J Korean Neurosurg Soc.
2017 Nov;60(6):717-722. 10.3340/jkns.2017.0404.010.
Delayed Burr Hole Surgery in Patients with Acute Subdural Hematoma: Clinical Analysis
- Affiliations
-
- 1Department of Neurosurgery, Ilsan Paik Hospital, Inje University College of Medicine, Goyang, Korea. hsrkmj@paik.ac.kr
- KMID: 2395792
- DOI: http://doi.org/10.3340/jkns.2017.0404.010
Abstract
OBJECTIVE
To evaluate the effectiveness and efficacy of delayed burr hole surgery in relation to the reduction of postoperative subdural hematoma (SDH) volume in patients with acute SDH.
METHODS
We retrospectively analyzed patients with acute SDH who received delayed burr hole surgery at our institute. Age, sex, Glasgow coma scale, maximal SDH thickness, volume of SDH, midline shifts, hounsfield unit (HU), and medical history of anticoagulant agent usage were recorded. Outcome measures were delayed operation day, reduction of SDH volume after operation, and the Glasgow outcome scale (GOS) score at discharge. The patients were divided two groups according to the post-operative reduction of volume of SDH (≥50%, group A; <50%, group B). We also analyzed variables and differences between two groups.
RESULTS
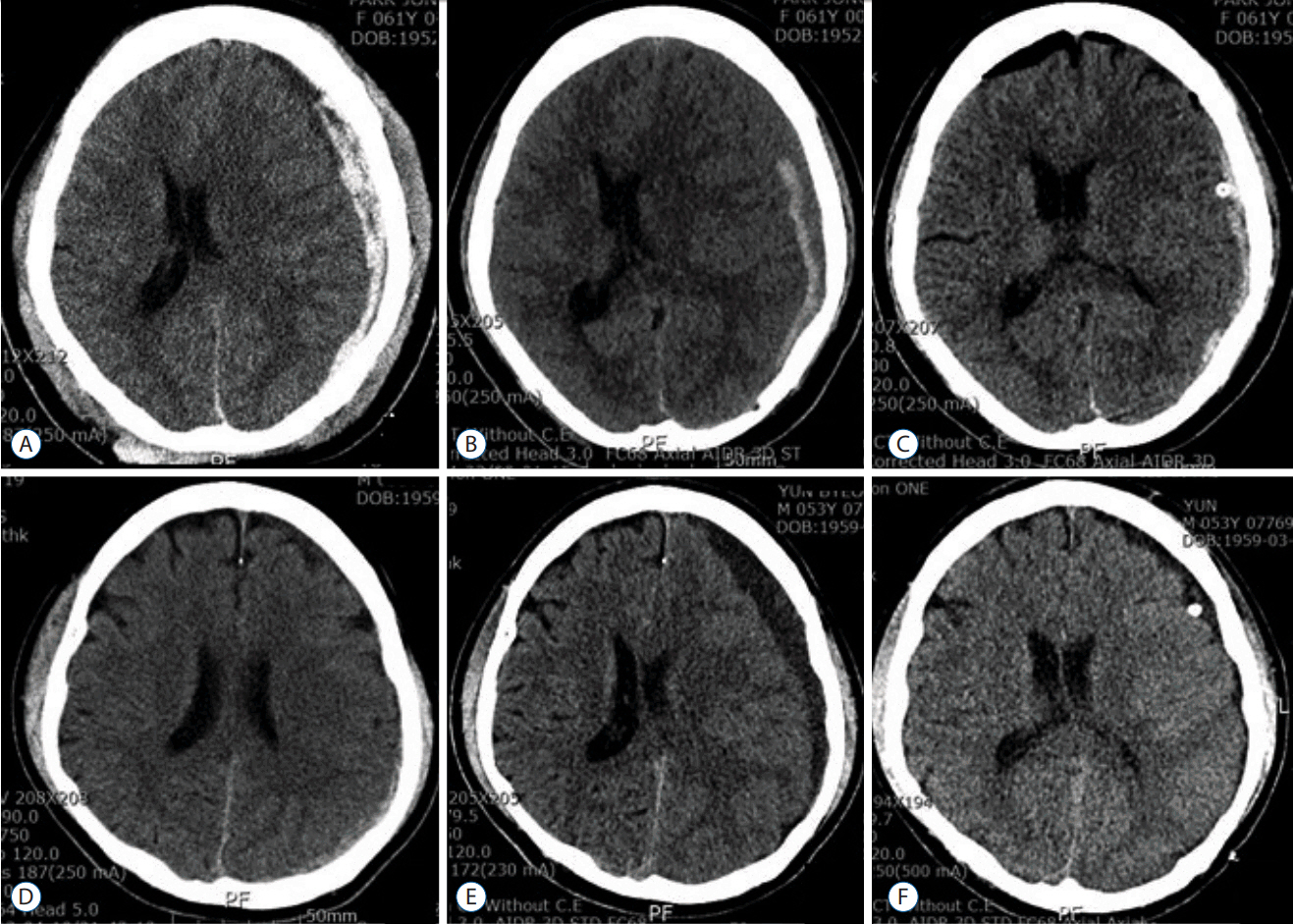
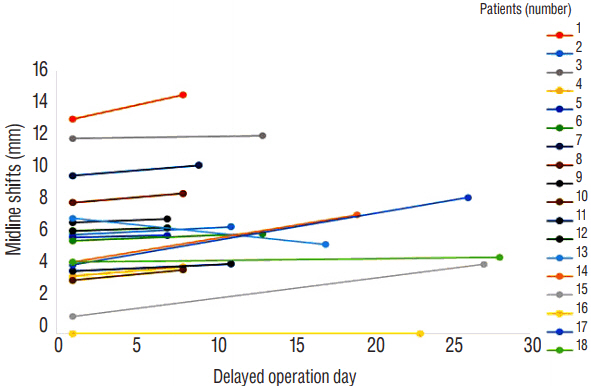
Eighteen patients were available for this analysis. The mean delayed of surgery was 13.9±7.5 days. Maximal thickness of SDH was changed from 10.0±3.5 mm to 12.2±3.7 mm. Volume of SDH was changed from 38.7±28.0 mL to 42.6±29.6 mL. Midline shifts were changed from 5.8±3.3 mm to 6.6±3.3 mm. HU were changed from 66.4±11.2 to 53.2±20.6. Post-operative reduction of SDH volume was 52.1±21.1%. Eleven patients (61%) had a discharge GOS score of 1 (good recovery). Ten patients (56%) were enrolled in group A. Midline shifting was greater in group A than in group B (7.4±3.3 vs. 3.0±2.4 mm; p<0.02). The delay of surgery was shorter for group A than group B (9.2±2.3 vs. 19.8±7.7 days; p<0.0008).
CONCLUSION
Among well selected patients, delayed burr hole surgery in patients with acute SDH may be effective for reduction of SDH volume. Further studies will be necessary to establish the effectiveness and safety of delayed burr hole surgery in patients with acute SDH.
Keyword
MeSH Terms
Figure
Reference
-
References
1. Bajsarowicz P, Prakash I, Lamoureux J, Saluja RS, Feyz M, Maleki M, et al. Nonsurgical acute traumatic subdural hematoma: what is the risk? J Neurosurg. 123:1176–1183. 2015.
Article2. Godlewski B, Pawelczyk A, Pawelczyk T, Ceranowicz K, Wojdyn M, Radek M. Retrospective analysis of operative treatment of a series of 100 patients with subdural hematoma. Neurol Med Chir (Tokyo). 53:26–33. 2013.
Article3. Izumihara A, Yamashita K, Murakami T. Acute subdural hematoma requiring surgery in the subacute or chronic stage. Neurol Med Chir (Tokyo). 53:323–328. 2013.
Article4. Karibe H, Hayashi T, Hirano T, Kameyama M, Nakagawa A, Tominaga T. Surgical management of traumatic acute subdural hematoma in adults: a review. Neurol Med Chir (Tokyo). 54:887–894. 2014.
Article5. Kim BJ, Park KJ, Park DH, Lim DJ, Kwon TH, Chung YG, et al. Risk factors of delayed surgical evacuation for initially nonoperative acute subdural hematomas following mild head injury. Acta Neurochir (Wien). 156:1605–1613. 2014.
Article6. Kuwahara S, Fukuoka M, Koan Y, Miyake H, Ono Y, Moriki A, et al. Diffusion-weighted imaging of traumatic subdural hematoma in the subacute stage. Neurol Med Chir (Tokyo). 45:464–469. 2005.
Article7. Laviv Y, Rappaport ZH. Risk factors for development of significant chronic subdural hematoma following conservative treatment of acute subdural hemorrhage. Br J Neurosurg. 28:733–738. 2014.
Article8. Lee JJ, Won Y, Yang T, Kim S, Choi CS, Yang J. Risk factors of chronic subdural hematoma progression after conservative management of cases with initially acute subdural hematoma. Korean J Neurotrauma. 11:52–57. 2015.
Article9. Lindvall P, Koskinen LO. Anticoagulants and antiplatelet agents and the risk of development and recurrence of chronic subdural haematomas. J Clin Neurosci. 16:1287–1290. 2009.
Article10. Mathew P, Oluoch-Olunya DL, Condon BR, Bullock R. Acute subdural haematoma in the conscious patient: outcome with initial non-operative management. Acta Neurochir (Wien). 121:100–108. 1993.
Article11. Rust T, Kiemer N, Erasmus A. Chronic subdural haematomas and anticoagulation or anti-thrombotic therapy. J Clin Neurosci. 13:823–827. 2006.
Article12. Torihashi K, Sadamasa N, Yoshida K, Narumi O, Chin M, Yamagata S. Independent predictors for recurrence of chronic subdural hematoma: a review of 343 consecutive surgical cases. Neurosurgery. 63:1125–1129. discussion 1129. 2008.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Delayed Onset Acute Subdural Hematoma after Burr Hole Drainage in a Patient with Chronic Subdural Hematoma and Liver Cirrhosis
- Evolution of Chronic Subdural Hematoma based on Brain CT findings and Appropriate Treatment Methods
- Cerebellar Hemorrhage after Burr Hole Drainage of Supratentorial Chronic Subdural Hematoma
- Factors Affecting the Reaccumulation of Chronic Subdural Hematoma after Burr-hole Trephination and Closed-system Drainage
- Burr hole drainage using urokinase for treatment of subacute subdural hematoma