Nat Prod Sci.
2018 Jun;24(2):115-118. 10.20307/nps.2018.24.2.115.
Ethyl Haematommate from Stereocaulon graminosum Schaer.: Isolation and Crystal Structure
- Affiliations
-
- 1The Laboratory of Natural Resource of Sumatra and Faculty of Pharmacy, Andalas University, 26163 Padang, Indonesia. friardi@ffarmasi.unand.ac.id
- 2School of Pharmacy and Pharmaceutical Sciences, Hoshi University, 2-4-41, Ebara, Shinagawa, Tokyo 142-8501, Japan. e-yonemochi@hoshi.ac.jp
- 3Current Address: Pharmaceutical Technology and Development, AstraZeneca, Pepparedsleden 1, Mölndal S-431 83, Sweden.
- KMID: 2416072
- DOI: http://doi.org/10.20307/nps.2018.24.2.115
Abstract
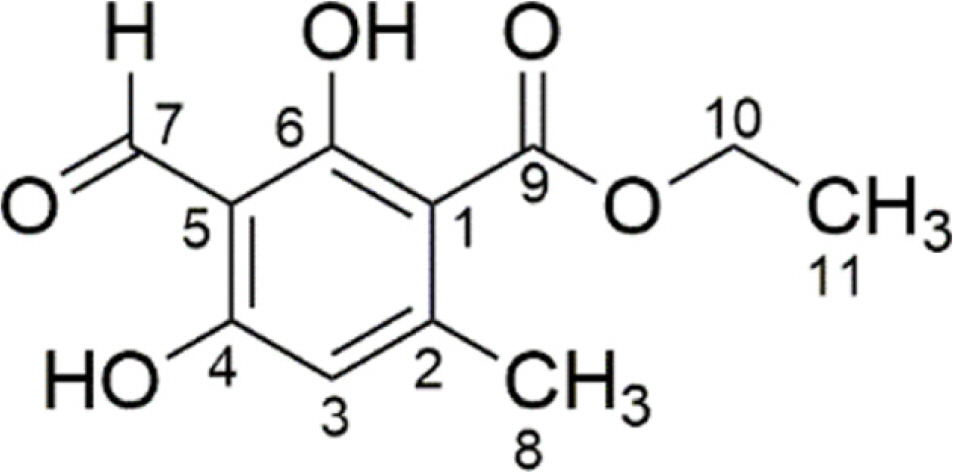
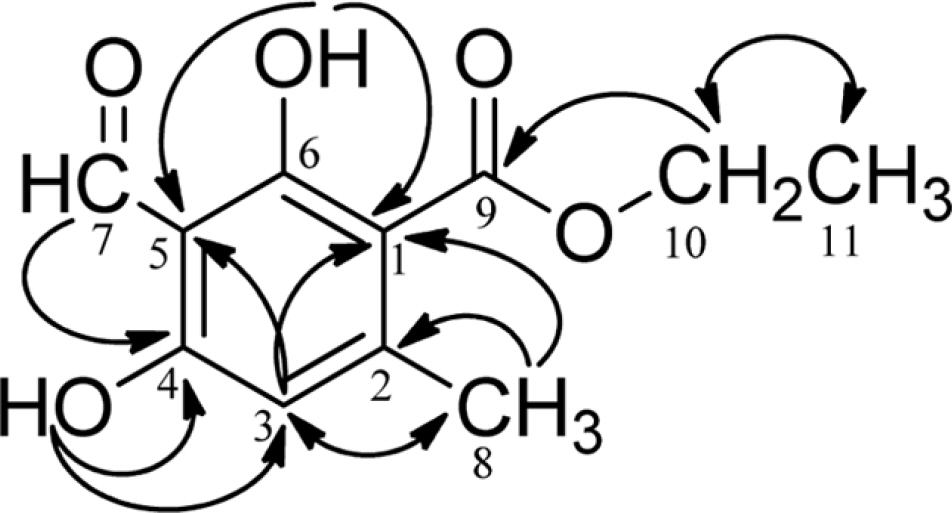
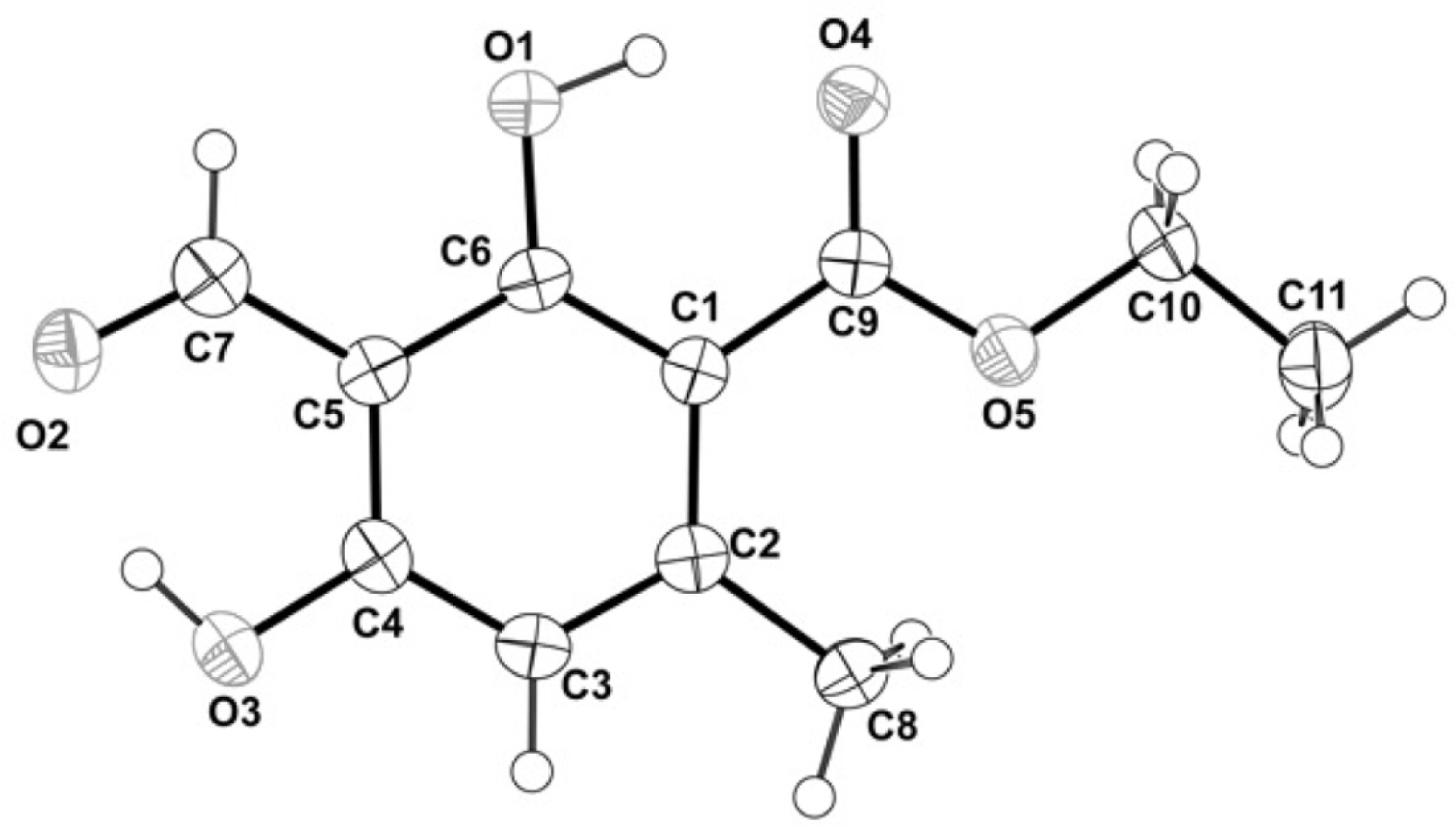
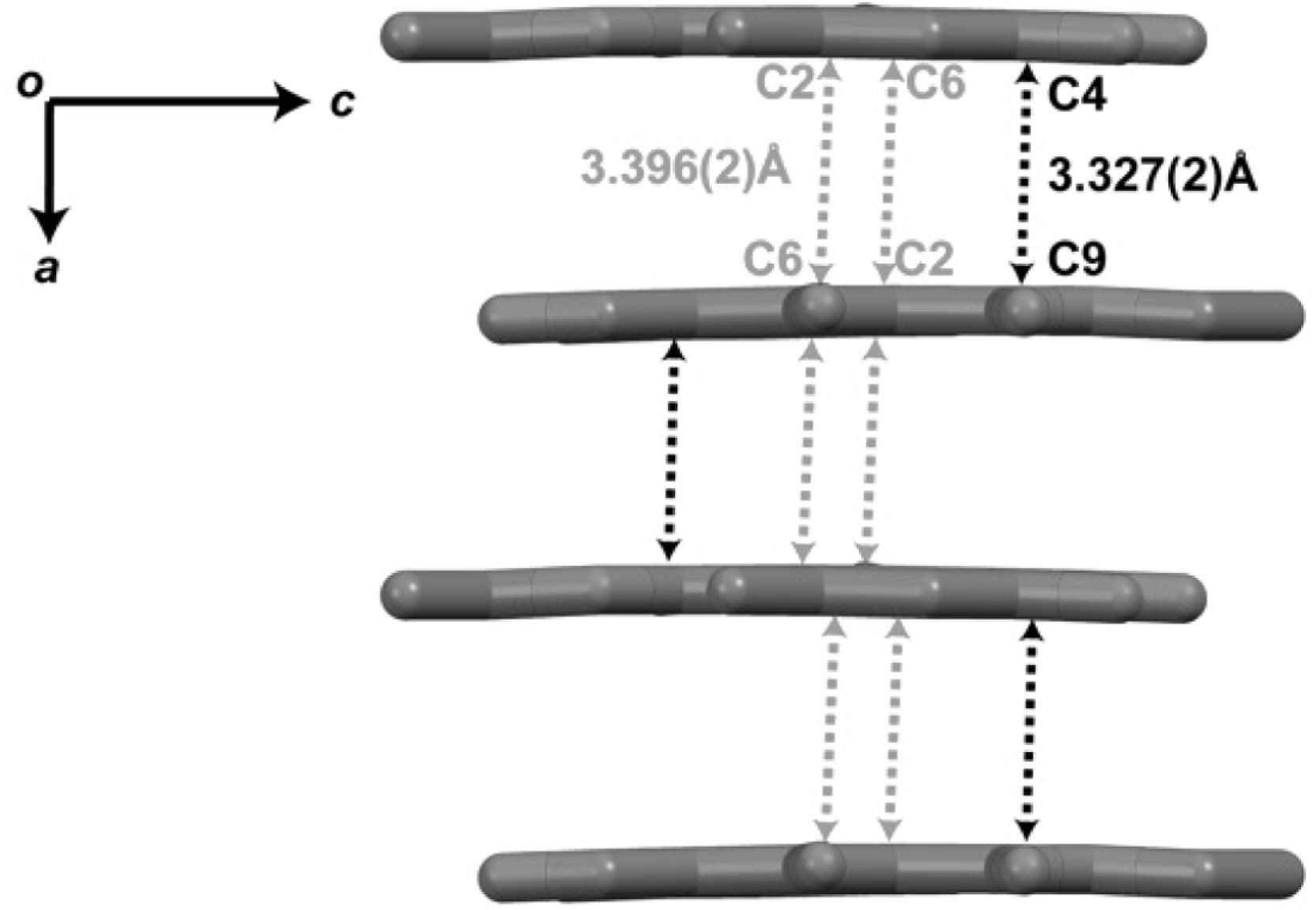
- Herein, we reported the phytochemical investigation of whole thallus Sumatran lichen, Stereocaulon graminosum Schaer, and isolated a mono aromatic compound, ethyl haematommate (1). The structure of compound 1 have been established based on spectroscopic data and confirmed by single crystal X-ray structure analysis.
Keyword
MeSH Terms
Figure
Reference
-
References
(1). Singh R.., Ranjan S.., Nayaka S.., Pathre U. V.., Shirke P. A.Acta Physiol. Plant. 2013. 35:1605–1615.(2). Ismed F.., Lohézic-Le Dévéhat F.., Delalande O.., Sinbandhit S.., Bakhtiar A.., Boustie J.Fitoterapia. 2012. 83:1693–1698.(3). Ismed F.., Lohézic-Le Dévéhat F.., Rouaud I.., Ferron S.., Bakhtiar A.., Boustie J. Z.Naturforsch. C. 2017. 72:55–62.(4). Stocker-Wörgötter E.Nat. Prod. Rep. 2008. 25:188–200.(5). Seshadri T. R.Proc. Indian Acad. Sci. 1944. 20:1–14.(6). Marante F. J. T.., Castellano A. G.., Rosas F. E.., Aguiar J. Q.., Barrera J. B. J.Chem. Ecol,. 2003. 29:2049–2071.
Article(7). Choudhary M. I.., Ali M.., Wahab A.., Khan A.., Rasheed S.., Shyaula S. L.., Rahman A.Sci. China Chem. 2011. 54:1926–1931.(8). Manojlovic N. T.., Vasiljevic P. J.., Maskovic P. Z.., Juskovic M.., Bogdanovic-Dusanovic G.Evid. Based Complement. Alternat. Med. 2012. 2012:452431.(9). Huneck S.., Yoshimura I.Identification of lichen substances. Springer;United States: 1996. . pp.p. 421–422.(10). Calculated using ABSCOR. Empirical Absorption Correction Based on Fourier Series Approximation; Rigaku: Texas, 1994. Based on Fourier Series Approximation; Rigaku: Texas,. 1994.(11). Burla M. C.., Caliandro R.., Camalli M.., Carrozzini B.., Cascarano G. L.., De Caro L.., Giacovazzo C.., Polidori G.., Spagna R. J.Appl. Cryst. 2005. 38:381–388.(12). Sheldrick G. M.Acta Crystallogr. A. 2008. 64:112–122.(13). Macrae C. F.., Bruno I. J.., Chisholm J. A.., Edgington P. R.., McCabe P.., Pidcock E.., Rodriguez-Monge L.., Taylor R.., van de Streek J.., Wood P. A. J.Appl. Cryst. 2008. 41:466–470.(14). Rao P. S.., Sarma K. G.., Seshadri T. R.Proc. Indian Acad. Sci. 1967. 66:1–14.(15). Putra O. D.., Furuishi T.., Yonemochi E.., Terada K.., Uekusa H.Cryst. Growth Des. 2016. 16:3577–3581.(16). Ismed F.., Farhan A.., Bakhtiar A.., Zaini E.., Nugraha Y. P.., Dwichandra Putra, O. Uekusa H.Acta Crystallogr. E Crystallogr. Commun. 2016. 72:1587–1589.(17). Putra O. D.., Umeda D.., Nugraha Y. P.., Furuishi T.., Nagase H.., Fukuzawa K.., Uekusa H.., Yonemochi E.CrystEngComm. 2017. 19:2614–2622.(18). Groom C. R.., Bruno I. J.., Lightfoot M. P.., Ward S.C.; Acta Crystallogr. B Struct. Sci. Cryst. Eng. Mater. 2016. 72:171–179.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Revision of the Lichen Genus Stereocaulon (Stereocaulaceae, Ascomycota) in South Korea
- Changes in surface content and crystal structure after fluoride gel or hydroxyapatite paste application on stripped enamel
- Lethal Effects of Helianthemum lippii (L.) on Acanthamoeba castellanii Cysts in Vitro
- Do Face Masks become Worthless after Only One Use in the COVID-19 Pandemic?
- A Case of Calcium Pyrophosphate Dihydrate Crystal Deposition Disease Associated with Gout