Korean J Pain.
2017 Oct;30(4):243-249. 10.3344/kjp.2017.30.4.243.
Practical statistics in pain research
- Affiliations
-
- 1Department of Anesthesia and Pain Medicine, Pusan National University School of Medicine, Yangsan, Korea. anesktk@pusan.ac.kr
- KMID: 2392900
- DOI: http://doi.org/10.3344/kjp.2017.30.4.243
Abstract
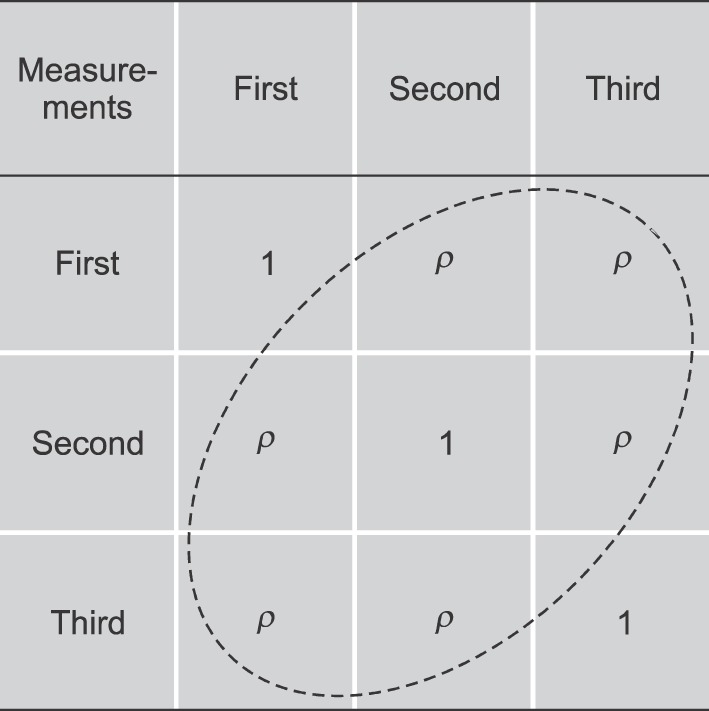
- Pain is subjective, while statistics related to pain research are objective. This review was written to help researchers involved in pain research make statistical decisions. The main issues are related with the level of scales that are often used in pain research, the choice of statistical methods between parametric or nonparametric statistics, and problems which arise from repeated measurements. In the field of pain research, parametric statistics used to be applied in an erroneous way. This is closely related with the scales of data and repeated measurements. The level of scales includes nominal, ordinal, interval, and ratio scales. The level of scales affects the choice of statistics between parametric or non-parametric methods. In the field of pain research, the most frequently used pain assessment scale is the ordinal scale, which would include the visual analogue scale (VAS). There used to be another view, however, which considered the VAS to be an interval or ratio scale, so that the usage of parametric statistics would be accepted practically in some cases. Repeated measurements of the same subjects always complicates statistics. It means that measurements inevitably have correlations between each other, and would preclude the application of one-way ANOVA in which independence between the measurements is necessary. Repeated measures of ANOVA (RMANOVA), however, would permit the comparison between the correlated measurements as long as the condition of sphericity assumption is satisfied. Conclusively, parametric statistical methods should be used only when the assumptions of parametric statistics, such as normality and sphericity, are established.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Comparison of modified thoracoabdominal nerve block through perichondral approach and subcostal transversus abdominis plane block for pain management in laparoscopic cholecystectomy: a randomized-controlled trial
Hye-Yeon Cho, In Eob Hwang, Mirang Lee, Wooil Kwon, Won Ho Kim, Ho-Jin Lee
Korean J Pain. 2023;36(3):382-391. doi: 10.3344/kjp.23114.
Reference
-
1. Yim KH, Nahm FS, Han KA, Park SY. Analysis of statistical methods and errors in the articles published in the Korean Journal of Pain. Korean J Pain. 2010; 23:35–41. PMID: 20552071.
Article2. Norman G. Likert scales, levels of measurement and the “laws” of statistics. Adv Health Sci Educ Theory Pract. 2010; 15:625–632. PMID: 20146096.
Article4. Price DD, McGrath PA, Rafii A, Buckingham B. The validation of visual analogue scales as ratio scale measures for chronic and experimental pain. Pain. 1983; 17:45–56. PMID: 6226917.
Article5. Dexter F, Chestnut DH. Analysis of statistical tests to compare visual analog scale measurements among groups. Anesthesiology. 1995; 82:896–902. PMID: 7717561.
Article6. Nunnally JC, Bernstein IH. Psychometric theory. 3rd ed. New York (NY): McGraw-Hill Book Company;1994. p. 115.7. Heeren T, D'Agostino R. Robustness of the two independent samples t-test when applied to ordinal scaled data. Stat Med. 1987; 6:79–90. PMID: 3576020.
Article8. Sullivan LM, D'Agostino RB Sr. Robustness and power of analysis of covariance applied to ordinal scaled data as arising in randomized controlled trials. Stat Med. 2003; 22:1317–1334. PMID: 12687657.
Article9. Lee Y. What repeated measures analysis of variances really tells us. Korean J Anesthesiol. 2015; 68:340–345. PMID: 26257845.
Article10. Kim TK. Understanding one-way ANOVA using conceptual figures. Korean J Anesthesiol. 2017; 70:22–26. PMID: 28184262.
Article11. Bland JM, Altman DG. Multiple significance tests: the Bonferroni method. BMJ. 1995; 310:170. PMID: 7833759.12. Rothman KJ. No adjustments are needed for multiple comparisons. Epidemiology. 1990; 1:43–46. PMID: 2081237.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Development of Teaching Model for Medical Statistics in Medical School Students
- Nonparametric statistical tests for the continuous data: the basic concept and the practical use
- The Dental Hygienists' Perception of the National Practical Examination
- Redesigning an anesthesiology resident training program to improve practical procedure competency
- A Factor Analysis of the Perspectives on the Coping Strategies about Practical Stress in Nursing Student