J Korean Med Sci.
2017 Dec;32(12):2021-2028. 10.3346/jkms.2017.32.12.2021.
Long-Term Survival Analysis of Intraperitoneal versus Intravenous Chemotherapy for Primary Ovarian Cancer and Comparison between Carboplatin- and Cisplatin-based Intraperitoneal Chemotherapy
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Institute of Women's Life Medical Science, Women's Cancer Center, Yonsei University College of Medicine, Seoul, Korea. san1@yuhs.ac
- KMID: 2396381
- DOI: http://doi.org/10.3346/jkms.2017.32.12.2021
Abstract
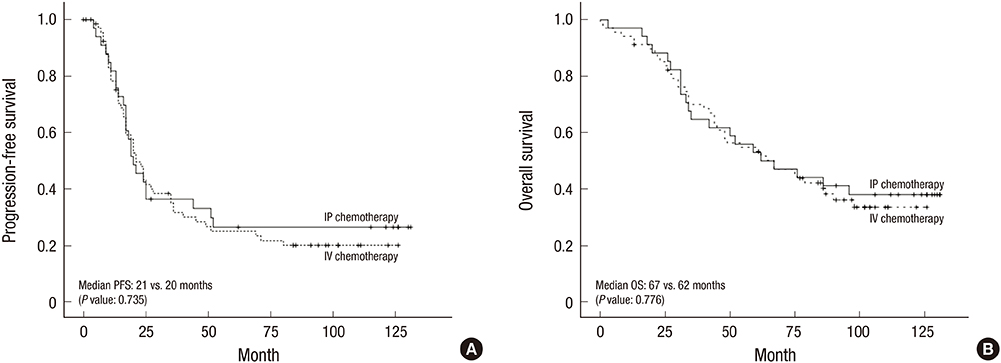
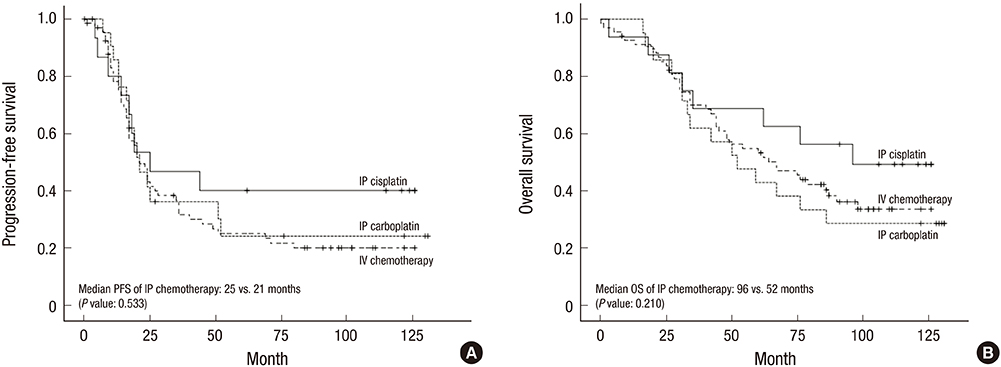
- In epithelial ovarian cancer (EOC), intraperitoneal (IP) administration of chemotherapy is an effective first-line treatment and may improve outcomes, compared with intravenous (IV) chemotherapy. We used Kaplan-Meier survival analysis to compare long-term survival between propensity score-matched patients with advanced EOC receiving IP (n = 34) vs. IV (n = 68) chemotherapy. Additionally, clinical features associated with carboplatin-based (n = 21) and cisplatin-based (n = 16) IP chemotherapy were analyzed and compared with those associated with IV chemotherapy. The IP and IV chemotherapy groups had a median follow-up duration of 67 (range, 3-131) and 62 (range, 0-126) months, respectively, with no significant difference in progression-free survival (PFS) (P = 0.735) and overall survival (OS) (P = 0.776). A significantly higher proportion of patients in the IV (91.2%) than in the IP (67.6%) chemotherapy group (P = 0.004) received ≥ 6 cycles. However, the frequency of toxic events (anemia, granulocytopenia, nausea/vomiting, abdominal pain, hepatotoxicity, neuromuscular effects) was significantly higher in the IP than in the IV group. Within the IP group, no significant differences were observed in PFS (P = 0.533) and OS (P = 0.210) between the cisplatin-based and carboplatin-based chemotherapy subgroups. The 10-year OS was 28.6% and 49.2% in carboplatin-based and cisplatin-based IP chemotherapy groups, respectively. Toxic events (granulocytopenia, leukopenia, nausea/vomiting, abdominal pain, hepatotoxicity, neuromuscular effects) were significantly more common in the cisplatin-based subgroup. In patients with EOC, cisplatin-based IP chemotherapy may be an acceptable alternative to IV chemotherapy regarding long-term survival, but toxicity must be addressed.
MeSH Terms
Figure
Reference
-
1. Kim SI, Lim MC, Lim J, Won YJ, Seo SS, Kang S, Park SY. Incidence of epithelial ovarian cancer according to histologic subtypes in Korea, 1999 to 2012. J Gynecol Oncol. 2016; 27:e5.2. Lee M, Chang MY, Yoo H, Lee KE, Chay DB, Cho H, Kim S, Kim YT, Kim JH. Clinical significance of CA125 level after the first cycle of chemotherapy on survival of patients with advanced ovarian cancer. Yonsei Med J. 2016; 57:580–587.3. Ozols RF. Treatment of ovarian cancer: current status. Semin Oncol. 1994; 21:1–9.4. Vaccarello L, Rubin SC, Vlamis V, Wong G, Jones WB, Lewis JL, Hoskins WJ. Cytoreductive surgery in ovarian carcinoma patients with a documented previously complete surgical response. Gynecol Oncol. 1995; 57:61–65.5. Alberts DS, Liu PY, Hannigan EV, O'Toole R, Williams SD, Young JA, Franklin EW, Clarke-Pearson DL, Malviya VK, DuBeshter B, et al. Intraperitoneal cisplatin plus intravenous cyclophosphamide versus intravenous cisplatin plus intravenous cyclophosphamide for stage III ovarian cancer. N Engl J Med. 1996; 335:1950–1955.6. Markman M, Bundy BN, Alberts DS, Fowler JM, Clark-Pearson DL, Carson LF, Wadler S, Sickel J. Phase III trial of standard-dose intravenous cisplatin plus paclitaxel versus moderately high-dose carboplatin followed by intravenous paclitaxel and intraperitoneal cisplatin in small-volume stage III ovarian carcinoma: an intergroup study of the Gynecologic Oncology Group, Southwestern Oncology Group, and Eastern Cooperative Oncology Group. J Clin Oncol. 2001; 19:1001–1007.7. Armstrong DK, Bundy B, Wenzel L, Huang HQ, Baergen R, Lele S, Copeland LJ, Walker JL, Burger RA. Gynecologic Oncology Group. Intraperitoneal cisplatin and paclitaxel in ovarian cancer. N Engl J Med. 2006; 354:34–43.8. Jaaback K, Johnson N. Intraperitoneal chemotherapy for the initial management of primary epithelial ovarian cancer. Cochrane Database Syst Rev. 2006; CD005340.9. Miyagi Y, Fujiwara K, Kigawa J, Itamochi H, Nagao S, Aotani E, Terakawa N, Kohno I. Sankai Gynecology Study Group (SGSG). Intraperitoneal carboplatin infusion may be a pharmacologically more reasonable route than intravenous administration as a systemic chemotherapy. A comparative pharmacokinetic analysis of platinum using a new mathematical model after intraperitoneal vs. intravenous infusion of carboplatin--a Sankai Gynecology Study Group (SGSG) study. Gynecol Oncol. 2005; 99:591–596.10. Fujiwara K, Nagao S, Kigawa J, Noma J, Akamatsu N, Miyagi Y, Numa F, Okada M, Aotani E. Phase II study of intraperitoneal carboplatin with intravenous paclitaxel in patients with suboptimal residual epithelial ovarian or primary peritoneal cancer: a Sankai Gynecology Cancer Study Group Study. Int J Gynecol Cancer. 2009; 19:834–837.11. Nagao S, Fujiwara K, Ohishi R, Nakanishi Y, Iwasa N, Shimizu M, Goto T, Shimoya K. Combination chemotherapy of intraperitoneal carboplatin and intravenous paclitaxel in suboptimally debulked epithelial ovarian cancer. Int J Gynecol Cancer. 2008; 18:1210–1214.12. Fujiwara K, Sakuragi N, Suzuki S, Yoshida N, Maehata K, Nishiya M, Koshida T, Sawai H, Aotani E, Kohno I. First-line intraperitoneal carboplatin-based chemotherapy for 165 patients with epithelial ovarian carcinoma: results of long-term follow-up. Gynecol Oncol. 2003; 90:637–643.13. Kim SW, Paek J, Nam EJ, Kim SH, Kim JH, Kim YT. The feasibility of carboplatin-based intraperitoneal chemotherapy in ovarian cancer. Eur J Obstet Gynecol Reprod Biol. 2010; 152:195–199.14. Bouchard-Fortier G, Rosen B, Vyarvelska I, Pasetka M, Covens A, Gien LT, Kupets R, Pulman K, Ferguson SE, Vicus D. A comparison of the toxicity and tolerability of two intraperitoneal chemotherapy regimens for advanced-stage epithelial ovarian cancer. Gynecol Oncol. 2016; 140:36–41.15. Makhija S, Leitao M, Sabbatini P, Bellin N, Almadrones L, Leon L, Spriggs DR, Barakat R. Complications associated with intraperitoneal chemotherapy catheters. Gynecol Oncol. 2001; 81:77–81.16. Crabtree JH, Chow KM. Peritoneal dialysis catheter insertion. Semin Nephrol. 2017; 37:17–29.17. Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van Oosterom AT, Christian MC, et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst. 2000; 92:205–216.18. National Cancer Institute (US). Cancer therapy evaluation program: common toxicity criteria [Internet]. accessed on 10 June 2017. Available at http://ctep.cancer.gov.19. Tewari D, Java JJ, Salani R, Armstrong DK, Markman M, Herzog T, Monk BJ, Chan JK. Long-term survival advantage and prognostic factors associated with intraperitoneal chemotherapy treatment in advanced ovarian cancer: a gynecologic oncology group study. J Clin Oncol. 2015; 33:1460–1466.20. Trimble EL, Alvarez RD. Intraperitoneal chemotherapy and the NCI clinical announcement. Gynecol Oncol. 2006; 103:S18–S19.21. Wright AA, Cronin A, Milne DE, Bookman MA, Burger RA, Cohn DE, Cristea MC, Griggs JJ, Keating NL, Levenback CF, et al. Use and effectiveness of intraperitoneal chemotherapy for treatment of ovarian cancer. J Clin Oncol. 2015; 33:2841–2847.22. Havrilesky LJ, Alvarez Secord A, Ehrisman JA, Berchuck A, Valea FA, Lee PS, Gaillard SL, Samsa GP, Cella D, Weinfurt KP, et al. Patient preferences in advanced or recurrent ovarian cancer. Cancer. 2014; 120:3651–3659.23. Van der Speeten K, Govaerts K, Stuart OA, Sugarbaker PH. Pharmacokinetics of the perioperative use of cancer chemotherapy in peritoneal surface malignancy patients. Gastroenterol Res Pract. 2012; 2012:378064.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Long-term survival after intraperitoneal chemotherapy with paclitaxel-cisplatin for recurrent primary peritoneal cancer resistant to multiple lines of intravenous chemotherapy
- New paradigm of intraperitoneal chemotherapy in ovarian carcinoma
- Chemotherapy for ovarian cancer
- Treatment of Peritoneal Carcinomatosis from Colorectal Cancer
- Effect of Radical Removal of Primary and Metastatic Lesions in Gastric Cancer with Peritoneal Seeding