Obstet Gynecol Sci.
2017 Sep;60(5):449-454. 10.5468/ogs.2017.60.5.449.
Gonadotropin releasing hormone antagonist administration for treatment of early type severe ovarian hyperstimulation syndrome: a case series
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Seoul National University Bundang Hospital, Seongnam, Korea. blasto@snubh.org
- 2Department of Obstetrics and Gynecology, Seoul National University Hospital, Seoul, Korea.
- KMID: 2393844
- DOI: http://doi.org/10.5468/ogs.2017.60.5.449
Abstract
OBJECTIVE
To report an efficacy of gonadotropin releasing hormone (GnRH) antagonist administration after freezing of all embryos for treatment of early type ovarian hyperstimulation syndrome (OHSS).
METHODS
In 10 women who developed fulminant early type OHSS after freezing of all embryos, GnRH antagonist (cetrorelix 0.25 mg per day) was started at the time of hospitalization and continued for 2 to 4 days. Fluid therapy and drainage of ascites was performed as usual.
RESULTS
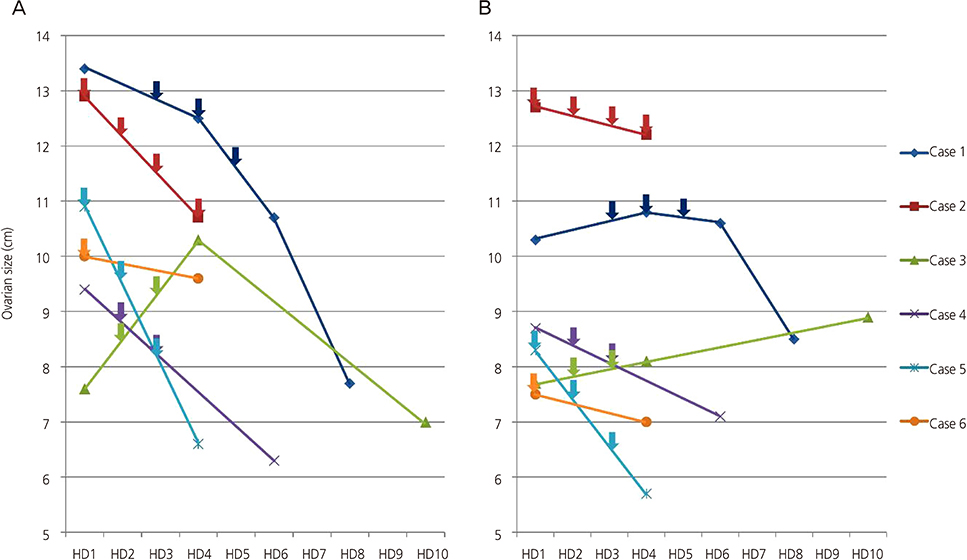
Early type OHSS was successfully treated without any complication. At hospitalization, the median (95% confidence interval [CI]) of the right and the left ovarian diameter was 10.0 cm (7.6 to 12.9 cm) and 8.5 cm (7.5 to 12.6 cm). After completion of GnRH antagonist administration, it was decreased to 7.4 cm (6.2 to 10.7 cm) (P=0.028) and 7.8 cm (5.7 to 12.2 cm) (P=0.116), respectively. The median duration of hospital stay was 6 days (3 to 11 days). Trans-abdominal drainage of ascites was performed in 2 women and drainage of ascites by percutaneous indwelling catheter was performed in 4 women. No side effect of GnRH antagonist was noted.
CONCLUSION
GnRH antagonist administration appears to be safe and effective for women with fulminant early type OHSS after freezing all embryos. Optimal dose or duration of GnRH antagonist should be further determined.
Keyword
MeSH Terms
Figure
Reference
-
1. Practice Committee of the American Society for Reproductive Medicine. Prevention and treatment of moderate and severe ovarian hyperstimulation syndrome: a guideline. Fertil Steril. 2016; 106:1634–1647.2. D'Angelo A, Amso NN. Embryo freezing for preventing ovarian hyperstimulation syndrome: a Cochrane review. Hum Reprod. 2002; 17:2787–2794.3. Mathur RS, Akande AV, Keay SD, Hunt LP, Jenkins JM. Distinction between early and late ovarian hyperstimulation syndrome. Fertil Steril. 2000; 73:901–907.4. Farquhar C, Marjoribanks J, Brown J, Fauser BC, Lethaby A, Mourad S, et al. Management of ovarian stimulation for IVF: narrative review of evidence provided for World Health Organization guidance. Reprod Biomed Online. 2017; 35:3–16.5. Rizk B, Smitz J. Ovarian hyperstimulation syndrome after superovulation using GnRH agonists for IVF and related procedures. Hum Reprod. 1992; 7:320–327.6. Endo T, Honnma H, Hayashi T, Chida M, Yamazaki K, Kitajima Y, et al. Continuation of GnRH agonist administration for 1 week, after hCG injection, prevents ovarian hyperstimulation syndrome following elective cryopreservation of all pronucleate embryos. Hum Reprod. 2002; 17:2548–2551.7. Wang YQ, Yu N, Xu WM, Xie QZ, Yan WJ, Wu GX, et al. Cetrotide administration in the early luteal phase in patients at high risk of ovarian hyperstimulation syndrome: a controlled clinical study. Exp Ther Med. 2014; 8:1855–1860.8. Lainas TG, Sfontouris IA, Zorzovilis IZ, Petsas GK, Lainas GT, Kolibianakis EM. Management of severe early ovarian hyperstimulation syndrome by re-initiation of GnRH antagonist. Reprod Biomed Online. 2007; 15:408–412.9. Lainas TG, Sfontouris IA, Zorzovilis IZ, Petsas GK, Lainas GT, Iliadis GS, et al. Management of severe OHSS using GnRH antagonist and blastocyst cryopreservation in PCOS patients treated with long protocol. Reprod Biomed Online. 2009; 18:15–20.10. Lainas TG, Sfontouris IA, Zorzovilis IZ, Petsas GK, Lainas GT, Alexopoulou E, et al. Live births after management of severe OHSS by GnRH antagonist administration in the luteal phase. Reprod Biomed Online. 2009; 19:789–795.11. Lainas GT, Kolibianakis EM, Sfontouris IA, Zorzovilis IZ, Petsas GK, Tarlatzi TB, et al. Outpatient management of severe early OHSS by administration of GnRH antagonist in the luteal phase: an observational cohort study. Reprod Biol Endocrinol. 2012; 10:69.12. Hosseini MA, Mahdavi A, Aleyasin A, Safdarian L, Bahmaee F. Treatment of ovarian hyperstimulation syndrome using gonadotropin releasing hormone antagonist: a pilot study. Gynecol Endocrinol. 2012; 28:853–855.13. Delvigne A, Rozenberg S. Review of clinical course and treatment of ovarian hyperstimulation syndrome (OHSS). Hum Reprod Update. 2003; 9:77–96.14. Schenker JG. Prevention and treatment of ovarian hyperstimulation. Hum Reprod. 1993; 8:653–659.15. Levin I, Almog B, Avni A, Baram A, Lessing JB, Gamzu R. Effect of paracentesis of ascitic fluids on urinary output and blood indices in patients with severe ovarian hyperstimulation syndrome. Fertil Steril. 2002; 77:986–988.16. Lan KC, Huang FJ, Lin YC, Kung FT, Lan TH, Chang SY. Significantly superior response in the right ovary compared with the left ovary after stimulation with follicle-stimulating hormone in a pituitary down-regulation regimen. Fertil Steril. 2010; 93:2269–2273.17. Choe SA, Ku SY, Jee BC, Suh CS, Kim SH, Choi YM, et al. Symmetry in number of retrieved oocytes between two ovaries: a possible predictor of in vitro fertilization outcome. Gynecol Endocrinol. 2011; 27:997–1000.18. Fukuda M, Fukuda K, Andersen CY, Byskov AG. Characteristics of human ovulation in natural cycles correlated with age and achievement of pregnancy. Hum Reprod. 2001; 16:2501–2507.19. Potashnik G, Insler V, Meizner I, Sternberg M. Frequency, sequence, and side of ovulation in women menstruating normally. Br Med J (Clin Res Ed). 1987; 294:219.20. Dominguez R, Cruz ME, Chavez R. Differences in the ovulatory ability between the right and left ovary are related to ovarian innervation. In : Hirshfield AN, editor. Growth factors and the ovary. Boston (MA): Springer;1989. p. 321–325.21. Latouche J, Crumeyrolle-Arias M, Jordan D, Kopp N, Augendre-Ferrante B, Cedard L, et al. GnRH receptors in human granulosa cells: anatomical localization and characterization by autoradiographic study. Endocrinology. 1989; 125:1739–1741.22. Minaretzis D, Jakubowski M, Mortola JF, Pavlou SN. Gonadotropin-releasing hormone receptor gene expression in human ovary and granulosa-lutein cells. J Clin Endocrinol Metab. 1995; 80:430–434.23. Asimakopoulos B, Nikolettos N, Nehls B, Diedrich K, Al-Hasani S, Metzen E. Gonadotropin-releasing hormone antagonists do not influence the secretion of steroid hormones but affect the secretion of vascular endothelial growth factor from human granulosa luteinized cell cultures. Fertil Steril. 2006; 86:636–641.24. Taylor PD, Hillier SG, Fraser HM. Effects of GnRH antagonist treatment on follicular development and angiogenesis in the primate ovary. J Endocrinol. 2004; 183:1–17.25. Fridén BE, Nilsson L. Gonadotrophin-releasing hormone-antagonist luteolysis during the preceding mid-luteal phase is a feasible protocol in ovarian hyperstimulation before in vitro fertilization. Acta Obstet Gynecol Scand. 2005; 84:812–816.26. Del Canto F, Sierralta W, Kohen P, Muñoz A, Strauss JF 3rd, Devoto L. Features of natural and gonadotropin-releasing hormone antagonist-induced corpus luteum regression and effects of in vivo human chorionic gonadotropin. J Clin Endocrinol Metab. 2007; 92:4436–4443.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The use of gonadotropin-releasing hormone antagonist post-ovulation trigger in ovarian hyperstimulation syndrome
- The effective of ultrashort-term gonadotropin-releasing hormone agonist administration in controlled ovarian hyperstimulation for in vitro fertilization
- Ovarian Hyper-Response to Administration of an GnRH-Agonist Without Gonadotropins
- Prediction and Prevention of Ovarian Hyperstimulation Syndrome
- GnRH analogue in controlled ovarian hyperstimulation for gonadotropin poor responder