Cancer Res Treat.
2015 Oct;47(4):747-756. 10.4143/crt.2014.099.
The Frequency of and Risk Factors for the Use of Bisphosphonates in the Adjuvant Setting of Primary Breast Cancer in Germany
- Affiliations
-
- 1Institute of Social Medicine and Epidemiology, University of Luebeck, Luebeck, Germany. eva-maria.fick@uksh.de
- 2Institute of Cancer Epidemiology e.V., University of Luebeck, Luebeck, Germany.
- KMID: 2403393
- DOI: http://doi.org/10.4143/crt.2014.099
Abstract
- PURPOSE
The aim of this cross-sectional health care study (use of bisphosphonates in primary tumors of the mammae, EBisMa) is to determine how often bisphosphonate medication is used in patients with non-metastatic primary breast cancer treatment, but who do not suffer from osteoporosis. Furthermore, we describe patients' characteristics and the most frequently used type of bisphosphonate in adjuvant therapy.
MATERIALS AND METHODS
The study population included primary breast cancer patients of four breast centers in northern Germany. Data on bisphosphonate therapy were collected by use of patient questionnaires; clinical data were extracted from the registers. Patients with and without prescribed bisphosphonate adjuvant treatment were tested for statistically significant differences regarding their characteristics.
RESULTS
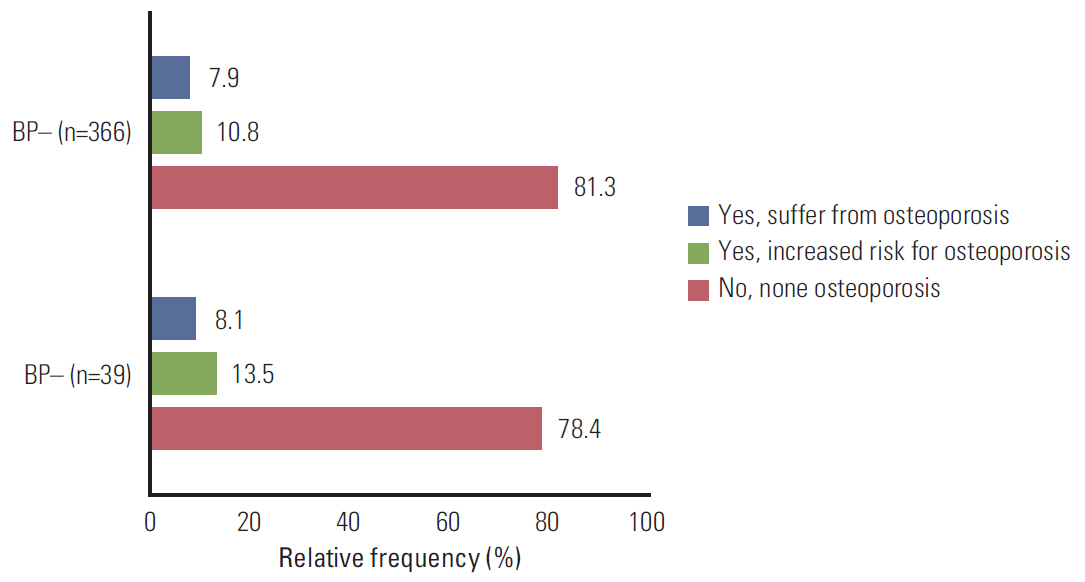
Four hundred seventy-four of 663 contacted patients participated in the study. Thirty-nine out of 474 patients (9.6%) were on adjuvant bisphosphonate therapy. Zoledronic acid was the most frequently reported bisphosphonate used for prevention of bone metastases. Compared to patients who did not report bisphosphonate medication, women who did report bisphosphonate therapy had a significantly higher advanced tumor stage (p < 0.001). Both the T2-T4 stage and N+ stage remained significant predictors in multivariate-adjusted regression models.
CONCLUSION
Bisphosphonates are rarely used in the adjuvant treatment of primary breast cancer. Patients with advanced tumor stage were more likely to use bisphosphonates in the adjuvant treatment of primary breast cancer. Further research is needed to identify patients who may benefit most from adjuvant bisphosphonate treatment.
MeSH Terms
Figure
Reference
-
References
1. Kaatsch P, Spix C, Hentschel S, Katalinic A, Luttmann S, Stegmaier C. Krebs in Deutschland 2009/2010. Berlin: Robert Koch-Institut;2013.2. Russell RG. Bisphosphonates: the first 40 years. Bone. 2011; 49:2–19.3. Gnant M, Mlineritsch B, Stoeger H, Luschin-Ebengreuth G, Heck D, Menzel C, et al. Adjuvant endocrine therapy plus zoledronic acid in premenopausal women with early-stage breast cancer: 62-month follow-up from the ABCSG-12 randomised trial. Lancet Oncol. 2011; 12:631–41.
Article4. Coleman RE, Marshall H, Cameron D, Dodwell D, Burkinshaw R, Keane M, et al. Breast-cancer adjuvant therapy with zoledronic acid. N Engl J Med. 2011; 365:1396–405.
Article5. Diel IJ, Jaschke A, Solomayer EF, Gollan C, Bastert G, Sohn C, et al. Adjuvant oral clodronate improves the overall survival of primary breast cancer patients with micrometastases to the bone marrow: a long-term follow-up. Ann Oncol. 2008; 19:2007–11.6. Diagnostik und therapie primärer und metastasierter mammakarzinome: bisphosphonate [Internet]. Arbeitsgemeinschaft Gynäkologische Onkologie e.V. 2009. [cited 2014 Dec 1]. Available from:http://www.ago-online.de/fileadmin/downloads/leitlinien/mamma/februar2009/g_mamma_09_1_0_d_26_bisphosphonates.pdf.7. Diagnostik und therapie primärer und metastasierter mammakarzinome: osteoonkologie und knochengesundheit [Internet]. Arbeitsgemeinschaft Gynäkologische Onkologie AGO e.V. 2014. [cited 2014 Dec 1]. Available from:http://www.ago-online.de/fileadmin/downloads/leitlinien/mamma/maerz2014/de/2014D_21_Osteoonkologie_und_Knochengesundheit.pdf.8. Paterson AH, Anderson SJ, Lembersky BC, Fehrenbacher L, Falkson CI, King KM, et al. Oral clodronate for adjuvant treatment of operable breast cancer (National Surgical Adjuvant Breast and Bowel Project protocol B-34): a multicentre, placebo-controlled, randomised trial. Lancet Oncol. 2012; 13:734–42.
Article9. Powles T, Paterson S, Kanis JA, McCloskey E, Ashley S, Tidy A, et al. Randomized, placebo-controlled trial of clodronate in patients with primary operable breast cancer. J Clin Oncol. 2002; 20:3219–24.
Article10. Saarto T, Vehmanen L, Virkkunen P, Blomqvist C. Ten-year follow-up of a randomized controlled trial of adjuvant clodronate treatment in node-positive breast cancer patients. Acta Oncol. 2004; 43:650–6.
Article11. Coleman R, de Boer R, Eidtmann H, Llombart A, Davidson N, Neven P, et al. Zoledronic acid (zoledronate) for postmenopausal women with early breast cancer receiving adjuvant letrozole (ZO-FAST study): final 60-month results. Ann Oncol. 2013; 24:398–405.
Article12. von Minckwitz G, Mobus V, Schneeweiss A, Huober J, Thomssen C, Untch M, et al. German adjuvant intergroup node-positive study: a phase III trial to compare oral ibandronate versus observation in patients with high-risk early breast cancer. J Clin Oncol. 2013; 31:3531–9.
Article13. Kokufu I, Kohno N, Yamamoto M, Takao S. Adjuvant pamidronate therapy prevents the development of bone metastases in breast cancer patients with four or more positive nodes. Oncol Lett. 2010; 1:247–52.
Article14. Kristensen B, Ejlertsen B, Mouridsen HT, Jensen MB, Andersen J, Bjerregaard B, et al. Bisphosphonate treatment in primary breast cancer: results from a randomised comparison of oral pamidronate versus no pamidronate in patients with primary breast cancer. Acta Oncol. 2008; 47:740–6.
Article15. Diagnostik und therapie primärer und metastasierter mammakarzinome: bisphosphonate und RANKL-antikörper [Internet] Arbeitsgemeinschaft Gynäkologische Onkologie AGO e.V. 2012. [cited 2014 Dec 1]. Available from:http://www.ago-online.de/fileadmin/downloads/leitlinien/mamma/maerz2012/16_2012D_Bisphosphonate_und_der_RANKL-Antikoerper_Denosumab.pdf.16. Fick EM, Anzeneder T, Katalinic A, Waldmann A. Bisphosphonates and their role in therapy for breast cancer: results from the PATH Biobank. Geburtshilfe Frauenheilkd. 2013; 73:412–21.17. Llombart A, Frassoldati A, Paija O, Sleeboom HP, Jerusalem G, Mebis J, et al. Immediate administration of zoledronic acid reduces aromatase inhibitor-associated bone loss in postmenopausal women with early breast cancer: 12-month analysis of the E-ZO-FAST trial. Clin Breast Cancer. 2012; 12:40–8.
Article18. Bundred NJ, Campbell ID, Davidson N, DeBoer RH, idtmann H, Monnier A, et al. Effective inhibition of aromatase inhibitor-associated bone loss by zoledronic acid in postmenopausal women with early breast cancer receiving adjuvant letrozole: ZO-FAST Study results. Cancer. 2008; 112:1001–10.19. Winter MC, Coleman RE. Bisphosphonates in the adjuvant treatment of breast cancer. Clin Oncol (R Coll Radiol). 2013; 25:135–45.
Article20. Hershman DL, McMahon DJ, Crew KD, Cremers S, Irani D, Cucchiara G, et al. Zoledronic acid prevents bone loss in premenopausal women undergoing adjuvant chemotherapy for early-stage breast cancer. J Clin Oncol. 2008; 26:4739–45.
Article21. Ibrahim T, Liverani C, Mercatali L, Sacanna E, Zanoni M, Fabbri F, et al. Cisplatin in combination with zoledronic acid: a synergistic effect in triple-negative breast cancer cell lines. Int J Oncol. 2013; 42:1263–70.
Article22. Neville-Webbe HL, Rostami-Hodjegan A, Evans CA, Coleman RE, Holen I. Sequence-and schedule-dependent enhancement of zoledronic acid induced apoptosis by doxorubicin in breast and prostate cancer cells. Int J Cancer. 2005; 113:364–71.23. Coleman RE, Winter MC, Cameron D, Bell R, Dodwell D, Keane MM, et al. The effects of adding zoledronic acid to neoadjuvant chemotherapy on tumour response: exploratory evidence for direct anti-tumour activity in breast cancer. Br J Cancer. 2010; 102:1099–105.
Article24. Ritterhoff N. Wie gut kennen patienten ihre krankheit und behandlung? Ein vergleich von patientenangaben, arz-tangaben und registerdaten in der onkologischen versorgung. Lübeck: Medizinische Fakultät, Institut für Krebsepidemiologie e.V., Universität zu Lübeck;2010.25. Waldmann A, Dreckschmidt J, Pritzkuleit R, Katalinic A. Testretest reliability of the OVIS Questionnaire: an instrument to evaluate oncological care from a patient's point of view. Gesundheitswesen. 2010; 72:707–13.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The evidence for adjuvant taxanes in early breast cancer
- Chemotherapy in Breast Cancer
- Adjuvant Chemotherapy for Breast Cancer
- Bone-modifying agents for bone metastasis in patients with breast cancer
- Interactions of Family History of Breast Cancer with Radiotherapy in Relation to the Risk of Breast Cancer Recurrence