Obstet Gynecol Sci.
2018 May;61(3):352-358. 10.5468/ogs.2018.61.3.352.
Retrospective study of combination chemotherapy with etoposide and ifosfamide in patients with heavily pretreated recurrent or persistent epithelial ovarian cancer
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. bgkim@skku.edu
- 2Department of Statistics, Hankuk University of Foreign Studies, Seoul, Korea.
- KMID: 2416123
- DOI: http://doi.org/10.5468/ogs.2018.61.3.352
Abstract
OBJECTIVE
This retrospective study is to evaluate the efficacy and toxicity of combination chemotherapy with etoposide and ifosfamide (ETI) in the management of pretreated recurrent or persistent epithelial ovarian cancer (EOC).
METHODS
Patients with recurrent or persistent EOC who had measurable disease and at least one chemotherapy regimen were to receive etoposide at a dose of 100 mg/m²/day intravenous (IV) on days 1 to 3 in combination with ifosfamide 1 g/m²/day IV on days 1 to 5, every 21 days.
RESULTS
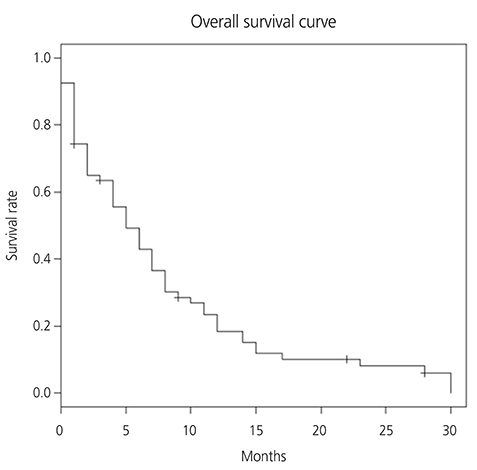
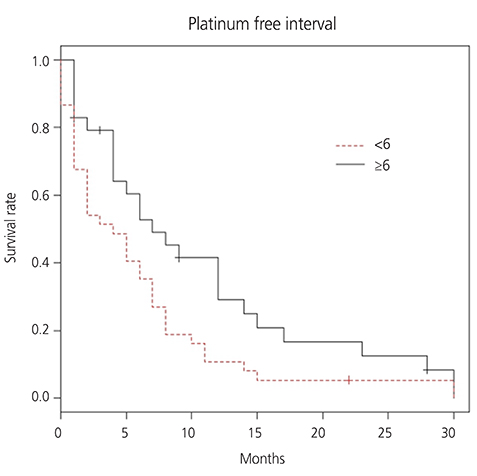
From August 2008 to August 2016, 66 patients were treated with ETI regimen. Most patients were heavily pretreated prior to ETI: 53 (80.3%) patients had received 3 or more chemotherapy regimens. The response rate (RR) of ETI chemotherapy was 18.2% and median duration of response was 6.8 months (range, 0-30). Median survival of all patients was 5 months at a median follow up of 7.2 months. Platinum-free interval (PFI) more than 6 months prior to ETI has statistically significant correlation with overall survival (OS; 9.2 vs. 5.6 months; P=0.029) and RR (34.5% vs. 5.4%; P < 0.010). However, treatment free interval before ETI, number of prior chemotherapy regimen, and optimality of primary surgery did not show significant difference for RR or OS. Grade 3 or 4 hematologic toxicities were observed in 7 cases (3%) of the 232 cycles of ETI.
CONCLUSION
The ETI combination regimen shows comparatively low toxicity and modest activity in heavily pretreated recurrent or persistent EOC patients with more than 6 months of PFI after last platinum treatment.
Keyword
MeSH Terms
Figure
Reference
-
1. Mirza MR, Monk BJ, Herrstedt J, Oza AM, Mahner S, Redondo A, et al. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016; 375:2154–2164.
Article2. McGuire WP, Hoskins WJ, Brady MF, Kucera PR, Partridge EE, Look KY, et al. Cyclophosphamide and cisplatin compared with paclitaxel and cisplatin in patients with stage III and stage IV ovarian cancer. N Engl J Med. 1996; 334:1–6.
Article3. Hanker LC, Loibl S, Burchardi N, Pfisterer J, Meier W, Pujade-Lauraine E, et al. The impact of second to sixth line therapy on survival of relapsed ovarian cancer after primary taxane/platinum-based therapy. Ann Oncol. 2012; 23:2605–2612.
Article4. Bookman MA, Malmstrom H, Bolis G, Gordon A, Lissoni A, Krebs JB, et al. Topotecan for the treatment of advanced epithelial ovarian cancer: an open-label phase II study in patients treated after prior chemotherapy that contained cisplatin or carboplatin and paclitaxel. J Clin Oncol. 1998; 16:3345–3352.
Article5. Bruchim I, Ben-Harim Z, Piura E, Haran G, Fishman A. Analysis of two topotecan treatment schedules in patients with recurrent ovarian cancer. J Chemother. 2016; 28:129–134.
Article6. Shapiro JD, Millward MJ, Rischin D, Michael M, Walcher V, Francis PA, et al. Activity of gemcitabine in patients with advanced ovarian cancer: responses seen following platinum and paclitaxel. Gynecol Oncol. 1996; 63:89–93.
Article7. Takei Y, Takahashi Y, Machida S, Taneichi A, Takahashi S, Nagashima T, et al. Response to and toxicity of gemcitabine for recurrent ovarian cancer according to number of previous chemotherapy regimens. J Obstet Gynaecol Res. 2017; 43:358–364.
Article8. Markman M, Kennedy A, Webster K, Peterson G, Kulp B, Belinson J. Phase 2 trial of liposomal doxorubicin (40 mg/m(2)) in platinum/paclitaxel-refractory ovarian and fallopian tube cancers and primary carcinoma of the peritoneum. Gynecol Oncol. 2000; 78:369–372.
Article9. Kucukoner M, Isikdogan A, Yaman S, Gumusay O, Unal O, Ulas A, et al. Oral etoposide for platinum-resistant and recurrent epithelial ovarian cancer: a study by the Anatolian Society of Medical Oncology. Asian Pac J Cancer Prev. 2012; 13:3973–3976.
Article10. Markman M, Kennedy A, Sutton G, Hurteau J, Webster K, Peterson G, et al. Phase 2 trial of single agent ifosfamide/mesna in patients with platinum/paclitaxel refractory ovarian cancer who have not previously been treated with an alkylating agent. Gynecol Oncol. 1998; 70:272–274.
Article11. Hainsworth JD, Greco FA. Etoposide: twenty years later. Ann Oncol. 1995; 6:325–341.
Article12. Maskens AP, Armand JP, Lacave AJ, De Jager RL, Hansen HH, Wolff JP. Phase II clinical trial of VP-16-213 in ovarian cancer. Cancer Treat Rep. 1981; 65:329–330.13. Tropé CG, Kisic J, Vergote I. Prognostic factors in platinum-resistant ovarian carcinoma treated with ifosfamide-etoposide. Eur J Gynaecol Oncol. 2000; 21:255–259.14. Kang H, Kim TJ, Choi CH, Lee JW, Lee JH, Bae DS, et al. Phase II study of combination chemotherapy with etoposide and ifosfamide in patients with heavily pretreated recurrent or persistent epithelial ovarian cancer. J Korean Med Sci. 2009; 24:945–950.
Article15. Furlanut M, Franceschi L. Pharmacology of ifosfamide. Oncology. 2003; 65:Suppl 2. 2–6.
Article16. Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009; 45:228–247.
Article17. Fujiwara K, Nagao S, Aotani E, Hasegawa K. Principle and evolving role of intraperitoneal chemotherapy in ovarian cancer. Expert Opin Pharmacother. 2013; 14:1797–1806.
Article18. Tomao F, D'Incalci M, Biagioli E, Peccatori FA, Colombo N. Restoring platinum sensitivity in recurrent ovarian cancer by extending the platinum-free interval: myth or reality? Cancer. 2017; 123:3450–3459.
Article19. Bolton KL, Chenevix-Trench G, Goh C, Sadetzki S, Ramus SJ, Karlan BY, et al. Association between BRCA1 and BRCA2 mutations and survival in women with invasive epithelial ovarian cancer. JAMA. 2012; 307:382–390.20. Ledermann JA. Benefits of enhancing the platinum-free interval in the treatment of relapsed ovarian cancer: more than just a hypothesis? Int J Gynecol Cancer. 2011; 21:Suppl 1. S9–S11.
Article21. Pignata S, Ferrandina G, Scarfone G, Scollo P, Odicino F, Selvaggi L, et al. Extending the platinum-free interval with a non-platinum therapy in platinum-sensitive recurrent ovarian cancer. Results from the SOCRATES Retrospective Study. Oncology. 2006; 71:320–326.22. See HT, Freedman RS, Kudelka AP, Burke TW, Gershenson DM, Tangjitgamol S, et al. Retrospective review: re-treatment of patients with ovarian cancer with carboplatin after platinum resistance. Int J Gynecol Cancer. 2005; 15:209–216.
Article23. Bruzzone M, Campora E, Merlini L, Giudici S, Bottero G, Iskra L, et al. Ifosfamide and etoposide salvage treatment in advanced ovarian cancer. J Chemother. 1991; 3:332–334.
Article24. Tropé C, Kaern J, Vergote I, Vossli S. A phase II study of etoposide combined with ifosfamide as second-line therapy in cisplatin-resistant ovarian carcinomas. Cancer Chemother Pharmacol. 1990; 26:Suppl. S45–S47.25. Aravantinos G, Dimopoulos MA, Kosmidis P, Bafaloukos D, Papadimitriou C, Kiamouris C, et al. Ifosfamide plus oral etoposide salvage chemotherapy for platinum-resistant paclitaxel-pretreated ovarian cancer. Ann Oncol. 2000; 11:607–612.
Article26. Shaheen M, Stender MJ, McClean JW, Look KY, Einhorn LH. Phase II study of ifosfamide plus daily oral etoposide in previously treated ovarian cancer: a Hoosier Oncology Group (HOG) study. Am J Clin Oncol. 2004; 27:229–231.27. Webber K, Friedlander M. Chemotherapy for epithelial ovarian, fallopian tube and primary peritoneal cancer. Best Pract Res Clin Obstet Gynaecol. 2017; 41:126–138.
Article28. Rothenberg ML, Liu PY, Wilczynski S, Nahhas WA, Winakur GL, Jiang CS, et al. Phase II trial of vinorelbine for relapsed ovarian cancer: a Southwest Oncology Group study. Gynecol Oncol. 2004; 95:506–512.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Phase II Study of Combination Chemotherapy with Etoposide and Ifosfamide in Patients with Heavily Pretreated Recurrent or Persistent Epithelial Ovarian Cancer
- Clinical Trial of IV Etoposide and Carboplatin and Cyclophosphamide Combination Chemotherapy Against Persistent or Recurrent Ovarian Cancer as 2nd or More Line Chemotherapy
- Clinical Efficacy of Ifosfamide-Based Regimen in Refractory of Relapsed Ovarian Cancer
- Fatal Ifosfamide-Induced Metabolic Encephalopathy in Patients with Recurrent Epithelial Ovarian Cancer: Report of Two Cases
- Ifosfamide and Etoposide in Relapsed Refractory Childhood Acute Lymphoblastic Leukemia