J Korean Soc Echocardiogr.
1996 Jul;4(1):13-21. 10.4250/jkse.1996.4.1.13.
Myocardial Velocity Gradient during Low-dose Dobutamine Stress Echocardiography in the Predietion of viable Myoeardium in the Infarct Zone
- Affiliations
-
- 1Department of Internal Medicine, Seoul National University, College of Medicine, Seoul, Korea.
- KMID: 2410401
- DOI: http://doi.org/10.4250/jkse.1996.4.1.13
Abstract
- BACKGROUND
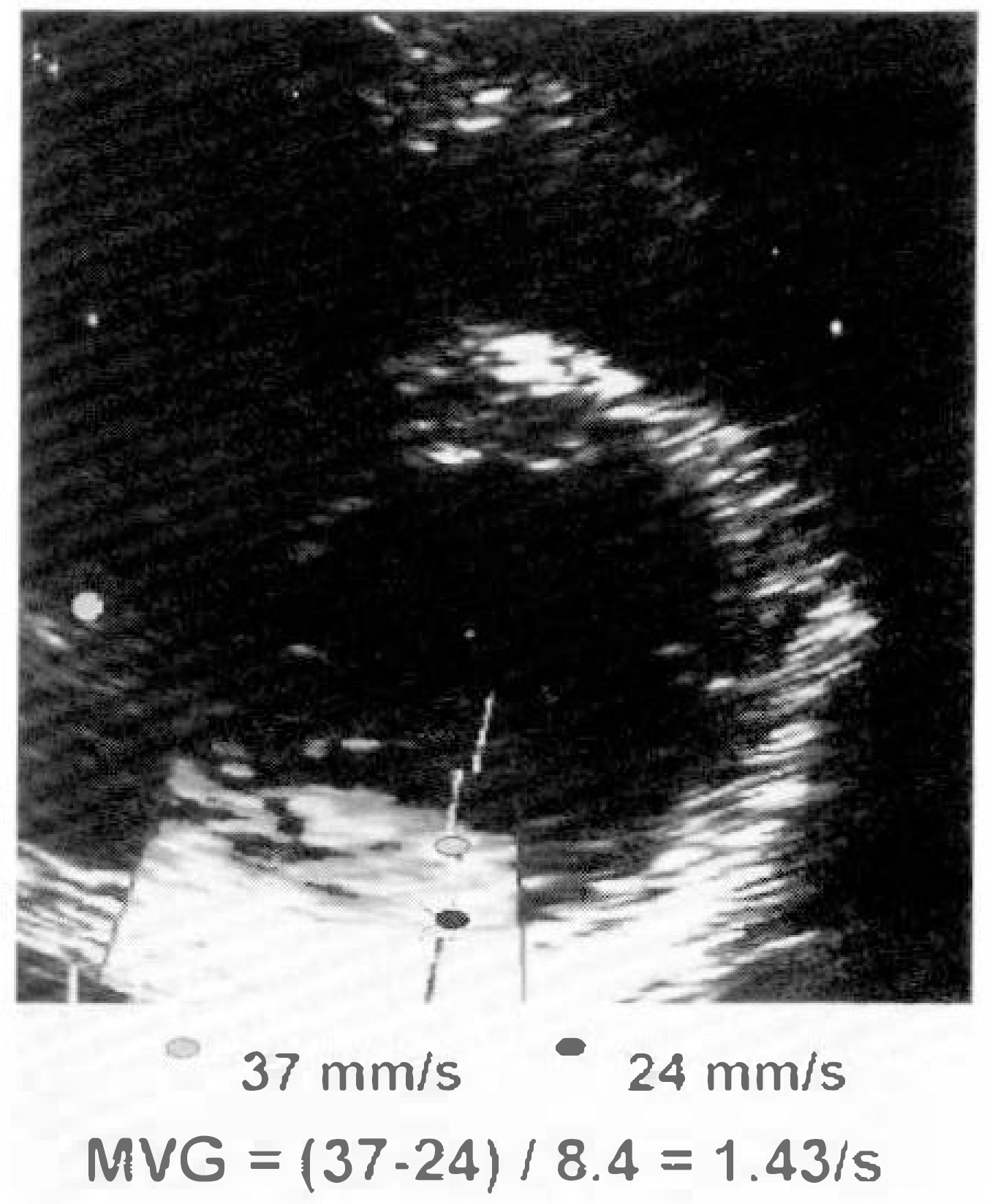
Clinical importance of predicting viability in the infarct zone has been well recognized and low dose dobutamine stress echocardiography has been used in this regards. Several experimental and clinical studies have shown that dobutamine can recruit a contraction reserve in postischemic viable but akinetic segment in patients with myocardial infarction. However, echocardiographic assessment of regional wall motion abnormality is relatively semiquantitative, ambiguous in differentiation between akinetic and hypokinetic segment. With the advent of Doppler tissue imaging, instantaneous wall motion velocity could be measured. This study was performed to evaluate the usefulness of myocardial velocity gradient during low-dose dobutamine stress echocardiography in the prediction of viable myocardium in the infarct zone. METHOD: We assessed left ventricular wall motion in 10 normal subjects, 20 patients with myocardial infarction (anteroseptal in 10, inferior in 10). Myocardial velocity gradient was defined as the difference in the wall motion velocities between the endocardium and the epicardium divided by wall thickness in the parasternal short axis view at the mid-ventricular level.
RESULTS
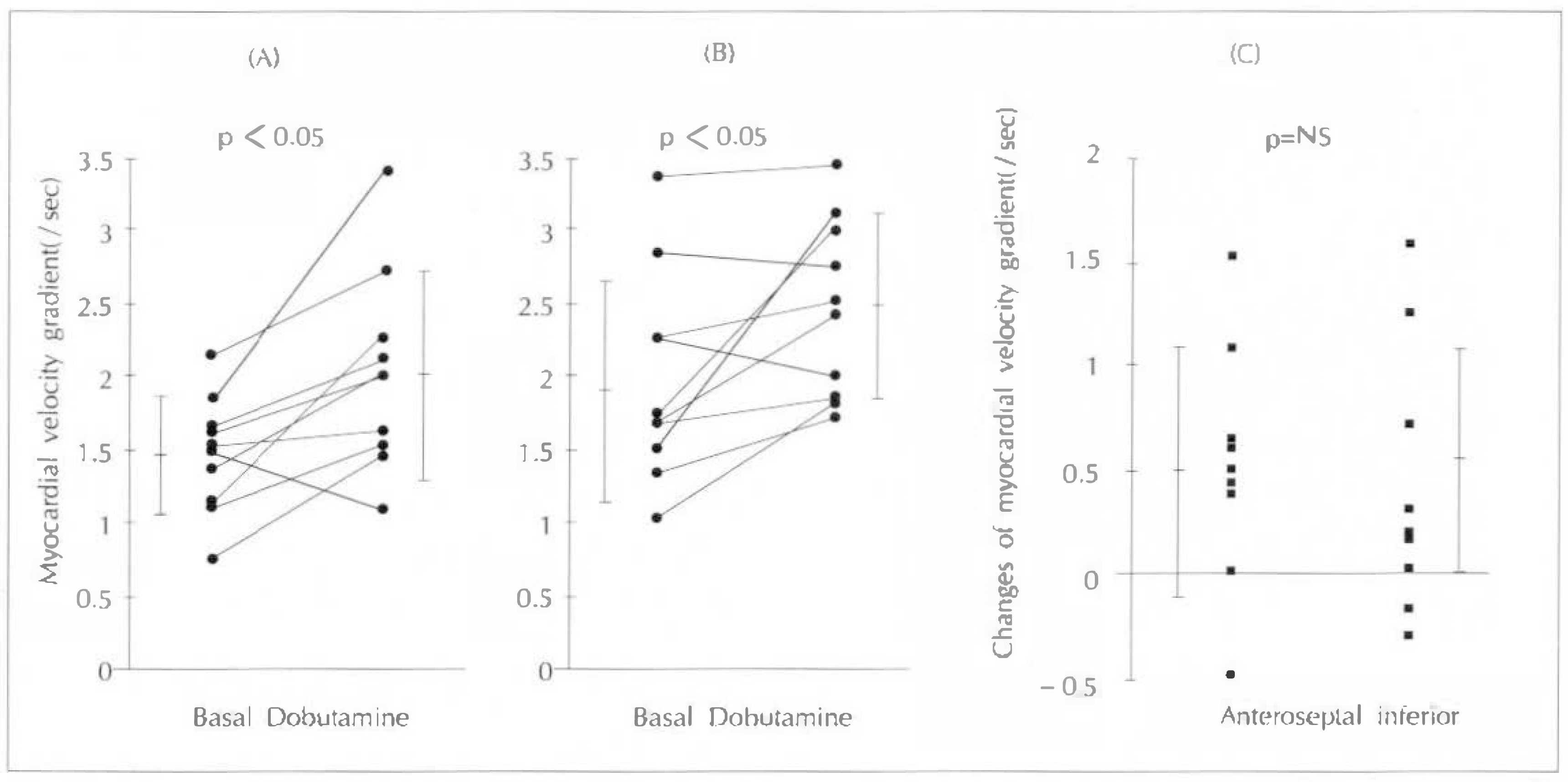
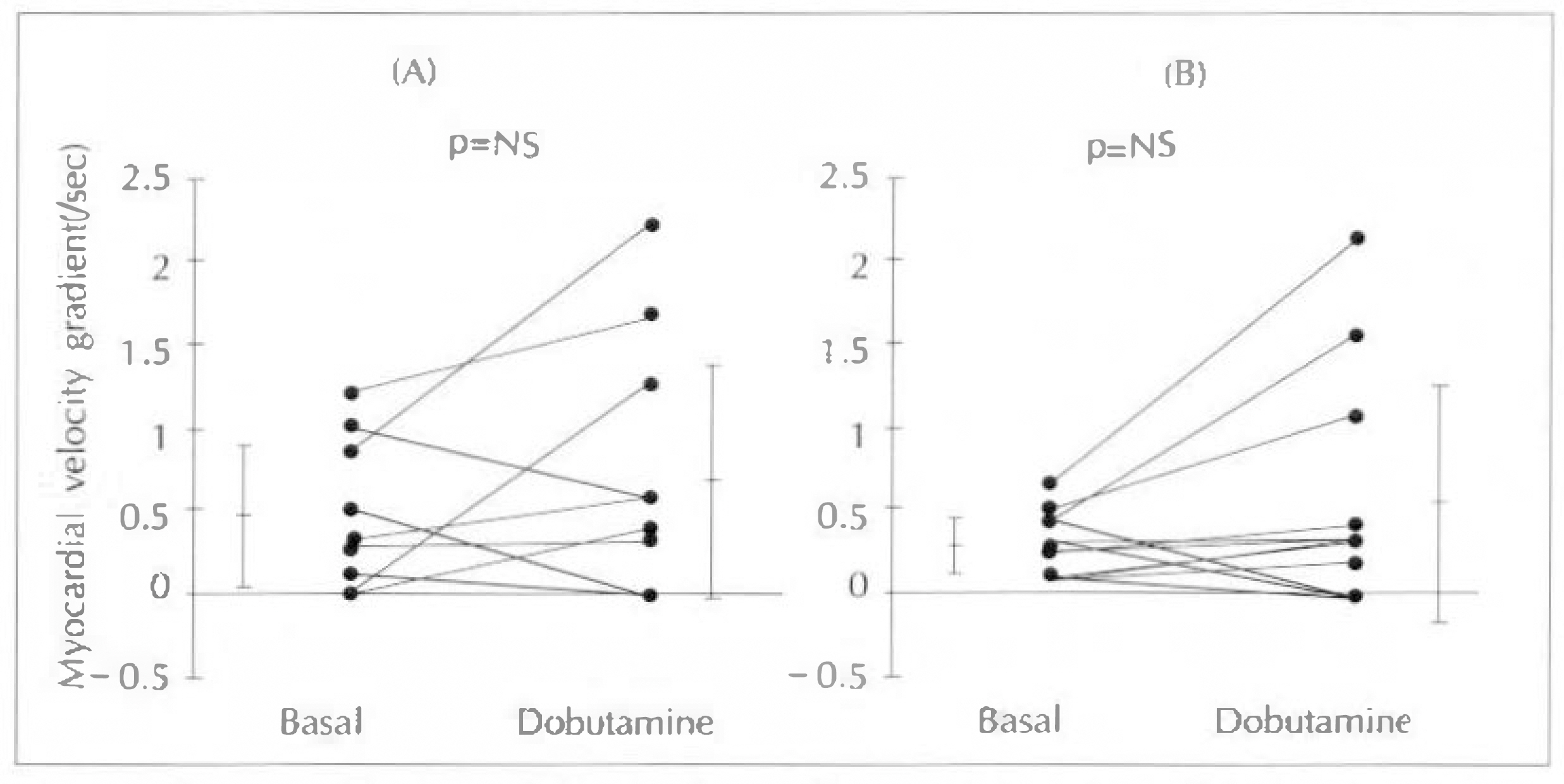
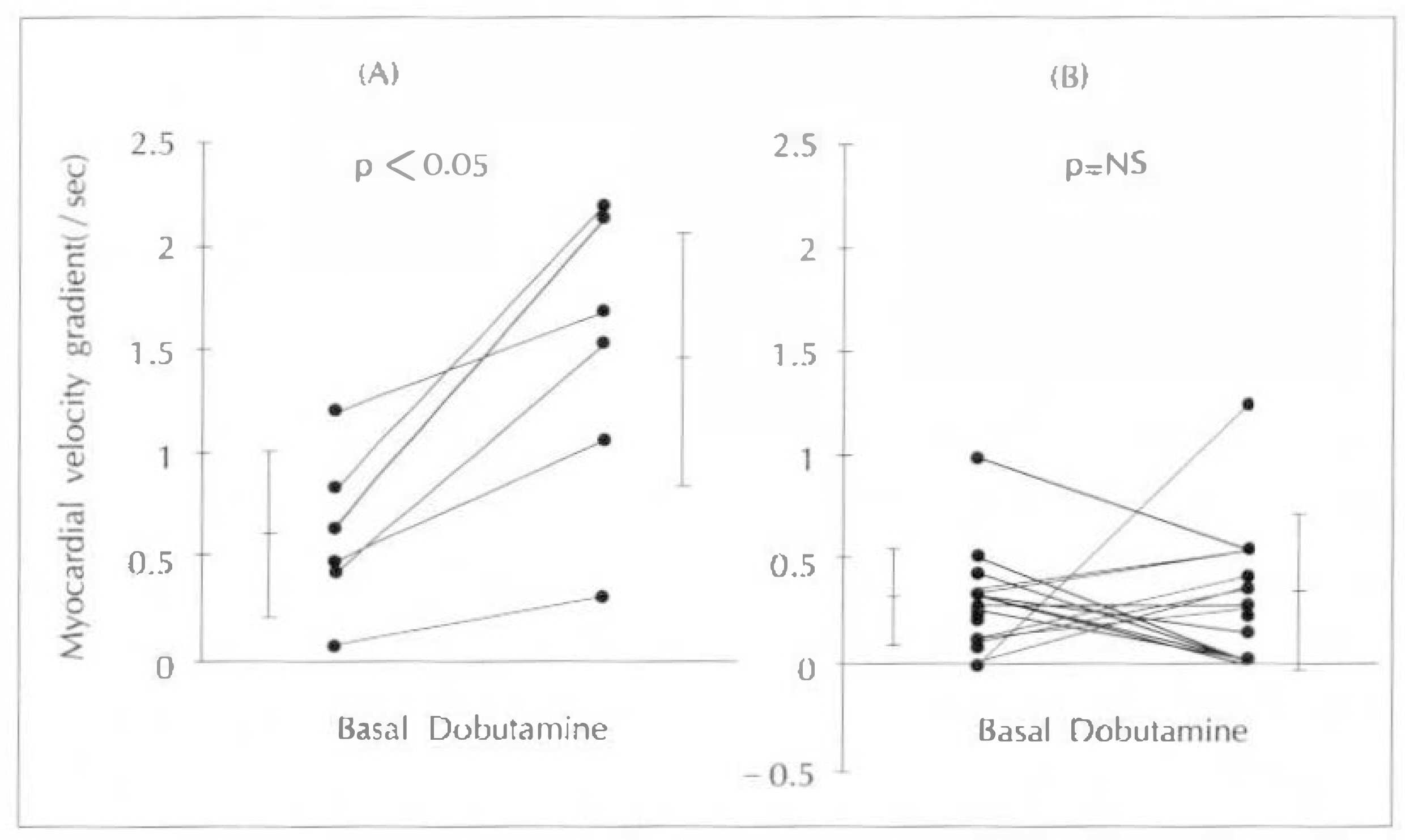
Myocardial velocity gradient in the infarct zones were significantly lower than the corresponding segments in normal subjects (anteroseptal 0.46±0.42/sec vs 1.47±0.39/sec, p < 0.01 ; inferior 0.31±0.18/sec vs 1.97±0.72/sec, p < 0.01). In normal subjects, myocardial velocity gradients during low-dose dobutamine infusion (5µg/kg/min), increased significantly compared to the resting myocardial velocity gradients (anteroseptal 1.47±0.39/sec to 2.04±0.68/sec, p < 0.01 ; inferior 1.97±0.72/sec to 2.48±0.62/sec, p < 0.05). However in the infarct zones, there were no sinificant increases in myocardial velocity gradients during low-dose dobutamine infusion(anteroseptal 0.46±0.42/sec to 0.69±0.76/sec, p=NS : inferior 0.31±0.18/sec to 0.62±0.72, p=NS). When the infarct zones were separated into two groups according to the presence of viable myocardium based on the 24 hour-delayed thallium 201 myocardial SPECT, infarct zones with viable myocardium (n=6, two in anteroseptal and four in inferior infarction)showed significant increase in myocardial velocity gradients during low-dose dobutamine infusion (0.61±0.39/sec to 1.48±0.71/sec, p < 0.05), while infarct zones without viable myocardium (n=14) did not show significant difference (0.29±0.25/sec to 0.30±0.34/sec, p=NS).
CONCLUSIONS
Myocardial velocity gradient could be an objective parameter in the prediction of viable myocardium with low-dose doubutamine stress echocardiography.
MeSH Terms
Figure
Reference
-
References
1). Weyman AE, Franklin TD, Egenes KM, Green D. Correlation between extent of abnormal regional wall motion and myocardial infarct size in chonically infarcted dogs. Circulation. 55(suppl 2):72. 1977.2). Heger J, et al. Cross-sectional echocardiography in acute myocardial infarction: Detection and localization of regional left ventricular asynergy. Circulation. 60:531–538. 1979.
Article3). Nixon JV, Narahara KA, Smitherman TC. Estimation of myocardial involvement in patients with acute myocardial infarction by two-dimensional echocardiography. Circulation. 62:1248–1255. 1980.
Article4). Weiss JV, Bulkley BH, Hutchins GM, Mason SJ. Two-dimensional echocardiographic recognition of myocardial injury in man: Comparison with postmortem studies. Circulation. 63:401–408. 1981.
Article5). Lieberman AN, et al. Two-dimensional echocardiography and infart size: Relationship of regional wall motion and thickening to the extent of myocardial infarction in the dog. Circulation. 63:739–746. 1981.6). Horowitz RS, et al. Immediate diagnosis of acute myocardial infarction by two-dimensional echocardiography. Circulation. 65:323–329. 1982.
Article7). Nieman M, et al. Serial evaluation of myocardial thickening and thinning in acute myocardial experimental infarction: Identification and quantification using two-dimensional echocardiography. Circulation. 66:174–180. 1982.8). Weyman AE, Franklin TD, Hogan RD, et al. Importance of temporal heterogeneity in assessing the contraction abnormalities associated with acute myocardial ischemia. Circulation. 70:102–112. 1984.
Article9). Clayton PD, Jeppson GM, Klausner SC. Should a fixed external reference system be used to analyze left ventrcular wall motion? Circulation. 65:1518–1521. 1982.10). Cigarroa GC, et al. Dobutamine stress echocardiogrphy identifies hibernating myocardium and predicts recovery of left ventricular function after coronary revascularization. Circulation. 88:430–436. 1993.11). Myers JH, Stirling MC, Choy M, Buda AJ, Gallagher KP. Direct measurement of inner and outer wall thickening dynamics with epicardial echocardiography. Circulation. 74:164–172. 1986.
Article12). Sutherland GR, Stewart MJ, Groundstroem KWE, et al. Color Doppler myocardial imaging: A new technique for the assessment of myocardial function. J Am Soc Echocadiogr. 7:441–458. 1994.
Article13). Miyatake K, Yamagishi M, Tanaka N, et al. A new method for the evaluation of left ventriular wall motion by color-coded tissue Doppler imaging: In vitro and in vivo studies. J Am Coll Cardiol. 23:717–724. 1995.14). Uematsu M, Miyatake K, Tanaka N, et al. Myocardial velocity gradient as a new indicator of regional left ventricular contraction: Detection by a two-dimensional tissue Doppler imaging technique. J Am Coll Cardiol. 26:217–223. 1995.
Article15). McDicken WN, Sutherland GR, Moran CM, Gordon LN. Colour Doppler velocity imaging of the myocardium. Ultrasound Med Bial. 18:651–654. 1992.
Article16). Flemming AD, Xia X, McDicken WN, Sutherland GR, Fenn L. Myocardial velocity gradients detected by Doppler imaging. Br J Radiol. 67:679–688. 1994.17). Schiller NB, et al. For the American Society of Echocardiography Committee of Standards, Subcommittee on Quantification of Two-Dimensional Echocardiograms. Recommendation for quantification of the left ventricle by two-dimensional echocardiography. J Am Soc Echodardiogr. 5:358–367. 1989.18). Barilla F, Gheorghiade M, Alam M, et al. Low-dose dobutamine in patients with acute myocardial infarction idendifies viable but not contractile myocardium and predicts the magnitude of improvement in wall motion abnormlities in response to coronary revascularization. Am Heart J. 122:1522–1531. 1991.19). Rahimtoola SH. A perspective on the three large multicenter randomized clinical trials of coronary bypass surgery for chronic stable angina. Circulation. 72(supp IV):V–123-V-135. 1985.20). Ross J Jr. Myocardial perfusion-contraction matching. Circulation. 83:1076–1083. 1991.21). Dilsizian V, Bonow RO. Current diagnostic techiques of assessing myocardial viability in patients with hibernating and stunned myocardium. Circulation. 87:1–20. 1993.22). Nesto RW, Cohn LH, Collins JJ, Wynne J, Holman L, Cohn PF. Inotropic contractile reserve: Useful predictor of increased 5-year survival and improved postoperative left ventricular function in patients with coronary artery disease and reduced ejection fraction. Am J Cardiol. 50:39–44. 1982.23). Popio KA, Gorlin R, Bechtel D, Levine JA. Postextrasystolic potentiation as a predictor of potential myocardial viability: Preoperative analyses compared with studies after coronary bypass surgery. Am J Cardiol. 39:944–951. 1977.
Article24). Bolli R, Zhu WX, Myers ML, Hartley CJ, Roberts R. Beta-adrenergic stimulation reverses postischemic myocardial dysfunction without producing subsequent functional deterioration. Am J Cardiol. 56:946–948. 1985.
Article25). Becker LC, Levine JH, DiPaula AF, Guarnieri T, Aversano T. Reversal of dysfunction in postischemic stunned myocardium by epinephrine and postextrasystolic potentiation. J Am Coll Cardiol. 7:580–589. 1986.
Article26). Malcom J, Arnold O. Braunwald E, Sandor T, Kloner RA. Inotropic stimulation of reperfused myocardium with dopamine: Effects on infarct size and myocardial function. J Am Coll Cardiol. 6:1026–1034. 1985.27). Dilsizian V, Rocco TP, Freedman NMT, Leon MB, Bonow RO. Enhanced detection of ischemic but viable myocardium by the reinjection of thallium after stress-redistribution imaging. N Engl J Med. 323:141–146. 1990.
Article28). Ragosta M, Beller GA, Watson DD, Kaul S, Gimple LW. Quantitative planar rest-redistribution 201 Tl imaging in detection of myocardial viability and prediction of improvement in left ventricular function after coronary bypass surgery in patients with severely depressed left ventricular function. Circulation. 87:1630–1641. 1993.29). Tillish J, Brunken R, Marshall R, Schwaiger M, Mandelkern M, Phelps M, Schelbert HR. Reversibility of cardiac wall-motion abnormalities predicted by positron tomography. N Engl J Med. 314:884–888. 1986.
Article30). Pierard LA, Delandsheere CM, Berthe C, Rigo P, Kulbertus HE. Identification of viable myocardium by echocardiography during dobutamine infusion in patients with myocardial infarction after thrombolytic therapy: Comparison with positron emission tomography. J Am Coll Cardiol. 5:1021–1031. 1990.31). DeFilippi CR, Willett DL, Irani WN, et al. Comparison of myocardial contrast echocardiography and low-dose dobutamine stress echocardiography in predicting recovery of left ventricular function after coronary revascularization in chronic ischemic heart disease. Circulation. 92:2863–2868. 1995.
Article32). Bodenheimer MM, Banka VS, Hermann GA, Trout RG, Pasdar H, Helfant R. Reversible asynergy: Histopathlogic and electrocardiographic correlations in patients with coronary artery disease. Circulation. 53:792–796. 1976.33). Villaneva FS, Glasheen WP, Sklenar J, Kaul S. Characterization of spatial patterns of flow within the reperfused myocardium by myocardial contrast echocardiography: Implications in determining extent of myocardial salvage. Circulation. 88:2596–2606. 1993.
Article34). Mijyatake K, Okamoto M, Kinoshita , et al. Clinical applications of a new type of real-time two-dimensional Doppler flow imaging system. Am J Cardiol. 54:857–868. 1984.35). Afiridi I, Kleiman NS, Raizner AE, Zoghbi WA. Dobutamine echocardiography in myocardial hibernation: Optimal dose and accuracy in predicting recovery of ventricular function after coronary angioplasty. Circulation. 91:663–670. 1995.36). Kaul S. Response of dysfunctional myocardium to dobutamine The eyes see what the mind knows!“. Circulation. 27:1608–1611. 1996.
Article37). Sklenar J, Camarano G, Goodman NC, Ismail S, Kaul S. Contractile versus microvascular reserve for the determination of the extent of myocardial salvage after reperfusion: The effect of residual stenosis. Circulation. In ress.
Article38). Kaul S, Pandian NG, Gillam LD, Newell JB, Okada RD, Weyman AE. Contrast echocardiography in acute myocardial ischemia. III. An in-vivo comparison of the extent of abnormal wall motion with the area at risk for necrosis. J Am Coll Cardiol. 7:383–392. 1986.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Usefulness of Low Dose Dobutamine Stress Echocardiography in Patients with Acute Myocardial Infarction
- The Usefulness of Dobutamine Stress Echocardiography for Evaluation of Viable Myocardium in Hibernating Myocardium
- Diagnosis of Latent Hypertrophic Obstructive Cardiomyopathy with Dobutamine Stress Echocardiography
- Correlations between Coronary Flow Reserve and the Presence of Viable Myocardium after Acute Anterior Myocardial Infarction
- Feasibility Study of Dobutamine Stress Transesophageal Echocardiography