Ann Lab Med.
2018 Jan;38(1):9-16. 10.3343/alm.2018.38.1.9.
Serum Macrophage Migration Inhibitory Factor as a Biomarker of Active Pulmonary Tuberculosis
- Affiliations
-
- 1Department of Clinical Laboratory, Wuxi Huishan People's Hospital, Wuxi, Jiangsu, China. kuaishougang@163.com
- 2Department of Clinical Laboratory, The Fifth People's Hospital of Wuxi, Affiliated to Jiangnan University, Wuxi, Jiangsu, China.
- 3Department of Respiratory Medicine, The Fifth People's Hospital of Wuxi, Affiliated to Jiangnan University, Wuxi, Jiangsu, China.
- KMID: 2397596
- DOI: http://doi.org/10.3343/alm.2018.38.1.9
Abstract
- BACKGROUND
Macrophage migration inhibitory factor (MIF), a pro-inflammatory cytokine with chemokine-like functions, has been shown to play a central role in several acute and chronic inflammatory diseases. However, limited information is available regarding the use of MIF as an inflammatory pathway marker in patients with tuberculosis. This study aimed to investigate the association of MIF with IFN-γ and TNF-α in active pulmonary tuberculosis (APTB) following anti-tuberculosis treatment.
METHODS
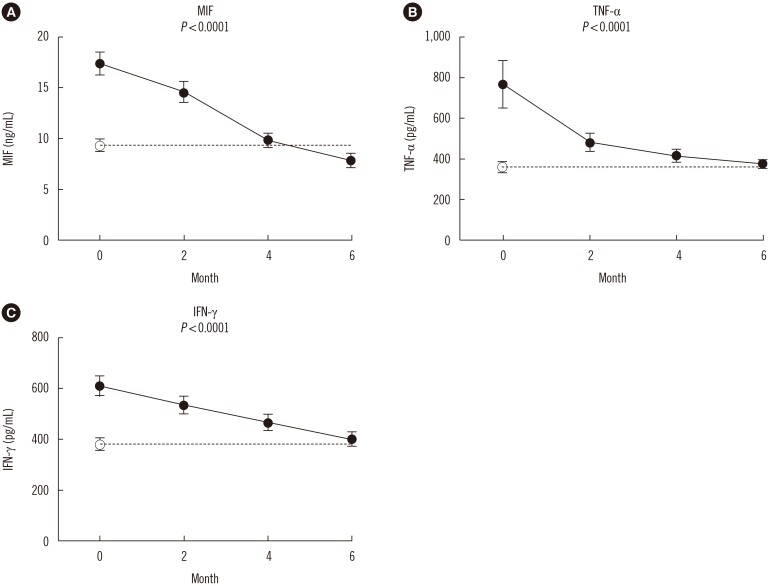
The MIF, TNF-α, and IFN-γ serum levels were determined in 47 patients with APTB by cytokine-specific ELISA at four phases: prior to anti-tuberculosis drug treatment (baseline), and following 2, 4, and 6 months of treatment. In addition, we measured the MIF, TNF-α, and IFN-γ serum levels in 50 health controls.
RESULTS
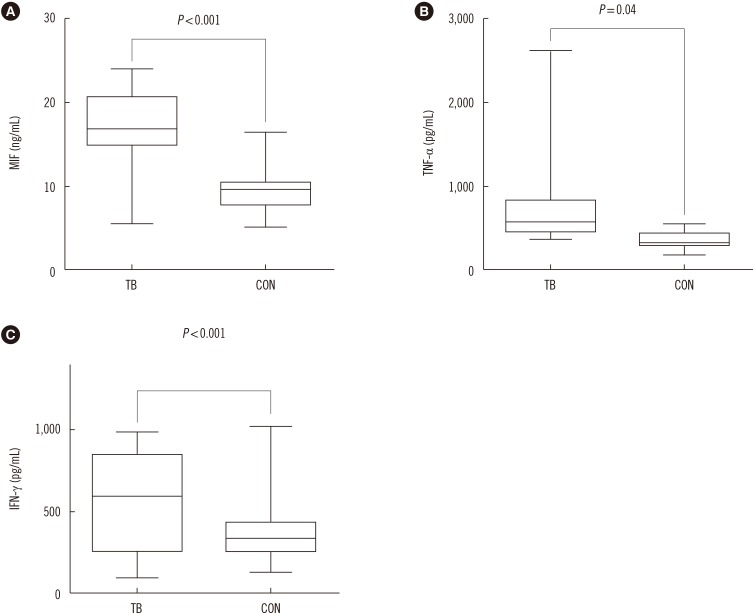
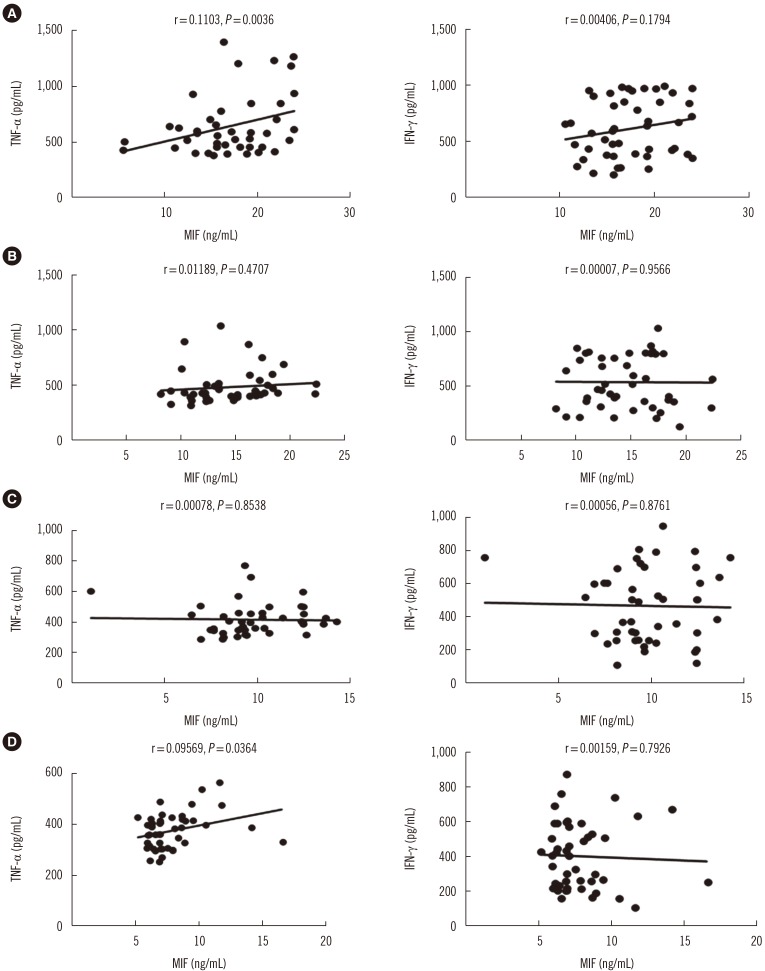
MIF serum levels were significantly elevated (P < 0.05) in patients with APTB prior to treatment compared with that in control subjects, and TNF-α≥449.7 pg/mL was associated with high MIF levels (≥13.1 ng/mL). MIF levels were significantly reduced (P < 0.01) following 2, 4, and 6 months of treatment, with variations in TNF-α and IFN-γ serum levels. MIF levels were positively correlated with the paired TNF-α level at baseline (r=0.1103, P=0.0316) and following 6 months of treatment (r=0.09569, P=0.0364).
CONCLUSIONS
A reduction in the MIF serum levels in patients with APTB following anti-tuberculosis treatment may positively affect host immune protection against Mycobacterium tuberculosis infection. Thus, serum MIF levels may constitute a useful marker for assessing therapy effectiveness in patients with APTB.
MeSH Terms
Figure
Reference
-
1. Calandra T, Roger T. Macrophage migration inhibitory factor: a regulator of innate immunity. Nat Rev Immunol. 2003; 3:791–800. PMID: 14502271.2. Gregory JL, Morand EF, Mckeown SJ, Ralph JA, Hall P, Yang YH, et al. Macrophage migration inhibitory factor induces macrophage recruitment via CC chemokine ligand 2. J Immunol. 2006; 177:8072–8079. PMID: 17114481.3. Tillmann S, Bernhagen J, Noels H. Arrest functions of the MIF ligand/receptor axes in atherogenesis. Front Immunol. 2013; 4:115. PMID: 23720662.4. Santos LL, Morand EF. Macrophage migration inhibitory factor: a key cytokine in RA, SLE and atherosclerosis. Clin Chim Acta. 2013; 399:1–7.5. Morand EF, Leech M, Bemhagen J. MIF: a new cytokine link between rheumatoid arthritis and atherosclerosis. Nat Rev Drug Discov. 2006; 5:399–410. PMID: 16628200.6. Baugh JA, Bucala R. Macrophage migration inhibitory factor. Crit Care Med. 2002; 30:S27–S35.7. Lolis E, Bucala R. Macrophage migration inhibitory factor. Expert Opin Ther Targets. 2003; 7:153–164. PMID: 12667094.8. Leng L, Bucala R. Macrophage migration inhibitory factor. Critical Care Med. 2005; 33:S475–S477. PMID: 16340426.9. Coban N, Onat A, Yildirim O, Can G, Erginelunaltuna N. Oxidative stress-mediated (sex-specific) loss of protection against type-2 diabetes by macrophage migration inhibitory factor (MIF)-173G/C polymorphism. Clin Chim Acta. 2015; 438:1–6. PMID: 25108206.10. Calandra T, Bernhagen J, Metz CN, Spiegel LA, Bacher M, Donnelly T, et al. MIF as a glucocorticoid-induced modulator of cytokine production. Nature. 1995; 377:68–71. PMID: 7659164.11. Awandare GA, Martinson JJ, Were T, Ouma C, Davenport GC, Ong'echa JM, et al. MIF (macrophage migration inhibitory factor) promoter polymorphism and susceptibility to severe malarial anemia. J Infect Dis. 2009; 200:629–637. PMID: 19591577.12. Yende S, Angus DC, Kong L, Kellum JA, Weissfeld L, Ferrell R, et al. The influence of macrophage migration inhibitory factor gene polymorphism on outcome from community-acquired pneumonia. FASEB J. 2009; 23:2403–2411. PMID: 19346297.13. Renner P, Roger T, Bochud PY, Sprong T, Sweep FC, Bochud M, et al. A functional microsatellite of the macrophage migration inhibitory factor gene associated with meningococcal disease. FASEB J. 2012; 26:907–916. PMID: 21990375.14. Cooper AM, Khader SA. The role of cytokines in the initiation, expansion, and control of cellular immunity to tuberculosis. Immunol Rev. 2008; 226:191–204. PMID: 19161425.15. Ameglio F, Casarini M, Capoluongo E, Mattia P, Puglisi G, Giosue S. Post-treatment changes of six cytokines in active pulmonary tuberculosis: differences between patients with stable or increased fibrosis. Int J Tuberc Lung Dis. 2005; 9:98–104. PMID: 15675558.16. Munk ME, Emoto M. Functions of T-cell subsets and cytokines in mycobacterial infections. Eur Respir J. 1995; 20:668–675.17. Boom WH, Canaday DH, Fulton SA, Gehring AJ, Rojas RE, Torres M. Human immunity to M. tuberculosis: T cell subsets and antigen processing. Tuberculosis. 2003; 83:98–106. PMID: 12758197.18. Denis M. Interferon-gamma-treated murine macrophages inhibit growth of tubercle bacilli via the generation of reactive nitrogen intermediates. Cell Immunol. 1991; 132:150–157. PMID: 1905984.19. Gutierrez MG, Master SS, Singh SB, Taylor GA, Colombo MI, Deretic V. Autophagy is a defense mechanism inhibiting BCG and Mycobacterium tuberculosis survival in infected macrophages. Cell. 2004; 119:753–766. PMID: 15607973.20. Jacobs M, Brown N, Allie N, Rvffel B. Fatal Mycobacterium bovis BCG infection in TNF-LT-alpha –deficient mice. Clin Immunol. 2000; 94:192–199. PMID: 10692238.21. Van CR, Ottenhoff TH, Vander Meer JW. Innate immunity to Mycobacterium tuberculosis. Clin Microbiol Rev. 2002; 15:294–309. PMID: 11932234.22. White DA, Fang L, Chan W, Morand EF, Kiriazis H, Duffy SJ, et al. Proinflammatory action of MIF in acute myocardial infarction via activation of peripheral blood mononuclear cells. PloS One. 2013; 8:e76206. PMID: 24098445.23. Llamas-Covarrubias MA, Valle Y, Bucala R, Navarro-Hernandez RE, Palafox-Sanchez CA, Padilla-Gutierrez JR, et al. Macrophage migration inhibitory factor (MIF): genetic evidence for participation in early onset and early stage rheumatoid arthritis. Cytokine. 2013; 61:759–765. PMID: 23402792.24. Vassalli P. The pathophysiology of tumor necrosis factors. Annu Rev Immunol. 1992; 10:411–452. PMID: 1590993.25. Pasparakis M, Alexopoulou L, Douni E, Kollias G. Tumour necrosis factors in immune regulation: everything that's interesting is. . .new! Cytokine Growth Factor Rev. 1996; 7:223–229. PMID: 8971477.26. Flynn JL, Chan J. Immunology of tuberculosis. Annu Rev Immunol. 2001; 19:93–129. PMID: 11244032.27. Berry MP, Graham CM, McNab FW, Bioch SA, Oni T, Wilkinson KA, et al. An interferon-inducible neutrophil-driven blood transcriptional signature in human tuberculosis. Nature. 2010; 466:973–977. PMID: 20725040.28. Bean AG, Roach DR, Briscoe H, France MP, Korner H, SedqwicK JD, et al. Structural deficiencies in granuloma formation in TNF gene-targeted mice underlie the heightened susceptibility to aerosol Mycobacterium tuberculosis infection, which is not compensated for by lymphotoxin. J Immunol. 1999; 162:3504–3511. PMID: 10092807.29. Chakravarty SD, Zhu G, Tsai MC, Mohan VP, Marino S, Kirschner DE, et al. Tumor necrosis factor blockade in chronic murine tuberculosis enhances granulomatous inflammation and disorganizes granulomas in the lungs. Infect Immun. 2008; 76:916–926. PMID: 18212087.30. Roach DR, Briscoe H, Saunders B, France MP, Riminton S, Britton WJ. Secreted lymphotoxin-α is essential for the control of an intracellular bacterial infection. J Exp Med. 2001; 193:239–246. PMID: 11208864.31. Botha T, Ryffel B. Reactivation of latent tuberculosis infection in TNF-deficient mice. J Immunol. 2003; 171:3110–3118. PMID: 12960337.32. Mohan VP, Scanga CA, Yu K, Scottd HM, Tanaka KE, Tsanq E, et al. Effects of tumor necrosis factor α on host immune response in chronic persistent tuberculosis: possible role for limiting pathology. Infect Immun. 2001; 69:1847–1855. PMID: 11179363.33. Smith SM, Klein MR, Malin AS, Sillah J, Mcadam KP, Dockrell HM, et al. Decreased IFN-gamma and increased IL-4 production by human CD8(+) T cells in response to Mycobacterium tuberculosis in tuberculosis patients. Tuberculosis. 2002; 82:7–13. PMID: 11914057.34. Sharma S, Sharma M, Roy S, Kumar P, Bose M. Mycobacterium tuberculosis induces high production of nitric oxide in coordination with production of tumor necrosis factor-alpha in patients with fresh active tuberculosis but not in MDR tuberculosis. Immunol Cell Biol. 2004; 82:377–382. PMID: 15283847.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Superoxide Generation by Blood Monocyte and Pulmonary Alveolar Macrophage in Patients with Pulmonary Tuberculosis
- Normal Serum Macrophage Migration Inhibitory Factor Concentrations of Korean people According to Age
- Elevated Serum Levels of Macrophage Migration Inhibitory Factor (MIF) in Patients with Behcet's Disease
- Macrophage migration inhibitory factor: a potential therapeutic target for rheumatoid arthritis
- The Activities of Leucocytic Acid Phosphatase in Pulmonary Tuberculosis