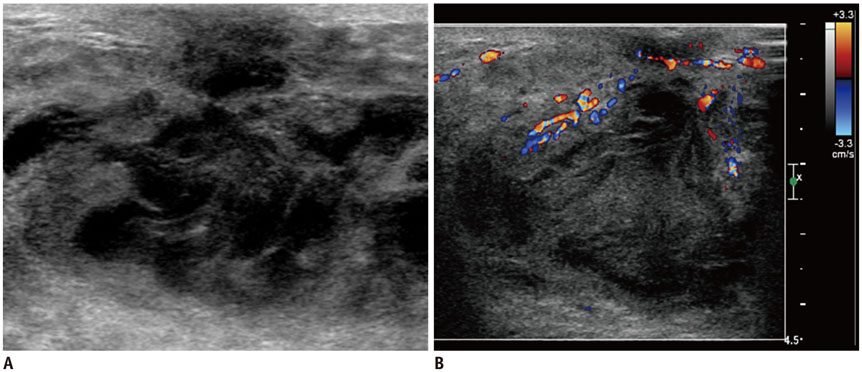
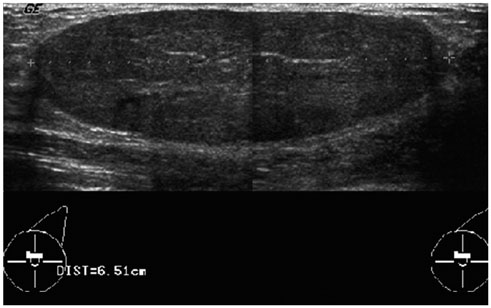
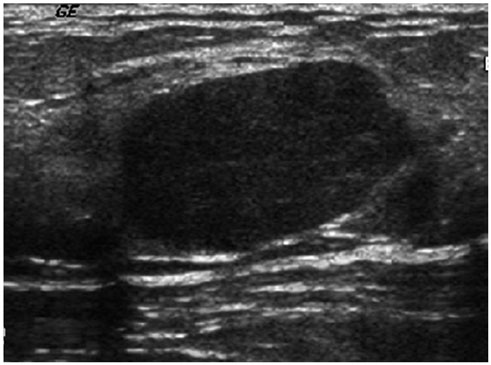
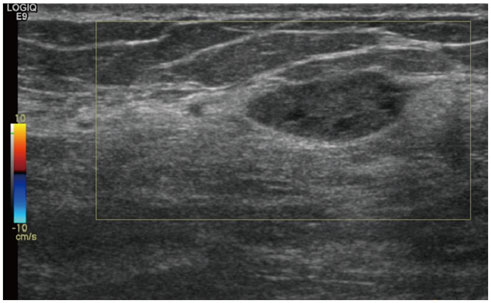
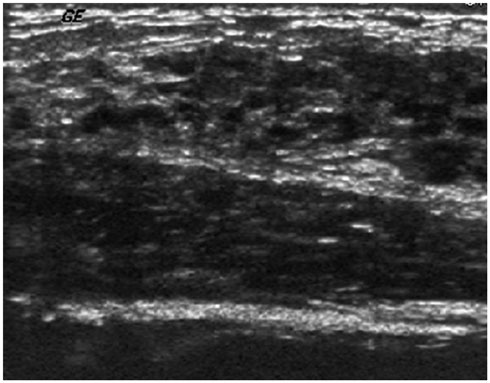
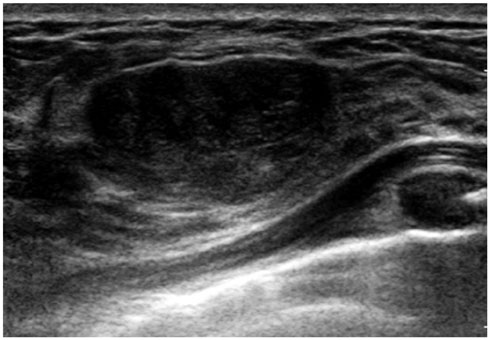
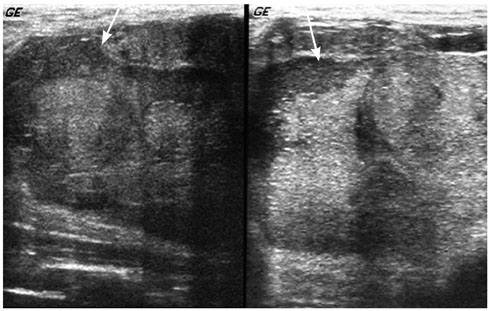
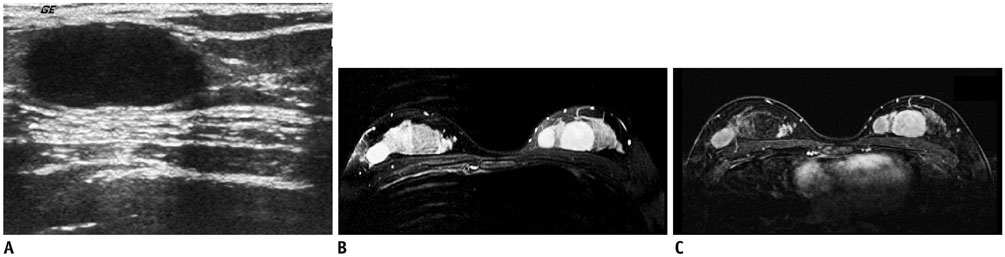
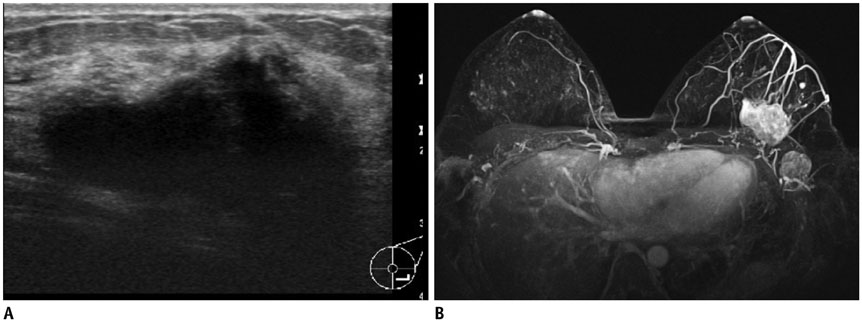
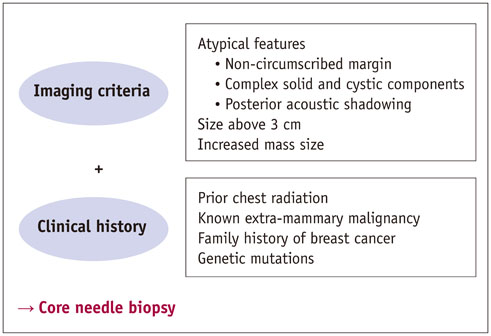
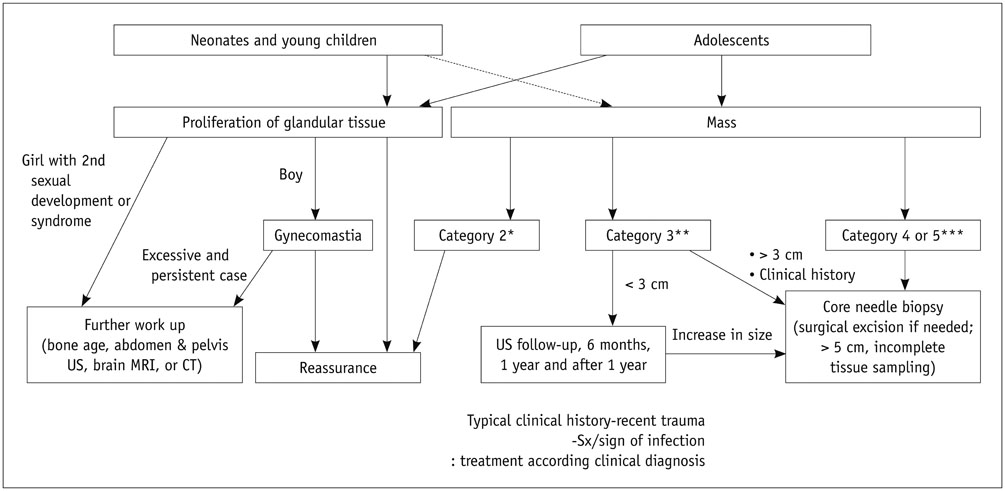
- Pediatric breast disease is uncommon, and primary breast carcinoma in children is extremely rare. Therefore, the approach used to address breast lesions in pediatric patients differs from that in adults in many ways. Knowledge of the normal imaging features at various stages of development and the characteristics of breast disease in the pediatric population can help the radiologist to make confident diagnoses and manage patients appropriately. Most breast diseases in children are benign or associated with breast development, suggesting a need for conservative treatment. Interventional procedures might affect the developing breast and are only indicated in a limited number of cases. Histologic examination should be performed in pediatric patients, taking into account the size of the lesion and clinical history together with the imaging findings. A core needle biopsy is useful for accurate diagnosis and avoidance of irreparable damage in pediatric patients. Biopsy should be considered in the event of abnormal imaging findings, such as non-circumscribed margins, complex solid and cystic components, posterior acoustic shadowing, size above 3 cm, or an increase in mass size. A clinical history that includes a risk factor for malignancy, such as prior chest irradiation, known concurrent cancer not involving the breast, or family history of breast cancer, should prompt consideration of biopsy even if the lesion has a probably benign appearance on ultrasonography.
Keyword
MeSH Terms
Figure
Reference
-
1. Valeur NS, Rahbar H, Chapman T. Ultrasound of pediatric breast masses: what to do with lumps and bumps. Pediatr Radiol. 2015; 45:1584–1599. quiz 1581-1583.
Article2. Chung EM, Cube R, Hall GJ, González C, Stocker JT, Glassman LM. From the archives of the AFIP: breast masses in children and adolescents: radiologic-pathologic correlation. Radiographics. 2009; 29:907–931.3. García CJ, Espinoza A, Dinamarca V, Navarro O, Daneman A, García H, et al. Breast US in children and adolescents. Radiographics. 2000; 20:1605–1612.
Article4. Kronemer KA, Rhee K, Siegel MJ, Sievert L, Hildebolt CF. Gray scale sonography of breast masses in adolescent girls. J Ultrasound Med. 2001; 20:491–496. quiz 498.
Article5. Sanchez R, Ladino-Torres MF, Bernat JA, Joe A, DiPietro MA. Breast fibroadenomas in the pediatric population: common and uncommon sonographic findings. Pediatr Radiol. 2010; 40:1681–1689.
Article6. Gutierrez JC, Housri N, Koniaris LG, Fischer AC, Sola JE. Malignant breast cancer in children: a review of 75 patients. J Surg Res. 2008; 147:182–188.
Article7. Kaneda HJ, Mack J, Kasales CJ, Schetter S. Pediatric and adolescent breast masses: a review of pathophysiology, imaging, diagnosis, and treatment. AJR Am J Roentgenol. 2013; 200:W204–W212.
Article8. Gao Y, Saksena MA, Brachtel EF, terMeulen DC, Rafferty EA. How to approach breast lesions in children and adolescents. Eur J Radiol. 2015; 84:1350–1364.
Article9. Baker JA, Soo MS, Rosen EL. Artifacts and pitfalls in sonographic imaging of the breast. AJR Am J Roentgenol. 2001; 176:1261–1266.
Article10. Durmaz E, Öztek MA, Arıöz Habibi H, Kesimal U, Sindel HT. Breast diseases in children: the spectrum of radiologic findings in a cohort study. Diagn Interv Radiol. 2017; 23:407–413.
Article11. Bock K, Duda VF, Hadji P, Ramaswamy A, Schulz-Wendtland R, Klose KJ, et al. Pathologic breast conditions in childhood and adolescence: evaluation by sonographic diagnosis. J Ultrasound Med. 2005; 24:1347–1354. quiz 1356-1357.12. Youn I, Park SH, Lim IS, Kim SJ. Ultrasound assessment of breast development: distinction between premature thelarche and precocious puberty. AJR Am J Roentgenol. 2015; 204:620–624.
Article13. Adler DD, Rebner M, Pennes DR. Accessory breast tissue in the axilla: mammographic appearance. Radiology. 1987; 163:709–711.
Article14. Mojallal A, La Marca S, Shipkov C, Sinna R, Braye F. Poland syndrome and breast tumor: a case report and review of the literature. Aesthet Surg J. 2012; 32:77–83.
Article15. Zhang F, Qi X, Xu Y, Zhou Y, Zhang Y, Fan L, et al. Breast cancer and Poland's syndrome: a case report and literature review. Breast J. 2011; 17:196–200.
Article16. Braunstein GD. Gynecomastia. N Engl J Med. 1993; 328:490–495.
Article17. Weinstein SP, Conant EF, Orel SG, Zuckerman JA, Bellah R. Spectrum of US findings in pediatric and adolescent patients with palpable breast masses. Radiographics. 2000; 20:1613–1621.
Article18. Huneeus A, Schilling A, Horvath E, Pinochet M, Carrasco O. Retroareolar cysts in the adolescent. J Pediatr Adolesc Gynecol. 2003; 16:45–49.
Article19. Welch ST, Babcock DS, Ballard ET. Sonography of pediatric male breast masses: gynecomastia and beyond. Pediatr Radiol. 2004; 34:952–957.
Article20. Soo MS, Kornguth PJ, Hertzberg BS. Fat necrosis in the breast: sonographic features. Radiology. 1998; 206:261–269.
Article21. Simmons PS. Diagnostic considerations in breast disorders of children and adolescents. Obstet Gynecol Clin North Am. 1992; 19:91–102.
Article22. Stavros AT, Thickman D, Rapp CL, Dennis MA, Parker SH, Sisney GA. Solid breast nodules: use of sonography to distinguish between benign and malignant lesions. Radiology. 1995; 196:123–134.
Article23. Sanders LM, Sharma P, El Madany M, King AB, Goodman KS, Sanders AE. Clinical breast concerns in low-risk pediatric patients: practice review with proposed recommendations. Pediatr Radiol. 2018; 48:186–195.
Article24. Patterson SK, Jorns JM. A case of juvenile papillomatosis, aka “Swiss cheese disease”. Breast J. 2013; 19:440–441.
Article25. Gill J, Greenall M. Juvenile papillomatosis and breast cancer. J Surg Educ. 2007; 64:234–236.
Article26. Tse GM, Niu Y, Shi HJ. Phyllodes tumor of the breast: an update. Breast Cancer. 2010; 17:29–34.
Article27. Cecen E, Uysal KM, Harmancioglu O, Balci P, Kupelioglu A, Canda T. Phyllodes tumor of the breast in an adolescent girl. Pediatr Hematol Oncol. 2008; 25:79–82.
Article28. Yabuuchi H, Soeda H, Matsuo Y, Okafuji T, Eguchi T, Sakai S, et al. Phyllodes tumor of the breast: correlation between MR findings and histologic grade. Radiology. 2006; 241:702–709.
Article29. Surov A, Holzhausen HJ, Wienke A, Schmidt J, Thomssen C, Arnold D, et al. Primary and secondary breast lymphoma: prevalence, clinical signs and radiological features. Br J Radiol. 2012; 85:e195–e205.
Article30. Wienbeck S, Meyer HJ, Uhlig J, Herzog A, Nemat S, Teifke A, et al. Radiological imaging characteristics of intramammary hematological malignancies: results from a german multicenter study. Sci Rep. 2017; 7:7435.
Article31. Kennedy RD, Boughey JC. Management of pediatric and adolescent breast masses. Semin Plast Surg. 2013; 27:19–22.
Article32. Murphy JJ, Morzaria S, Gow KW, Magee JF. Breast cancer in a 6-year-old child. J Pediatr Surg. 2000; 35:765–767.
Article33. Horowitz DP, Sharma CS, Connolly E, Gidea-Addeo D, Deutsch I. Secretory carcinoma of the breast: results from the survival, epidemiology and end results database. Breast. 2012; 21:350–353.
Article34. Ha SM, Chae EY, Cha JH, Kim HH, Shin HJ, Choi WJ. Association of BRCA mutation types, imaging features, and pathologic findings in patients with breast cancer with BRCA1 and BRCA2 mutations. AJR Am J Roentgenol. 2017; 209:920–928.
Article35. Lee MV, Katabathina VS, Bowerson ML, Mityul MI, Shetty AS, Elsayes KM, et al. BRCA-associated cancers: role of imaging in screening, diagnosis, and management. Radiographics. 2017; 37:1005–1023.36. Mendelson EB, Böhm-Vélez M, Berg WA, et al. ACR BI-RADS ultrasound. ACR BI-RADS Atlas. Breast imaging reporting and data system. 5th ed. Reston, VA: American College of Radiology;2013. p. 128–130.37. Gordon PB, Gagnon FA, Lanzkowsky L. Solid breast masses diagnosed as fibroadenoma at fine-needle aspiration biopsy: acceptable rates of growth at long-term follow-up. Radiology. 2003; 229:233–238.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Common and Uncommon Conditions of Breast Disease in Children and Adolescents: A Pictorial Review
- Metabolic syndrome in children and adolescents
- Dizziness in children and adolescents
- Clinical Analysis of Female Patients Under 20 Years of Age with Benign Breast Masses
- Social Services Information for Children and Adolescents with Diabetes Mellitus