J Clin Neurol.
2014 Jan;10(1):24-31.
Clinical and Pathological Features of Korean Patients with DNM2-Related Centronuclear Myopathy
- Affiliations
-
- 1Department of Neurology, Pusan National University School of Medicine, Busan, Korea. dskim@pusan.ac.kr
- 2Medical Research Institute, Pusan National University Hospital, Busan, Korea.
- 3Department of Neurology, Gangnam Severance Hospital, Yonsei University College of Medicine, Seoul, Korea.
- 4Department of Neurology, Inje University School of Medicine, Busan, Korea.
- 5Department of Pathology, Pusan National University School of Medicine, Busan, Korea.
- 6Research Institute for Convergence of Biomedical Research and Technology, Pusan National University Yangsan Hospital, Yangsan, Korea.
Abstract
- BACKGROUND AND PURPOSE
Centronuclear myopathy (CNM) is characterized by the presence of central nuclei within a large number of muscle fibers. Mutations of the dynamin 2 gene (DNM2) are common causes of autosomal dominant or sporadic CNM. The aim of this study was to characterize the clinical and pathological features of CNM relative to the presence of DNM2 mutations.
METHODS
Six patients with clinical and pathological features of CNM were recruited. Detailed clinical and pathological findings were analyzed according to the presence of DNM2 mutations.
RESULTS
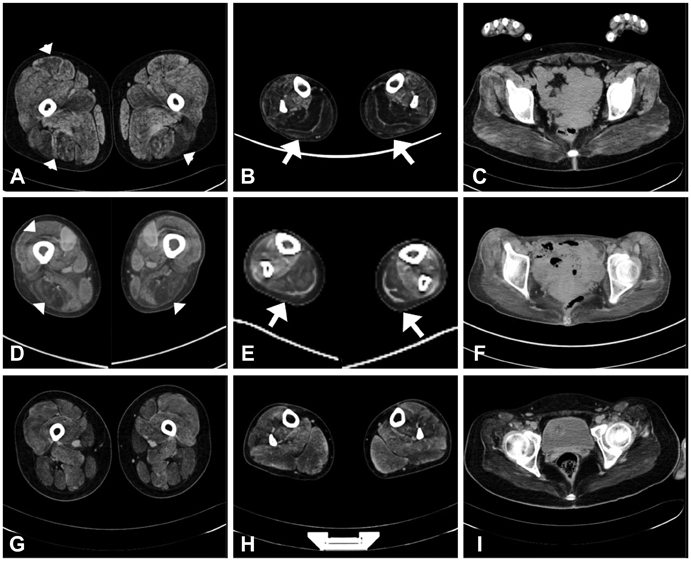
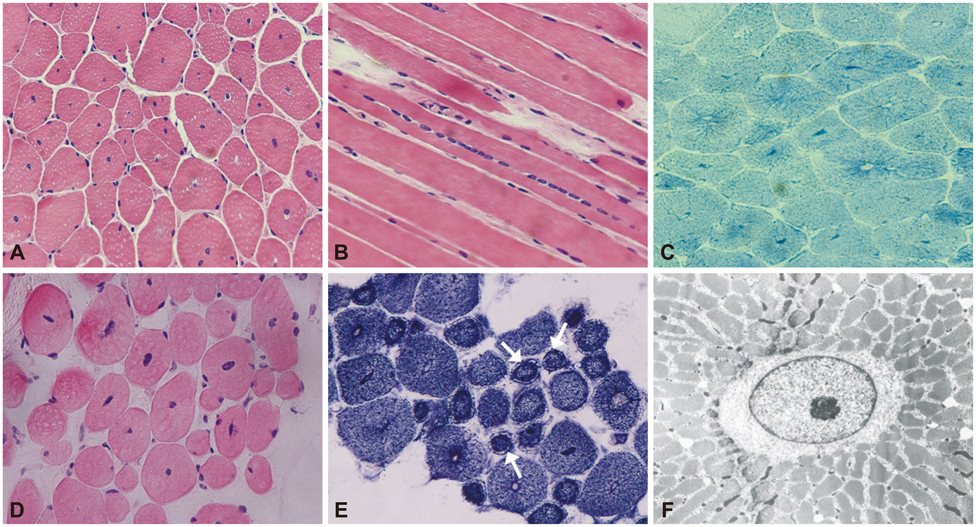
We detected DNM2 mutations in four of the six sporadic CNM patients, and identified the following distinct clinical and pathological features in those patients with DNM2 mutations: preferential involvement of the distal lower limbs, typical nuclear centralization, and radially distributed sarcoplasmic strands in muscle pathology. In contrast, those without DNM2 mutations exhibited rather diffuse muscular involvement, and nuclear internalization and myofibrillar disorganization were more pronounced features of their muscle pathology.
CONCLUSIONS
These findings suggest the presence of specific features in Korean CNM patients. A detailed clinical and pathological examination of CNM patients would be helpful for molecular genetic analyses of this condition.
Keyword
MeSH Terms
Figure
Reference
-
1. Spiro AJ, Shy GM, Gonatas NK. Myotubular myopathy. Persistence of fetal muscle in an adolescent boy. Arch Neurol. 1966; 14:1–14.2. Laporte J, Hu LJ, Kretz C, Mandel JL, Kioschis P, Coy JF, et al. A gene mutated in X-linked myotubular myopathy defines a new putative tyrosine phosphatase family conserved in yeast. Nat Genet. 1996; 13:175–182.
Article3. Bitoun M, Maugenre S, Jeannet PY, Lacène E, Ferrer X, Laforêt P, et al. Mutations in dynamin 2 cause dominant centronuclear myopathy. Nat Genet. 2005; 37:1207–1209.
Article4. Nicot AS, Toussaint A, Tosch V, Kretz C, Wallgren-Pettersson C, Iwarsson E, et al. Mutations in amphiphysin 2 (BIN1) disrupt interaction with dynamin 2 and cause autosomal recessive centronuclear myopathy. Nat Genet. 2007; 39:1134–1139.
Article5. Wilmshurst JM, Lillis S, Zhou H, Pillay K, Henderson H, Kress W, et al. RYR1 mutations are a common cause of congenital myopathies with central nuclei. Ann Neurol. 2010; 68:717–726.
Article6. Züchner S, Noureddine M, Kennerson M, Verhoeven K, Claeys K, De Jonghe P, et al. Mutations in the pleckstrin homology domain of dynamin 2 cause dominant intermediate Charcot-Marie-Tooth disease. Nat Genet. 2005; 37:289–294.
Article7. Jeannet PY, Bassez G, Eymard B, Laforêt P, Urtizberea JA, Rouche A, et al. Clinical and histologic findings in autosomal centronuclear myopathy. Neurology. 2004; 62:1484–1490.
Article8. Melberg A, Kretz C, Kalimo H, Wallgren-Pettersson C, Toussaint A, Böhm J, et al. Adult course in dynamin 2 dominant centronuclear myopathy with neonatal onset. Neuromuscul Disord. 2010; 20:53–56.
Article9. Bitoun M, Durieux AC, Prudhon B, Bevilacqua JA, Herledan A, Sakanyan V, et al. Dynamin 2 mutations associated with human diseases impair clathrin-mediated receptor endocytosis. Hum Mutat. 2009; 30:1419–1427.
Article10. Susman RD, Quijano-Roy S, Yang N, Webster R, Clarke NF, Dowling J, et al. Expanding the clinical, pathological and MRI phenotype of DNM2-related centronuclear myopathy. Neuromuscul Disord. 2010; 20:229–237.
Article11. Böhm J, Biancalana V, Dechene ET, Bitoun M, Pierson CR, Schaefer E, et al. Mutation spectrum in the large GTPase dynamin 2, and genotype-phenotype correlation in autosomal dominant centronuclear myopathy. Hum Mutat. 2012; 33:949–959.
Article12. Koutsopoulos OS, Koch C, Tosch V, Böhm J, North KN, Laporte J. Mild functional differences of dynamin 2 mutations associated to centronuclear myopathy and Charcot-Marie Tooth peripheral neuropathy. PLoS One. 2011; 6:e27498.13. Fischer D, Herasse M, Bitoun M, Barragán-Campos HM, Chiras J, Laforêt P, et al. Characterization of the muscle involvement in dynamin 2-related centronuclear myopathy. Brain. 2006; 129:1463–1469.
Article14. Catteruccia M, Fattori F, Codemo V, Ruggiero L, Maggi L, Tasca G, et al. Centronuclear myopathy related to dynamin 2 mutations: clinical, morphological, muscle imaging and genetic features of an Italian cohort. Neuromuscul Disord. 2013; 23:229–238.
Article15. Bitoun M, Bevilacqua JA, Prudhon B, Maugenre S, Taratuto AL, Monges S, et al. Dynamin 2 mutations cause sporadic centronuclear myopathy with neonatal onset. Ann Neurol. 2007; 62:666–670.
Article16. Hanisch F, Müller T, Dietz A, Bitoun M, Kress W, Weis J, et al. Phenotype variability and histopathological findings in centronuclear myopathy due to DNM2 mutations. J Neurol. 2011; 258:1085–1090.
Article17. Romero NB. Centronuclear myopathies: a widening concept. Neuromuscul Disord. 2010; 20:223–228.
Article18. Bevilacqua JA, Bitoun M, Biancalana V, Oldfors A, Stoltenburg G, Claeys KG, et al. "Necklace" fibers, a new histological marker of late-onset MTM1-related centronuclear myopathy. Acta Neuropathol. 2009; 117:283–291.
Article19. Mori-Yoshimura M, Okuma A, Oya Y, Fujimura-Kiyono C, Nakajima H, Matsuura K, et al. Clinicopathological features of centronuclear myopathy in Japanese populations harboring mutations in dynamin 2. Clin Neurol Neurosurg. 2012; 114:678–683.
Article