Chonnam Med J.
2017 May;53(2):140-146. 10.4068/cmj.2017.53.2.140.
Clinical Significance of p53 Protein Expression, Beta-catenin Expression and HER2 Expression for Epstein-Barr Virus-associated Gastric Cancer
- Affiliations
-
- 1Department of Oncology/Hematology, Kyungpook National University Medical Center, Kyungpook National University Cancer Research Institute, Kyungpook National University School of Medicine, Daegu, Korea. jkk21c@knu.ac.kr, bwkang@knu.ac.kr
- 2Department of Pathology, Kyungpook National University Hospital, Kyungpook National University School of Medicine, Daegu, Korea.
- 3Department of Surgery, Kyungpook National University Hospital, Kyungpook National University School of Medicine, Daegu, Korea.
- KMID: 2379287
- DOI: http://doi.org/10.4068/cmj.2017.53.2.140
Abstract
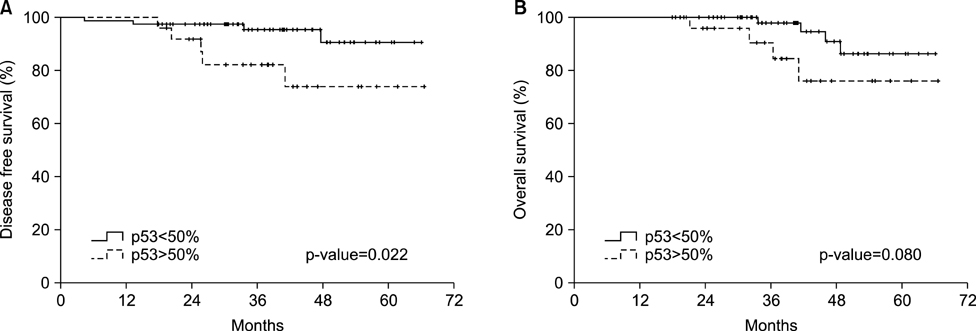
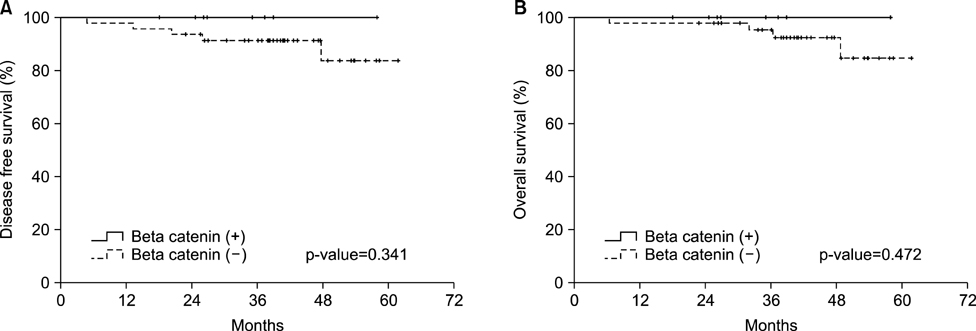
- This study assessed the expression of the p53 protein, beta-catenin, and HER2 and their prognostic implications in patients with EBV-associated gastric cancer (EBVaGC). After reviewing 1318 consecutive cases of surgically resected or endoscopic submucosal dissected gastric cancers, 117 patients were identified as EBV-positive using EBV-encoded RNA in-situ hybridization. The immunohistochemistry results were interpreted as follows: strong p53 nuclear expression in at least 50% of tumor nuclei was interpreted as a positive result, strong beta-catenin expression in at least 10% of cytoplasmic nuclei was interpreted as a positive result, and moderate or strong complete or basolateral membrane staining in 10% of tumor cells was interpreted as a positive result for HER2. Immunohistochemical staining for p53 was performed on tumor tissue from 105 patients, among whom 25 (23.8%) tested positive. Meanwhile, beta-catenin expression was positive in 10 patients (17.5%) and HER2 expression was positive in 8 patients (6.8%). The positive expression of p53 was significantly associated with a high T stage (p=0.006). More patients with lymph node metastasis were p53-positive (p=0.013). In the univariate analysis, the p53-positive patients showed significantly decreased disease-free survival (DFS) when compared with the p53-negative patients (p=0.022), although the p53 status was only marginally associated with overall survival (OS) (p=0.080). However, p53 expression showed no prognostic significance on DFS in the multivariate analysis. Moreover, beta-catenin and HER2 showed no association with DFS and OS in the survival analysis. The current study found a significant correlation between p53 expression and tumor progression and lymph node metastases in patients with EBVaGC.
Keyword
MeSH Terms
Figure
Reference
-
1. Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014; 513:202–209.2. Tsao SW, Tsang CM, To KF, Lo KW. The role of Epstein-Barr virus in epithelial malignancies. J Pathol. 2015; 235:323–333.
Article3. Gonzalez RS, Messing S, Tu X, McMahon LA, Whitney-Miller CL. Immunohistochemistry as a surrogate for molecular subtyping of gastric adenocarcinoma. Hum Pathol. 2016; 56:16–21.
Article4. Szkaradkiewicz A, Karpiński TM, Majewski J, Malinowska K, Goślińska-Kuźniarek O, Linke K. The participation of p53 and bcl-2 proteins in gastric carcinomas associated with Helicobacter-pylori and/or Epstein-Barr Virus (EBV). Pol J Microbiol. 2015; 64:211–216.
Article5. Kang BW, Seo AN, Yoon S, Bae HI, Jeon SW, Kwon OK, et al. Prognostic value of tumor-infiltrating lymphocytes in Epstein-Barr virus-associated gastric cancer. Ann Oncol. 2016; 27:494–501.
Article6. Belyi VA, Ak P, Markert E, Wang H, Hu W, Puzio-Kuter A, et al. The origins and evolution of the p53 family of genes. Cold Spring Harb Perspect Biol. 2010; 2:a001198.
Article7. Bellini MF, Cadamuro AC, Succi M, Proenca MA, Silva AE. Alterations of the TP53 gene in gastric and esophageal carcinogenesis. J Biomed Biotechnol. 2012; 2012:891961.8. Tahara T, Shibata T, Okamoto Y, Yamazaki J, Kawamura T, Horiguchi N, et al. Mutation spectrum of TP53 gene predicts clinicopathological features and survival of gastric cancer. Oncotarget. 2016; 7:42252–42260.
Article9. Lee JH, Kim SH, Han SH, An JS, Lee ES, Kim YS. Clinicopathological and molecular characteristics of Epstein-Barr virus-associated gastric carcinoma: a meta-analysis. J Gastroenterol Hepatol. 2009; 24:354–365.
Article10. Chiurillo MA. Role of the Wnt/β-catenin pathway in gastric cancer: an in-depth literature review. World J Exp Med. 2015; 5:84–102.
Article11. Staal FJ, Arens R. Wnt signaling as master regulator of T lymphocyte responses: implications for transplant therapy. Transplantation. 2016; DOI: 10.1097/TP.0000000000001393. [Epub ahead of print].12. Bang YJ, Van Cutsem E, Feyereislova A, Chung HC, Shen L, Sawaki A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010; 376:687–697.
Article13. Li Z, Lai Y, Sun L, Zhang X, Liu R, Feng G, et al. PD-L1 expression is associated with massive lymphocyte infiltration and histology in gastric cancer. Hum Pathol. 2016; 55:182–189.
Article14. Zhang YW, Zhao XX, Tan C, Zhang ZG, Jiang Y, Chen JN, et al. Epstein-Barr virus latent membrane protein 2A suppresses the expression of HER2 via a pathway involving TWIST and YB-1 in Epstein-Barr virus-associated gastric carcinomas. Oncotarget. 2015; 6:207–220.
Article15. Irkkan C, Balci S, Guler Tezel G, Akinci B, Yalcin B, Guler G. Comparison of clinicopathologic parameters and survivals between Epstein-Barr Virus-positive and Her2-positive gastric cancers. Appl Immunohistochem Mol Morphol. 2016; DOI: 10.1097/PAI.0000000000000353. [Epub ahead of print].
Article16. Sukawa Y, Yamamoto H, Nosho K, Kunimoto H, Suzuki H, Adachi Y, et al. Alterations in the human epidermal growth factor receptor 2-phosphatidylinositol 3-kinase-v-Akt pathway in gastric cancer. World J Gastroenterol. 2012; 18:6577–6586.
Article17. Takada K. Epstein-Barr virus and gastric carcinoma. Mol Pathol. 2000; 53:255–261.
Article18. Young LS, Rickinson AB. Epstein-Barr virus: 40 years on. Nat Rev Cancer. 2004; 4:757–768.
Article19. Yau TO, Tang CM, Yu J. Epigenetic dysregulation in Epstein-Barr virus-associated gastric carcinoma: disease and treatments. World J Gastroenterol. 2014; 20:6448–6456.
Article20. Giudice A, D'Arena G, Crispo A, Tecce MF, Nocerino F, Grimaldi M, et al. Role of viral miRNAs and epigenetic modifications in Epstein-Barr Virus-associated gastric carcinogenesis. Oxid Med Cell Longev. 2016; 2016:6021934.
Article21. Liu Y, Kulesz-Martin M. p53 protein at the hub of cellular DNA damage response pathways through sequence-specific and non-sequence-specific DNA binding. Carcinogenesis. 2001; 22:851–860.
Article22. Sivachandran N, Dawson CW, Young LS, Liu FF, Middeldorp J, Frappier L. Contributions of the Epstein-Barr virus EBNA1 protein to gastric carcinoma. J Virol. 2012; 86:60–68.
Article23. Shukla SK, Jha HC, El-Naccache DW, Robertson ES. An EBV recombinant deleted for residues 130-159 in EBNA3C can deregulate p53/Mdm2 and Cyclin D1/CDK6 which results in apoptosis and reduced cell proliferation. Oncotarget. 2016; 7:18116–18134.
Article24. Li L, Li W, Xiao L, Xu J, Chen X, Tang M, et al. Viral oncoprotein LMP1 disrupts p53-induced cell cycle arrest and apoptosis through modulating K63-linked ubiquitination of p53. Cell Cycle. 2012; 11:2327–2336.
Article25. Guo L, Tang M, Yang L, Xiao L, Bode AM, Li L, et al. Epstein-Barr virus oncoprotein LMP1 mediates survivin upregulation by p53 contributing to G1/S cell cycle progression in nasopharyngeal carcinoma. Int J Mol Med. 2012; 29:574–580.
Article26. Yang CF, Peng LX, Huang TJ, Yang GD, Chu QQ, Liang YY, et al. Cancer stem-like cell characteristics induced by EB virus-encoded LMP1 contribute to radioresistance in nasopharyngeal carcinoma by suppressing the p53-mediated apoptosis pathway. Cancer Lett. 2014; 344:260–271.
Article27. Chen HC, Chen HJ, Khan MA, Rao ZZ, Wan XX, Tan B, et al. Genetic mutations of p53 and k-ras in gastric carcinoma patients from Hunan, China. Tumour Biol. 2011; 32:367–373.
Article28. Imai S, Koizumi S, Sugiura M, Tokunaga M, Uemura Y, Yamamoto N, et al. Gastric carcinoma: monoclonal epithelial malignant cells expressing Epstein-Barr virus latent infection protein. Proc Natl Acad Sci U S A. 1994; 91:9131–9135.
Article29. Yildirim M, Kaya V, Demirpence O, Gunduz S, Bozcuk H. Prognostic significance of p53 in gastric cancer: a meta-analysis. Asian Pac J Cancer Prev. 2015; 16:327–332.
Article30. Begnami MD, Fregnani JH, Nonogaki S, Soares FA. Evaluation of cell cycle protein expression in gastric cancer: cyclin B1 expression and its prognostic implication. Hum Pathol. 2010; 41:1120–1127.
Article31. Goncalves AR, Carneiro AJ, Martins I, de Faria PA, Ferreira MA, de Mello EL, et al. Prognostic significance of p53 protein expression in early gastric cancer. Pathol Oncol Res. 2011; 17:349–355.
Article32. Ye YW, Zhang X, Zhou Y, Wu J, Zhao C, Yuan L, et al. The correlations between the expression of FGFR4 protein and clinicopathological parameters as well as prognosis of gastric cancer patients. J Surg Oncol. 2012; 106:872–879.
Article33. Lazăr D, Tăban S, Sporea I, Dema A, Cornianu M, Lazăr E, et al. The immunohistochemical expression of the p53-protein in gastric carcinomas. Correlation with clinicopathological factors and survival of patients. Rom J Morphol Embryol. 2010; 51:249–257.34. Murakami D, Tsujitani S, Osaki T, Saito H, Katano K, Tatebe S, et al. Expression of phosphorylated Akt (pAkt) in gastric carcinoma predicts prognosis and efficacy of chemotherapy. Gastric Cancer. 2007; 10:45–51.
Article35. Tsujitani S, Saito H, Wakatsuki T, Ikeguchi M, Shirabe K, Morita M, et al. Relationship between expression of apoptosis-related proteins and the efficacy of postoperative chemotherapy in patients with T3 gastric cancer. Surg Today. 2012; 42:225–232.
Article36. Scanlon CS, Van Tubergen EA, Inglehart RC, D'Silva NJ. Biomarkers of epithelial-mesenchymal transition in squamous cell carcinoma. J Dent Res. 2013; 92:114–121.
Article37. Schmalhofer O, Brabletz S, Brabletz T. E-cadherin, beta-catenin, and ZEB1 in malignant progression of cancer. Cancer Metastasis Rev. 2009; 28:151–166.
Article38. Behrens J. Cadherins and catenins: role in signal transduction and tumor progression. Cancer Metastasis Rev. 1999; 18:15–30.39. Li LF, Wei ZJ, Sun H, Jiang B. Abnormal β-catenin immunohistochemical expression as a prognostic factor in gastric cancer: a meta-analysis. World J Gastroenterol. 2014; 20:12313–12321.
Article40. Retterspitz MF, Monig SP, Schreckenberg S, Schneider PM, Holscher AH, Dienes HP. Expression of {beta}-catenin, MUC1 and c-met in diffuse-type gastric carcinomas: correlations with tumour progression and prognosis. Anticancer Res. 2010; 30:4635–4641.41. Yaguchi T, Goto Y, Kido K, Mochimaru H, Sakurai T, Tsukamoto N, et al. Immune suppression and resistance mediated by constitutive activation of Wnt/β-catenin signaling in human melanoma cells. J Immunol. 2012; 189:2110–2117.
Article42. Gravalos C, Jimeno A. HER2 in gastric cancer: a new prognostic factor and a novel therapeutic target. Ann Oncol. 2008; 19:1523–1529.
Article43. Hynes NE, Stern DF. The biology of erbB-2/neu/HER-2 and its role in cancer. Biochim Biophys Acta. 1994; 1198:165–184.44. Liang JW, Zhang JJ, Zhang T, Zheng ZC. Clinicopathological and prognostic significance of HER2 overexpression in gastric cancer: a meta-analysis of the literature. Tumour Biol. 2014; 35:4849–4858.
Article45. Lee HS, Chang MS, Yang HK, Lee BL, Kim WH. Epstein-barr virus-positive gastric carcinoma has a distinct protein expression profile in comparison with epstein-barr virus-negative carcinoma. Clin Cancer Res. 2004; 10:1698–1705.
Article46. Moritani S, Kushima R, Sugihara H, Hattori T. Phenotypic characteristics of Epstein-Barr-virus-associated gastric carcinomas. J Cancer Res Clin Oncol. 1996; 122:750–756.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Significance of Cyclin E, p53, E-cadherin, and beta-Catenin Expressions in Gastric Adenocarcinomas
- Expression of p53 Protein and Proliferating Cell Nuclear Antigen in Epstein - Barr Virus-associated Gastric Adenocarcinoma
- Expression of Beta-catenin and E-cadherin in Early Gastric Cancer: Correlation with Clinicopathologic Parameters
- beta-Catenin Expression in Gastric Carcinogenesis
- Expression of E-cadherin and beta-catenin in Human Adenocarcinoma, Adenoma, and Ulcer of Stomach