J Korean Soc Transplant.
2017 Dec;31(4):200-206. 10.4285/jkstn.2017.31.4.200.
Predictors of Avascular Necrosis after Kidney Transplantation
- Affiliations
-
- 1Department of Surgery, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. djhan@amc.seoul.kr
- 2Division of Nephrology, Department of Internal Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- KMID: 2401571
- DOI: http://doi.org/10.4285/jkstn.2017.31.4.200
Abstract
- BACKGROUND
Risk factors for bone avascular necrosis (AVN), a common late complication after kidney transplantation (KT), are not well known.
METHODS
Patients that underwent living-donor KT at Asan Medical Center between January 2009 and July 2016 were included in this retrospective study to determine the incidence and risk factors for AVN after KT.
RESULTS
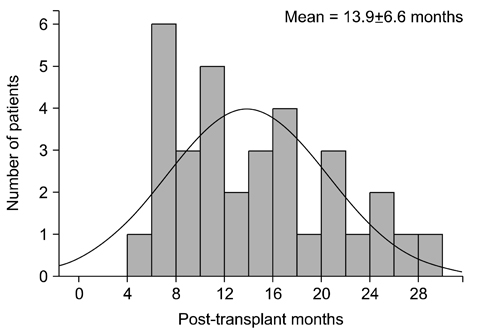
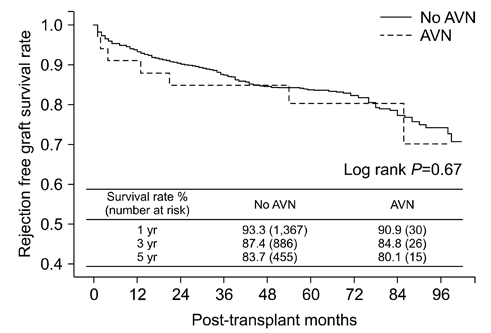
Among 1,570 patients that underwent living-donor KT, 33 (2.1%) developed AVN during a mean follow-up of 49.8±25.0months. Additionally, AVN was diagnosed at a mean of 13.9±6.6 months after KT. The mean cumulative corticosteroid dose during the last follow-up in patients without AVN (9,108±3,400 mg) was higher than that that in patients with AVN (4,483±1,114 mg) until AVN development (P < 0.01). More patients among those with AVN (n=4, 12.1%) underwent steroid pulse treatment because of biopsy-proven rejections during the first 6 months after KT than patients without AVN (n=68, 4.4%; P=0.04). Female (hazard ratio [HR], 2.29; P=0.04) and steroid pulse treatment during the first 6 months (HR, 2.31; P=0.02) were significant AVN risk factors as revealed by the Cox proportional multivariate analysis. However, no significant differences in rejection-free graft survival rates were observed between the two groups (P=0.67).
CONCLUSIONS
Steroid pulse treatment within 6 months of KT and being female were independent risk factors for AVN development.
MeSH Terms
Figure
Reference
-
1. Kubo T, Yamazoe S, Sugano N, Fujioka M, Naruse S, Yoshimura N, et al. Initial MRI findings of non-traumatic osteonecrosis of the femoral head in renal allograft recipients. Magn Reson Imaging. 1997; 15:1017–1023.
Article2. McAvoy S, Baker KS, Mulrooney D, Blaes A, Arora M, Burns LJ, et al. Corticosteroid dose as a risk factor for avascular necrosis of the bone after hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2010; 16:1231–1236.
Article3. Saito M, Ueshima K, Fujioka M, Ishida M, Goto T, Arai Y, et al. Corticosteroid administration within 2 weeks after renal transplantation affects the incidence of femoral head osteonecrosis. Acta Orthop. 2014; 85:266–270.
Article4. Karapinar L, Gurkan A, Kacar S, Polat O. Post-transplant femoral head avascular necrosis: a selective investigation with MRI. Ann Transplant. 2007; 12:27–31.5. Chan KL, Mok CC. Glucocorticoid-induced avascular bone necrosis: diagnosis and management. Open Orthop J. 2012; 6:449–457.
Article6. Drescher W, Schlieper G, Floege J, Eitner F. Steroid-related osteonecrosis: an update. Nephrol Dial Transplant. 2011; 26:2728–2731.7. Hirota Y, Hirohata T, Fukuda K, Mori M, Yanagawa H, Ohno Y, et al. Association of alcohol intake, cigarette smoking, and occupational status with the risk of idiopathic osteonecrosis of the femoral head. Am J Epidemiol. 1993; 137:530–538.
Article8. Zonana-Nacach A, Barr SG, Magder LS, Petri M. Damage in systemic lupus erythematosus and its association with corticosteroids. Arthritis Rheum. 2000; 43:1801–1808.
Article9. Shah KN, Racine J, Jones LC, Aaron RK. Pathophysiology and risk factors for osteonecrosis. Curr Rev Musculoskelet Med. 2015; 8:201–209.
Article10. Orban HB, Cristescu V, Dragusanu M. Avascular necrosis of the femoral head. Maedica (Buchar). 2009; 4:26–34.11. Hernigou P. Avascular necrosis of head of femur. Indian J Orthop. 2009; 43:1–2.
Article12. Paydas S, Balal M, Demir E, Sertdemir Y, Erken U. Avascular osteonecrosis and accompanying anemia, leucocytosis, and decreased bone mineral density in renal transplant recipients. Transplant Proc. 2011; 43:863–866.
Article13. Abbott KC, Koff J, Bohen EM, Oglesby RJ, Agodoa LY, Lentine KL, et al. Maintenance immunosuppression use and the associated risk of avascular necrosis after kidney transplantation in the United States. Transplantation. 2005; 79:330–336.
Article14. Hedri H, Cherif M, Zouaghi K, Abderrahim E, Goucha R, Ben Hamida F, et al. Avascular osteonecrosis after renal transplantation. Transplant Proc. 2007; 39:1036–1038.
Article15. Schulte CM, Beelen DW. Avascular osteonecrosis after allogeneic hematopoietic stem-cell transplantation: diagnosis and gender matter. Transplantation. 2004; 78:1055–1063.
Article16. Campbell S, Sun CL, Kurian S, Francisco L, Carter A, Kulkarni S, et al. Predictors of avascular necrosis of bone in long-term survivors of hematopoietic cell transplantation. Cancer. 2009; 115:4127–4135.
Article17. Haas M, Sis B, Racusen LC, Solez K, Glotz D, Colvin RB, et al. Banff 2013 meeting report: inclusion of c4d-negative antibody-mediated rejection and antibody-associated arterial lesions. Am J Transplant. 2014; 14:272–283.
Article18. Barbhaiya M, Dong Y, Sparks JA, Losina E, Costenbader KH, Katz JN. Administrative algorithms to identify avascular necrosis of bone among patients undergoing upper or lower extremity magnetic resonance imaging: a validation study. BMC Musculoskelet Disord. 2017; 18:268.
Article19. Maus U, Roth A, Tingart M, Rader C, Jager M, Noth U, et al. S3 Guideline. Part 3: non-traumatic avascular necrosis in adults: surgical treatment of atraumatic avascular femoral head necrosis in adults. Z Orthop Unfall. 2015; 153:498–507.20. Shibatani M, Fujioka M, Arai Y, Takahashi K, Ueshima K, Okamoto M, et al. Degree of corticosteroid treatment within the first 2 months of renal transplantation has a strong influence on the incidence of osteonecrosis of the femoral head. Acta Orthop. 2008; 79:631–636.
Article21. Weinstein RS. Glucocorticoid-induced osteonecrosis. Endocrine. 2012; 41:183–190.
Article22. Serrano OK, Kandaswamy R, Gillingham K, Chinnakotla S, Dunn TB, Finger E, et al. Rapid discontinuation of prednisone in kidney transplant recipients: 15-year outcomes from the University of Minnesota. Transplantation. 2017; 101:2590–2598.23. Lausten GS, Lemser T, Jensen PK, Egfjord M. Necrosis of the femoral head after kidney transplantation. Clin Transplant. 1998; 12:572–574.24. Dilisio MF. Osteonecrosis following short-term, low-dose oral corticosteroids: a population-based study of 24 million patients. Orthopedics. 2014; 37:e631–e636.
Article25. Sakai T, Sugano N, Kokado Y, Takahara S, Ohzono K, Yoshikawa H. Tacrolimus may be associated with lower osteonecrosis rates after renal transplantation. Clin Orthop Relat Res. 2003; (415):163–170.
Article26. Kenzora JE. Ischemic necrosis of femoral head. Part I. Accumulative cell stress: a hypothesis for the etiology of idiopathic osteonecrosis. Instr Course Lect. 1983; 32:242–252.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Avascuar Necrosis of Bone after Kidney Transplantation
- Avascular Necrosis After the Fracture of the Neck of the Talus
- Femoral Head and Neck Fractures developed in Avascular Necrosis of the Femoral Head
- Acute Retinal Necrosis Syndrome in an Immunosuppressed Kidney Transplant Patient
- Prednisolone Influence on Avascular Osteonecrosis after Renal Transplantation