Int J Stem Cells.
2018 Jun;11(1):1-12. 10.15283/ijsc17061.
Mesenchymal Stem Cell Therapy for Ischemic Heart Disease: Systematic Review and Meta-analysis
- Affiliations
-
- 1Department of Preventive Medicine, College of Medicine, The Catholic University of Korea, Seoul, Korea. y1693@catholic.ac.kr
- 2Division of Cardiology, Department of Internal Medicine, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- 3Cheong-yang Branch Office of the Community Health Center, Cheongyang, Korea.
- 4Medical Library, The Catholic University of Korea, Seoul, Korea.
- 5Catholic High-Performance Cell Therapy Center & Department of Medical Life Science, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- KMID: 2413496
- DOI: http://doi.org/10.15283/ijsc17061
Abstract
- BACKGROUND AND OBJECTIVES
Mesenchymal stem cells (MSC) have emerged as breakthrough treatments for myocardial infarction. However, the efficacy of MSC remains unclear. The aim of the study was to evaluate treatment effect of MSC in terms of mechanical, regenerative, and clinical outcomes for patients with myocardial infarction (MI) using meta-analysis.
METHODS
A systematic search and critical review of MEDLINE, EMBASE, and Cochrane database literature published from inception through December 2017 was performed. The inclusion criteria were randomized controlled trials, studies on patients with myocardial infarction, and studies compared with placebo as a control group.
RESULTS
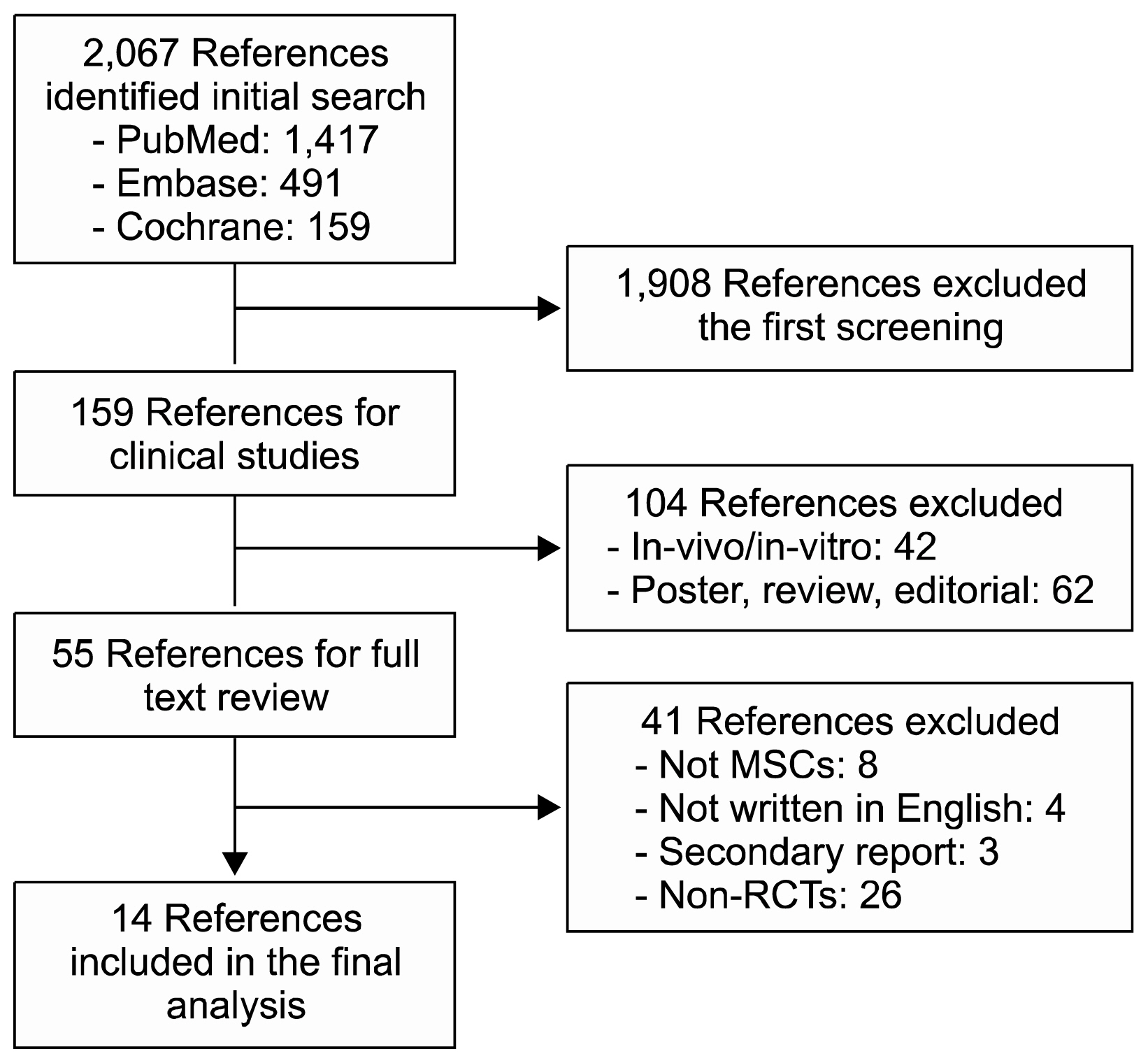
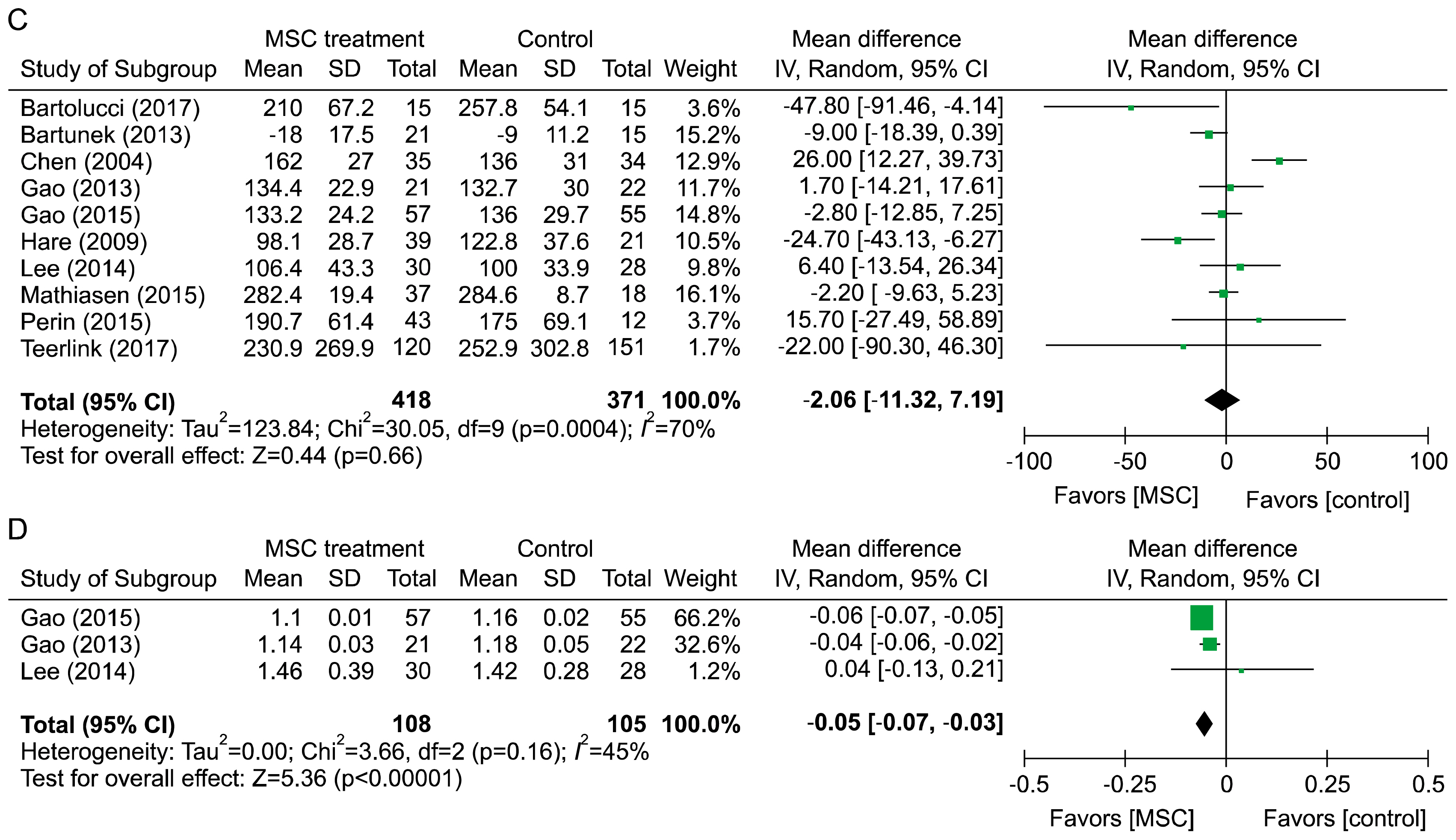
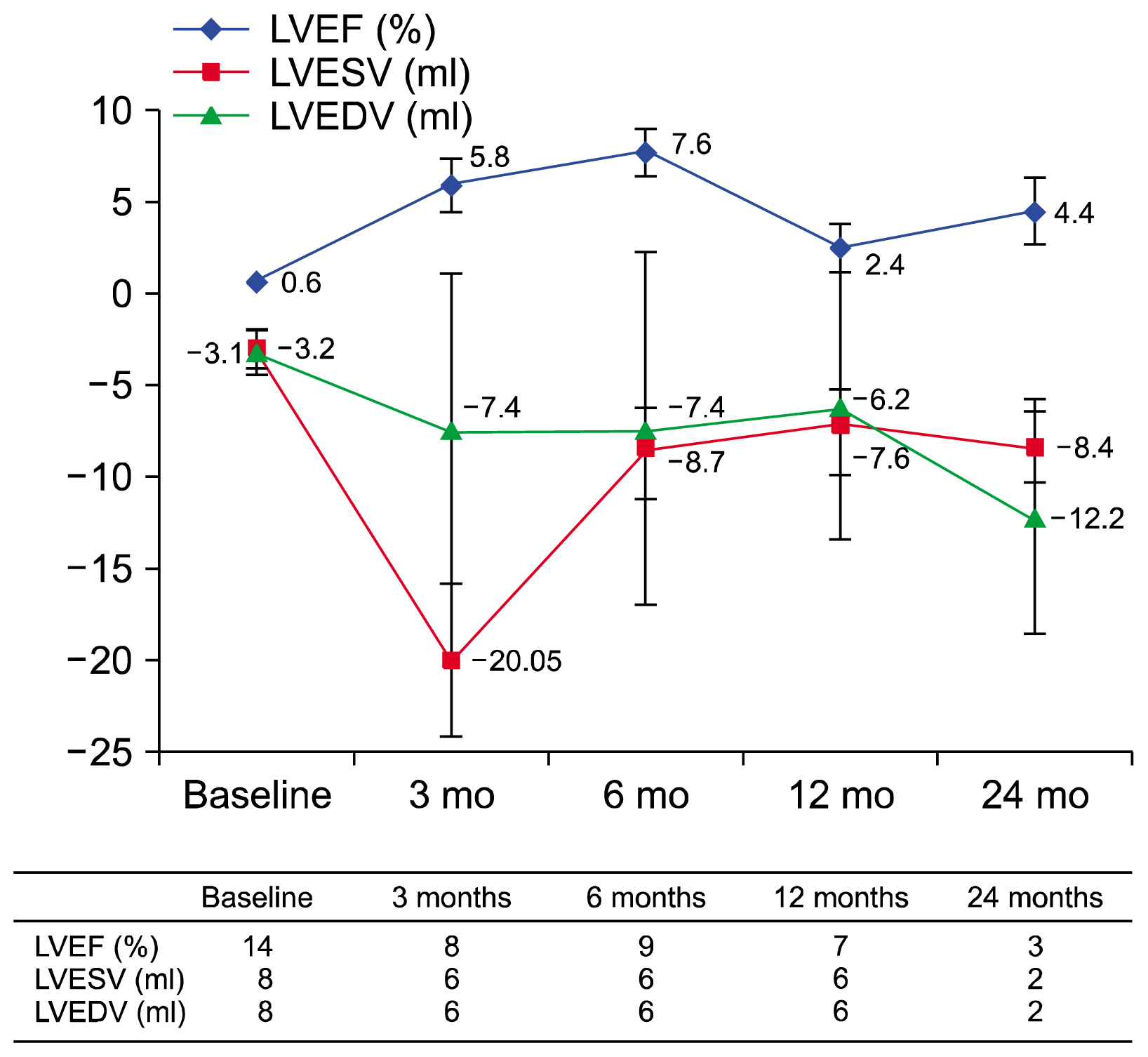
A total of 950 patients from 14 randomized placebo controlled trials were included in the final meta-analysis. MSC treatment showed benefits for mechanical, regenerative, and clinical outcomes. In terms of mechanical outcomes, the LVEF of the MSC treatment group increased by 3.84% (95% CI: 2.32~5.35, I 2=43) and the effect was maintained for up to 24 months. Regenerative outcomes were measured by scar mass and WMSI. Scar mass was reduced by −1.13 (95% CI: −1.80 to −0.46, I 2=71) and WMSI was reduced by −0.05 (95% CI: −0.07 to −0.03, I 2=45) at 6 months after MSC treatment. Mortality rate and incidence of re-hospitalization for HF in MSC group patients trended toward reduced incidence compared to the control group, although this was not statistically significant because of the low event rate.
CONCLUSIONS
The findings of this meta-analysis indicate that MSCs can be beneficial in improving heart function in the treatment of MI. However, the efficacy of MSCs must be further explored through large randomized controlled trials based on rigorous research design.
MeSH Terms
Figure
Reference
-
References
1. Lipinski MJ, Biondi-Zoccai GG, Abbate A, Khianey R, Sheiban I, Bartunek J, Vanderheyden M, Kim HS, Kang HJ, Strauer BE, Vetrovec GW. Impact of intracoronary cell therapy on left ventricular function in the setting of acute myocardial infarction: a collaborative systematic review and meta-analysis of controlled clinical trials. J Am Coll Cardiol. 2007; 50:1761–1767. DOI: 10.1016/j.jacc.2007.07.041. PMID: 17964040.
Article2. Martin-Rendon E, Brunskill SJ, Hyde CJ, Stanworth SJ, Mathur A, Watt SM. Autologous bone marrow stem cells to treat acute myocardial infarction: a systematic review. Eur Heart J. 2008; 29:1807–1818. DOI: 10.1093/eurheartj/ehn220. PMID: 18523058.
Article3. Zhang SN, Sun AJ, Ge JB, Yao K, Huang ZY, Wang KQ, Zou YZ. Intracoronary autologous bone marrow stem cells transfer for patients with acute myocardial infarction: a meta-analysis of randomised controlled trials. Int J Cardiol. 2009; 136:178–185. DOI: 10.1016/j.ijcard.2008.04.071.
Article4. Jeong H, Yim HW, Cho Y, Park HJ, Jeong S, Kim HB, Hong W, Kim H. The effect of rigorous study design in the research of autologous bone marrow-derived mononuclear cell transfer in patients with acute myocardial infarction. Stem Cell Res Ther. 2013; 4:82. DOI: 10.1186/scrt233. PMID: 23849537. PMCID: 3854784.
Article5. Gyöngyösi M, Wojakowski W, Lemarchand P, Lunde K, Tendera M, Bartunek J, Marban E, Assmus B, Henry TD, Traverse JH, Moyé LA, Sürder D, Corti R, Huikuri H, Miettinen J, Wöhrle J, Obradovic S, Roncalli J, Malliaras K, Pokushalov E, Romanov A, Kastrup J, Bergmann MW, Atsma DE, Diederichsen A, Edes I, Benedek I, Benedek T, Pejkov H, Nyolczas N, Pavo N, Bergler-Klein J, Pavo IJ, Sylven C, Berti S, Navarese EP, Maurer G. ACCRUE Investigators. Meta-Analysis of Cell-based CaRdiac stUdiEs (ACCRUE) in patients with acute myocardial infarction based on individual patient data. Circ Res. 2015; 116:1346–1360. DOI: 10.1161/CIRCRESAHA.116.304346. PMID: 25700037. PMCID: 4509791.
Article6. Fisher SA, Zhang H, Doree C, Mathur A, Martin-Rendon E. Stem cell treatment for acute myocardial infarction. Cochrane Database Syst Rev. 2015; (9):CD006536. PMID: 26419913.
Article7. Fisher SA, Doree C, Mathur A, Taggart DP, Martin-Rendon E. Stem cell therapy for chronic ischaemic heart disease and congestive heart failure. Cochrane Database Syst Rev. 2016; 12:CD007888. PMID: 28012165.
Article8. Izadpanah R, Trygg C, Patel B, Kriedt C, Dufour J, Gimble JM, Bunnell BA. Biologic properties of mesenchymal stem cells derived from bone marrow and adipose tissue. J Cell Biochem. 2006; 99:1285–1297. DOI: 10.1002/jcb.20904. PMID: 16795045. PMCID: 4048742.
Article9. Draper JS, Moore HD, Ruban LN, Gokhale PJ, Andrews PW. Culture and characterization of human embryonic stem cells. Stem Cells Dev. 2004; 13:325–336. DOI: 10.1089/scd.2004.13.325. PMID: 15345125.
Article10. Lee K, Majumdar MK, Buyaner D, Hendricks JK, Pittenger MF, Mosca JD. Human mesenchymal stem cells maintain transgene expression during expansion and differentiation. Mol Ther. 2001; 3:857–866. DOI: 10.1006/mthe.2001.0327. PMID: 11407899.
Article11. Toma C, Pittenger MF, Cahill KS, Byrne BJ, Kessler PD. Human mesenchymal stem cells differentiate to a cardiomyocyte phenotype in the adult murine heart. Circulation. 2002; 105:93–98. DOI: 10.1161/hc0102.101442. PMID: 11772882.
Article12. Ikegami Y, Miyoshi S, Nishiyama N, Hida N, Okamoto K, Miyado K, Segawa K, Ogawa S, Umezawa A. Serum-independent cardiomyogenic transdifferentiation in human endometrium-derived mesenchymal cells. Artif Organs. 2010; 34:280–288. DOI: 10.1111/j.1525-1594.2009.00859.x. PMID: 20420609.
Article13. Xu W, Zhang X, Qian H, Zhu W, Sun X, Hu J, Zhou H, Chen Y. Mesenchymal stem cells from adult human bone marrow differentiate into a cardiomyocyte phenotype in vitro. Exp Biol Med (Maywood). 2004; 229:623–631. DOI: 10.1177/153537020422900706.
Article14. Chullikana A, Majumdar AS, Gottipamula S, Krishnamurthy S, Kumar AS, Prakash VS, Gupta PK. Randomized, double-blind, phase I/II study of intravenous allogeneic mesenchymal stromal cells in acute myocardial infarction. Cytotherapy. 2015; 17:250–261. DOI: 10.1016/j.jcyt.2014.10.009.
Article15. Hare JM, Traverse JH, Henry TD, Dib N, Strumpf RK, Schulman SP, Gerstenblith G, DeMaria AN, Denktas AE, Gammon RS, Hermiller JB Jr, Reisman MA, Schaer GL, Sherman W. A randomized, double-blind, placebo-controlled, dose-escalation study of intravenous adult human mesenchymal stem cells (prochymal) after acute myocardial infarction. J Am Coll Cardiol. 2009; 54:2277–2286. DOI: 10.1016/j.jacc.2009.06.055. PMID: 19958962. PMCID: 3580848.
Article16. Perin EC, Borow KM, Silva GV, DeMaria AN, Marroquin OC, Huang PP, Traverse JH, Krum H, Skerrett D, Zheng Y, Willerson JT, Itescu S, Henry TD. A phase II dose-escalation study of allogeneic mesenchymal precursor cells in patients with ischemic or nonischemic heart failure. Circ Res. 2015; 117:576–584. DOI: 10.1161/CIRCRESAHA.115.306332. PMID: 26148930.
Article17. FDA. Guidance for Industry Cellular Therapy for Cardiac Disease [Internet]. [cited 2013 May 15]. Available from: http://www.fda.gov/downloads/BiologicsBloodVaccines/GuidanceComplianceRegulatoryInformation/Guidances/CellularandGeneTherapy/UCM164345.pdf.18. Strauer BE, Brehm M, Zeus T, Köstering M, Hernandez A, Sorg RV, Kögler G, Wernet P. Repair of infarcted myocardium by autologous intracoronary mononuclear bone marrow cell transplantation in humans. Circulation. 2002; 106:1913–1918. DOI: 10.1161/01.CIR.0000034046.87607.1C. PMID: 12370212.
Article19. Cintron G, Johnson G, Francis G, Cobb F, Cohn JN. Prognostic significance of serial changes in left ventricular ejection fraction in patients with congestive heart failure. The V-HeFT VA Cooperative Studies Group. Circulation. 1993; 87(6 Suppl):VI17–V23. PMID: 8500235.20. Karantalis V, Hare JM. Use of mesenchymal stem cells for therapy of cardiac disease. Circ Res. 2015; 116:1413–1430. DOI: 10.1161/CIRCRESAHA.116.303614. PMID: 25858066. PMCID: 4429294.
Article21. Higgins JPT, AD , Sterne JAC. Assessing risk of bias in included studies. Higgins JPT, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions. Version 5.1.0 (updated March 2011). Cochrane Collaboration;2011.
Article22. Huedo-Medina TB, Sánchez-Meca J, Marín-Martínez F, Botella J. Assessing heterogeneity in meta-analysis: Q statistic or I2 index? Psychol Methods. 2006; 11:193–206. DOI: 10.1037/1082-989X.11.2.193. PMID: 16784338.23. de Jong R, Houtgraaf JH, Samiei S, Boersma E, Duckers HJ. Intracoronary stem cell infusion after acute myocardial infarction: a meta-analysis and update on clinical trials. Circ Cardiovasc Interv. 2014; 7:156–167. DOI: 10.1161/CIRCINTERVENTIONS.113.001009. PMID: 24668227.24. Lee JW, Lee SH, Youn YJ, Ahn MS, Kim JY, Yoo BS, Yoon J, Kwon W, Hong IS, Lee K, Kwan J, Park KS, Choi D, Jang YS, Hong MK. A randomized, open-label, multicenter trial for the safety and efficacy of adult mesenchymal stem cells after acute myocardial infarction. J Korean Med Sci. 2014; 29:23–31. DOI: 10.3346/jkms.2014.29.1.23. PMID: 24431901. PMCID: 3890472.
Article25. Ascheim DD, Gelijns AC, Goldstein D, Moye LA, Smedira N, Lee S, Klodell CT, Szady A, Parides MK, Jeffries NO, Skerrett D, Taylor DA, Rame JE, Milano C, Rogers JG, Lynch J, Dewey T, Eichhorn E, Sun B, Feldman D, Simari R, O’Gara PT, Taddei-Peters WC, Miller MA, Naka Y, Bagiella E, Rose EA, Woo YJ. Mesenchymal precursor cells as adjunctive therapy in recipients of contemporary left ventricular assist devices. Circulation. 2014; 129:2287–2296. DOI: 10.1161/CIRCULATIONAHA.113.007412. PMID: 24682346. PMCID: 4243683.
Article26. Bartunek J, Behfar A, Dolatabadi D, Vanderheyden M, Ostojic M, Dens J, El Nakadi B, Banovic M, Beleslin B, Vrolix M, Legrand V, Vrints C, Vanoverschelde JL, Crespo-Diaz R, Homsy C, Tendera M, Waldman S, Wijns W, Terzic A. Cardiopoietic stem cell therapy in heart failure: the C-CURE (Cardiopoietic stem Cell therapy in heart failURE) multicenter randomized trial with lineage- specified biologics. J Am Coll Cardiol. 2013; 61:2329–2338. DOI: 10.1016/j.jacc.2013.02.071. PMID: 23583246.27. Gao LR, Pei XT, Ding QA, Chen Y, Zhang NK, Chen HY, Wang ZG, Wang YF, Zhu ZM, Li TC, Liu HL, Tong ZC, Yang Y, Nan X, Guo F, Shen JL, Shen YH, Zhang JJ, Fei YX, Xu HT, Wang LH, Tian HT, Liu DQ, Yang Y. A critical challenge: dosage-related efficacy and acute complication intracoronary injection of autologous bone marrow mesenchymal stem cells in acute myocardial infarction. Int J Cardiol. 2013; 168:3191–3199. DOI: 10.1016/j.ijcard.2013.04.112. PMID: 23651816.
Article28. Gao LR, Chen Y, Zhang NK, Yang XL, Liu HL, Wang ZG, Yan XY, Wang Y, Zhu ZM, Li TC, Wang LH, Chen HY, Chen YD, Huang CL, Qu P, Yao C, Wang B, Chen GH, Wang ZM, Xu ZY, Bai J, Lu D, Shen YH, Guo F, Liu MY, Yang Y, Ding YC, Yang Y, Tian HT, Ding QA, Li LN, Yang XC, Hu X. Intracoronary infusion of Wharton’s jelly-derived mesenchymal stem cells in acute myocardial infarction: double-blind, randomized controlled trial. BMC Med. 2015; 13:162. DOI: 10.1186/s12916-015-0399-z.
Article29. Chen SL, Fang WW, Ye F, Liu YH, Qian J, Shan SJ, Zhang JJ, Chunhua RZ, Liao LM, Lin S, Sun JP. Effect on left ventricular function of intracoronary transplantation of autologous bone marrow mesenchymal stem cell in patients with acute myocardial infarction. Am J Cardiol. 2004; 94:92–95. DOI: 10.1016/j.amjcard.2004.03.034. PMID: 15219514.
Article30. Chen S, Liu Z, Tian N, Zhang J, Yei F, Duan B, Zhu Z, Lin S, Kwan TW. Intracoronary transplantation of autologous bone marrow mesenchymal stem cells for ischemic cardiomyopathy due to isolated chronic occluded left anterior descending artery. J Invasive Cardiol. 2006; 18:552–556. PMID: 17090821.31. Heldman AW, DiFede DL, Fishman JE, Zambrano JP, Trachtenberg BH, Karantalis V, Mushtaq M, Williams AR, Suncion VY, McNiece IK, Ghersin E, Soto V, Lopera G, Miki R, Willens H, Hendel R, Mitrani R, Pattany P, Feigenbaum G, Oskouei B, Byrnes J, Lowery MH, Sierra J, Pujol MV, Delgado C, Gonzalez PJ, Rodriguez JE, Bagno LL, Rouy D, Altman P, Foo CW, da Silva J, Anderson E, Schwarz R, Mendizabal A, Hare JM. Transendocardial mesenchymal stem cells and mononuclear bone marrow cells for ischemic cardiomyopathy: the TAC-HFT randomized trial. JAMA. 2014; 311:62–73. DOI: 10.1001/jama.2013.282909. PMCID: 4111133.
Article32. Mathiasen AB, Qayyum AA, Jørgensen E, Helqvist S, Fischer-Nielsen A, Kofoed KF, Haack-Sørensen M, Ekblond A, Kastrup J. Bone marrow-derived mesenchymal stromal cell treatment in patients with severe ischaemic heart failure: a randomized placebo-controlled trial (MSC-HF trial). Eur Heart J. 2015; 36:1744–1753. DOI: 10.1093/eurheartj/ehv136. PMID: 25926562.
Article33. Bartolucci J, Verdugo FJ, González PL, Larrea RE, Abarzua E, Goset C, Rojo P, Palma I, Lamich R, Pedreros PA, Valdivia G, Lopez VM, Nazzal C, Alcayaga-Miranda F, Cuenca J, Brobeck MJ, Patel AN, Figueroa FE, Khoury M. Safety and efficacy of the intravenous infusion of umbilical cord mesenchymal stem cells in patients with heart failure: a phase 1/2 randomized controlled trial (RIMECARD Trial [Randomized Clinical Trial of Intravenous Infusion Umbilical Cord Mesenchymal Stem Cells on Cardiopathy]). Circ Res. 2017; 121:1192–1204. DOI: 10.1161/CIRCRESAHA.117.310712. PMID: 28974553.
Article34. Teerlink JR, Metra M, Filippatos GS, Davison BA, Bartunek J, Terzic A, Gersh BJ, Povsic TJ, Henry TD, Alexandre B, Homsy C, Edwards C, Seron A, Wijns W, Cotter G. CHART Investigators. Benefit of cardiopoietic mesenchymal stem cell therapy on left ventricular remodelling: results from the Congestive Heart Failure Cardiopoietic Regenerative Therapy (CHART-1) study. Eur J Heart Fail. 2017; 19:1520–1529. DOI: 10.1002/ejhf.898. PMID: 28560782.
Article35. Quevedo HC, Hatzistergos KE, Oskouei BN, Feigenbaum GS, Rodriguez JE, Valdes D, Pattany PM, Zambrano JP, Hu Q, McNiece I, Heldman AW, Hare JM. Allogeneic mesenchymal stem cells restore cardiac function in chronic ischemic cardiomyopathy via trilineage differentiating capacity. Proc Natl Acad Sci U S A. 2009; 106:14022–14027. DOI: 10.1073/pnas.0903201106. PMID: 19666564. PMCID: 2729013.
Article36. Afzal MR, Samanta A, Shah ZI, Jeevanantham V, Abdel-Latif A, Zuba-Surma EK, Dawn B. Adult bone marrow cell therapy for ischemic heart disease: evidence and insights from randomized controlled trials. Circ Res. 2015; 117:558–575. DOI: 10.1161/CIRCRESAHA.114.304792. PMID: 26160853. PMCID: 4553075.
Article37. van der Spoel TI, Gathier WA, Koudstaal S, van Slochteren F, Of Lorkeers SJ, Sluijter JP, Hoefer IE, Steendijk P, Cramer MJ, Doevendans PA, van Belle E, Chamuleau SA. Autologous mesenchymal stem cells show more benefit on systolic function compared to bone marrow mononuclear cells in a porcine model of chronic myocardial infarction. J Cardiovasc Transl Res. 2015; 8:393–403. DOI: 10.1007/s12265-015-9643-3. PMID: 26382088. PMCID: 4623074.
Article38. van der Spoel TI, Jansen of Lorkeers SJ, Agostoni P, van Belle E, Gyöngyösi M, Sluijter JP, Cramer MJ, Doevendans PA, Chamuleau SA. Human relevance of pre-clinical studies in stem cell therapy: systematic review and meta-analysis of large animal models of ischaemic heart disease. Cardiovasc Res. 2011; 91:649–658. DOI: 10.1093/cvr/cvr113. PMID: 21498423.
Article39. Oh H, Bradfute SB, Gallardo TD, Nakamura T, Gaussin V, Mishina Y, Pocius J, Michael LH, Behringer RR, Garry DJ, Entman ML, Schneider MD. Cardiac progenitor cells from adult myocardium: homing, differentiation, and fusion after infarction. Proc Natl Acad Sci U S A. 2003; 100:12313–12318. DOI: 10.1073/pnas.2132126100.
Article40. Kocher AA, Schuster MD, Szabolcs MJ, Takuma S, Burkhoff D, Wang J, Homma S, Edwards NM, Itescu S. Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat Med. 2001; 7:430–436. DOI: 10.1038/86498. PMID: 11283669.
Article41. Gnecchi M, Zhang Z, Ni A, Dzau VJ. Paracrine mechanisms in adult stem cell signaling and therapy. Circ Res. 2008; 103:1204–1219. DOI: 10.1161/CIRCRESAHA.108.176826. PMID: 19028920. PMCID: 2667788.
Article42. Boyle AJ, McNiece IK, Hare JM. Mesenchymal stem cell therapy for cardiac repair. Methods Mol Biol. 2010; 660:65–84. DOI: 10.1007/978-1-60761-705-1_5. PMID: 20680813.
Article43. Piao H, Youn TJ, Kwon JS, Kim YH, Bae JW, Bora-Sohn , Kim DW, Cho MC, Lee MM, Park YB. Effects of bone marrow derived mesenchymal stem cells transplantation in acutely infarcting myocardium. Eur J Heart Fail. 2005; 7:730–738. DOI: 10.1016/j.ejheart.2004.09.019. PMID: 16158494.
Article44. Katritsis DG, Sotiropoulou PA, Karvouni E, Karabinos I, Korovesis S, Perez SA, Voridis EM, Papamichail M. Transcoronary transplantation of autologous mesenchymal stem cells and endothelial progenitors into infarcted human myocardium. Catheter Cardiovasc Interv. 2005; 65:321–329. DOI: 10.1002/ccd.20406. PMID: 15954106.
Article45. Katritsis DG, Sotiropoulou P, Giazitzoglou E, Karvouni E, Papamichail M. Electrophysiological effects of intracoronary transplantation of autologous mesenchymal and endothelial progenitor cells. Europace. 2007; 9:167–171. DOI: 10.1093/europace/eul184. PMID: 17272327.
Article46. Jeong JO, Han JW, Kim JM, Cho HJ, Park C, Lee N, Kim DW, Yoon YS. Malignant tumor formation after transplantation of short-term cultured bone marrow mesenchymal stem cells in experimental myocardial infarction and diabetic neuropathy. Circ Res. 2011; 108:1340–1347. DOI: 10.1161/CIRCRESAHA.110.239848. PMID: 21493893. PMCID: 3109741.
Article47. Miura M, Miura Y, Padilla-Nash HM, Molinolo AA, Fu B, Patel V, Seo BM, Sonoyama W, Zheng JJ, Baker CC, Chen W, Ried T, Shi S. Accumulated chromosomal instability in murine bone marrow mesenchymal stem cells leads to malignant transformation. Stem Cells. 2006; 24:1095–1103. DOI: 10.1634/stemcells.2005-0403.
Article48. Djouad F, Plence P, Bony C, Tropel P, Apparailly F, Sany J, Noël D, Jorgensen C. Immunosuppressive effect of mesenchymal stem cells favors tumor growth in allogeneic animals. Blood. 2003; 102:3837–3844. DOI: 10.1182/blood-2003-04-1193. PMID: 12881305.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Current Concepts in Stem Cell Therapy for Cardiovascular Diseases: What We Know and Don't Know
- Current Trends and Prospect of Cell Therapy using Hematopoietic Stem Cells
- Clinical Trials of Adult Stem Cell Therapy in Patients with Ischemic Stroke
- Mesenchymal Stem Cell Therapy for Intrac table Neonatal Disorders
- Therapeutic Effects of Mesenchymal Stem Cells for Patients with Chronic Liver Diseases: Systematic Review and Meta-analysis