Allergic Rhinitis in Preschool Children and the Clinical Utility of FeNO
- Affiliations
-
- 1Department of Pediatrics, Childhood Asthma Atopy Center, Research Center for the Standardization of Allergic Diseases, University of Ulsan College of Medicine, Asan Medical Center, Seoul, Korea. sjhong@amc.seoul.kr
- 2Department of Pediatrics, Chonnam National University Hospital, Gwangju, Korea.
- 3Department of Pediatrics, Hallym University Sacred Heart Hospital, Hallym University College of Medicine, Anyang, Korea.
- 4Department of Pediatrics, Gyeongsang National University Changwon Hospital, Gyeongsang, Korea.
- 5Department of Pediatrics, CHA Bundang Medical Center, College of Medicine, CHA University, Seongnam, Korea.
- 6Department of Pediatrics, Dankook University Hospital, Cheonan, Korea.
- 7Department of Pediatrics, Seoul National University Bundang Hospital, Seongnam, Korea.
- 8Department of Pediatrics, Inje University Sanggye Paik Hospital, Inje University College of Medicine, Seoul, Korea.
- 9Department of Pediatrics, Gangneung Asan Hospital, Gangneung, Korea.
- 10Department of Pediatrics, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 11Division of Allergy and Chronic Respiratory Diseases, Center for Biomedical Sciences, Korea National Institute of Health, Korea Center for Disease Control and Prevention, Cheongju, Korea.
- KMID: 2378194
- DOI: http://doi.org/10.4168/aair.2017.9.4.314
Abstract
- PURPOSE
The nature of allergic rhinitis (AR) in preschool aged children remains incompletely characterized. This study aimed to investigate the prevalence of AR and its associated risk factors in preschool-aged children and to assess the clinical utility of fractional exhaled nitric oxide (FeNO).
METHODS
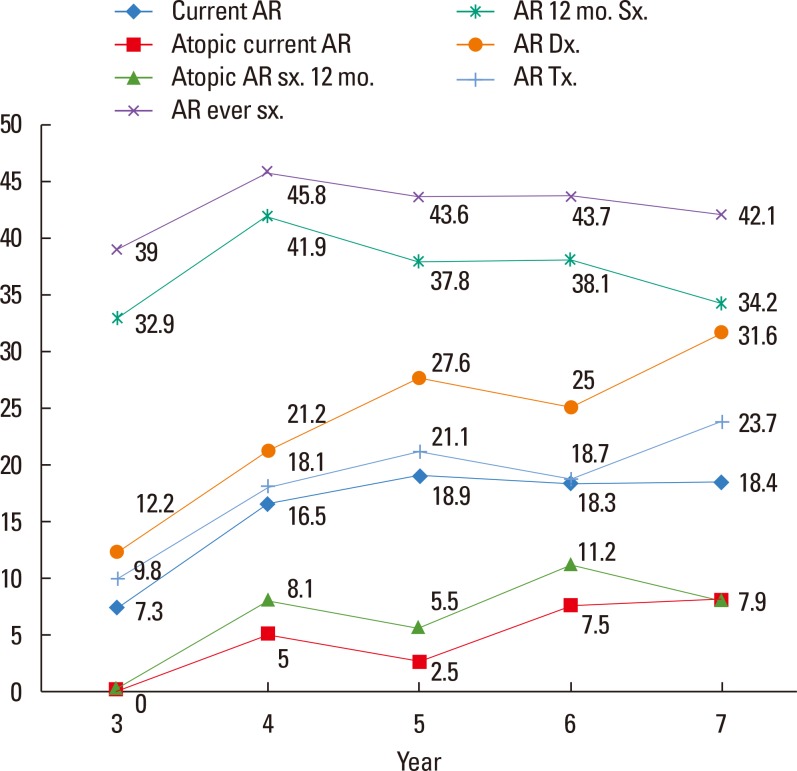
This general population-based, cross-sectional survey included 933 preschool-aged (3- to 7-year-old) children from Korea. Current AR was defined as having nasal symptoms within the last 12 months and physician-diagnosed AR.
RESULTS
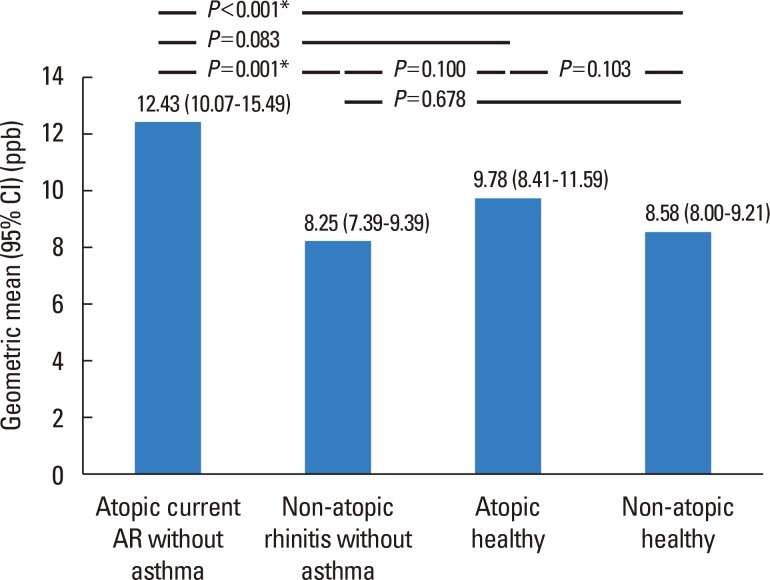
The prevalence of current AR in preschool children was 17.0% (156/919). Mold exposure (adjusted odds ratio [aOR], 1.67; 95% confidence interval [CI], 1.15-2.43) and the use of antibiotics (aOR, 1.97; 95% CI, 1.33-2.90) during infancy were associated with an increased risk of current AR, whereas having an older sibling (aOR, 0.52; 95% CI, 0.35-0.75) reduced the risk. Children with current atopic AR had significantly higher geometric mean levels of FeNO compared to those with non-atopic rhinitis (12.43; range of 1standard deviation [SD], 7.31-21.14 vs 8.25; range of 1SD, 5.62-12.10, P=0.001) or non-atopic healthy children (8.58; range of 1SD, 5.51-13.38, P<0.001). The FeNO levels were higher in children with current atopic AR compared with atopic healthy children (9.78; range of 1SD, 5.97-16.02, P=0.083).
CONCLUSIONS
Mold exposure and use of antibiotics during infancy increases the risk of current AR, whereas having an older sibling reduces it. Children with current atopic AR exhibit higher levels of FeNO compared with non-atopic rhinitis cases, suggesting that FeNO levels may be a useful discriminatory marker for subtypes of AR in preschool children.
Keyword
MeSH Terms
Figure
Cited by 6 articles
-
Effects of Antibiotics on the Development of Asthma and Other Allergic Diseases in Children and Adolescents
Do Hyun Kim, Kyungdo Han, Soo Whan Kim
Allergy Asthma Immunol Res. 2018;10(5):457-465. doi: 10.4168/aair.2018.10.5.457.Measurement of Exhaled Nitric Oxide in Children: A Comparison Between NObreath® and NIOX VERO® Analyzers
Yoko Inoue, Sakura Sato, Tetsuharu Manabe, Eishi Makita, Masako Chiyotanda, Kyohei Takahashi, Hitoshi Yamamoto, Noriyuki Yanagida, Motohiro Ebisawa
Allergy Asthma Immunol Res. 2018;10(5):478-489. doi: 10.4168/aair.2018.10.5.478.Effects of Vacuuming Mattresses on Allergic Rhinitis Symptoms in Children
You Hoon Jeon, Yong Ju Lee, Myung Hyun Sohn, Hae Ran Lee
Allergy Asthma Immunol Res. 2019;11(5):655-663. doi: 10.4168/aair.2019.11.5.655.Research on pediatric allergic rhinitis in Korea
Kyung Suk Lee, Yeong Ho Rha
Allergy Asthma Respir Dis. 2018;6(Suppl 1):S58-S65. doi: 10.4168/aard.2018.6.S1.S58.Chinese Herbal Medicine to Treat Allergic Rhinitis: Evidence From a Meta-Analysis
Xu Zhang, Feng Lan, Yuan Zhang, Luo Zhang
Allergy Asthma Immunol Res. 2018;10(1):34-42. doi: 10.4168/aair.2018.10.1.34.Indoor pet ownership in infancy is a risk factor for the development of sensitization to pets and asthma in childhood
Sungsu Jung, Soo Ran Noh, So-Yeon Lee, Jisun Yoon, Hyun-Ju Cho, Young-Ho Kim, Dong In Suh, Song-I Yang, Ji-won Kwon, Gwang Cheon Jang, Yong Han Sun, Sung-Il Woo, You-Sook Youn, Kang Seo Park, Eun Lee, Hwa Jin Cho, Myung-Hee Kook, Hye Ryoung Yi, Hai Lee Chung, Ja Hyeong Kim, Hyung Young Kim, Jin A Jung, Hyang-Ok Woo, Soo-Jong Hong
Allergy Asthma Respir Dis. 2019;7(2):99-105. doi: 10.4168/aard.2019.7.2.99.
Reference
-
1. Chen Y, Wong GW, Li J. Environmental exposure and genetic predisposition as risk factors for asthma in China. Allergy Asthma Immunol Res. 2016; 8:92–100. PMID: 26739401.
Article2. Yoon HI. Respiratory review of 2014: asthma. Tuberc Respir Dis (Seoul). 2014; 77:237–242. PMID: 25580139.
Article3. Meyers DA, Bleecker ER, Holloway JW, Holgate ST. Asthma genetics and personalised medicine. Lancet Respir Med. 2014; 2:405–415. PMID: 24794577.
Article4. Vahedi G, Richard AC, O'Shea JJ. Enhancing the understanding of asthma. Nat Immunol. 2014; 15:701–703. PMID: 25045871.
Article5. Tizaoui K, Kaabachi W, Hamzaoui K, Hamzaoui A. Association of single nucleotide polymorphisms in toll-like receptor genes with asthma risk: a systematic review and meta-analysis. Allergy Asthma Immunol Res. 2015; 7:130–140. PMID: 25729620.
Article6. Zhao CN, Fan Y, Huang JJ, Zhang HX, Gao T, Wang C, et al. The association of GSDMB and ORMDL3 gene polymorphisms with asthma: a meta-analysis. Allergy Asthma Immunol Res. 2015; 7:175–185. PMID: 25729625.7. Huang J, Wu S, Barrera J, Matthews K, Pan D. The hippo signaling pathway coordinately regulates cell proliferation and apoptosis by inactivating Yorkie, the Drosophila Homolog of YAP. Cell. 2005; 122:421–434. PMID: 16096061.
Article8. Angus L, Moleirinho S, Herron L, Sinha A, Zhang X, Niestrata M, et al. Willin/FRMD6 expression activates the hippo signaling pathway kinases in mammals and antagonizes oncogenic YAP. Oncogene. 2012; 31:238–250. PMID: 21666719.
Article9. Zhao B, Wei X, Li W, Udan RS, Yang Q, Kim J, et al. Inactivation of YAP oncoprotein by the hippo pathway is involved in cell contact inhibition and tissue growth control. Genes Dev. 2007; 21:2747–2761. PMID: 17974916.
Article10. Alarcón C, Zaromytidou AI, Xi Q, Gao S, Yu J, Fujisawa S, et al. Nuclear CDKs drive Smad transcriptional activation and turnover in BMP and TGFβ pathways. Cell. 2009; 139:757–769. PMID: 19914168.
Article11. Strano S, Munarriz E, Rossi M, Castagnoli L, Shaul Y, Sacchi A, et al. Physical interaction with Yes-associated protein enhances p73 transcriptional activity. J Biol Chem. 2001; 276:15164–15173. PMID: 11278685.
Article12. Vassilev A, Kaneko KJ, Shu H, Zhao Y, DePamphilis ML. TEAD/TEF transcription factors utilize the activation domain of YAP65, a Src/Yes-associated protein localized in the cytoplasm. Genes Dev. 2001; 15:1229–1241. PMID: 11358867.
Article13. Mahoney JE, Mori M, Szymaniak AD, Varelas X, Cardoso WV. The hippo pathway effector Yap controls patterning and differentiation of airway epithelial progenitors. Dev Cell. 2014; 30:137–150. PMID: 25043473.
Article14. Lange AW, Sridharan A, Xu Y, Stripp BR, Perl AK, Whitsett JA. Hippo/Yap signaling controls epithelial progenitor cell proliferation and differentiation in the embryonic and adult lung. J Mol Cell Biol. 2015; 7:35–47. PMID: 25480985.
Article15. Ungvári I, Hullám G, Antal P, Kiszel PS, Gézsi A, Hadadi É, et al. Evaluation of a partial genome screening of two asthma susceptibility regions using bayesian network based bayesian multilevel analysis of relevance. PLoS One. 2012; 7:e33573. PMID: 22432035.
Article16. Ungvári I, Hadadi E, Virág V, Bikov A, Nagy A, Semsei ÁF, et al. Implication of BIRC5 in asthma pathogenesis. Int Immunol. 2012; 24:293–301. PMID: 22336533.17. Wenzel SE. Asthma: defining of the persistent adult phenotypes. Lancet. 2006; 368:804–813. PMID: 16935691.
Article18. Antal P, Millinghoffer A, Hullám G, Hajós G, Sárközy P, Gézsi A, et al. Bayesian, systems-based, multilevel analysis of associations for complex phenotypes: from interpretation to decisions. In : Sinoquet C, Mourad R, editors. Probabilistic graphical models for genetics, genomics, and postgenomics. Oxford: Oxford University Press;2014. p. 318–362.19. Antal P, Millinghoffer A, Hullám G, Hajós G, Szalai C, Falus A. A bioinformatic platform for a bayesian, multiphased, multilevel analysis in immunogenomics. In : Flower DR, Davies M, Ranganathan S, editors. Bioinformatics for immunomics. New York (NY): Springer;2010. p. 157–185.20. Hullám G, Antal P, Szalai C, Falus A. Evaluation of a Bayesian model-based approach in GA studies. JMLR Workshop Conf Proc. 2010; 8:30–43.21. Antal P, Millinghoffer A, Hullám G, Szalai C, Falus AA. Bayesian view of challenges in feature selection: multilevel analysis, feature aggregation, multiple targets, redundancy and interaction. JMLR Workshop Conf Proc. 2008; 4:74–89.22. Gézsi A, Lautner-Csorba O, Erdélyi DJ, Hullám G, Antal P, Semsei ÁF, et al. In interaction with gender a common CYP3A4 polymorphism may influence the survival rate of chemotherapy for childhood acute lymphoblastic leukemia. Pharmacogenomics J. 2015; 15:241–247. PMID: 25266680.
Article23. Lautner-Csorba O, Gézsi A, Erdélyi DJ, Hullám G, Antal P, Semsei ÁF, et al. Roles of genetic polymorphisms in the folate pathway in childhood acute lymphoblastic leukemia evaluated by Bayesian relevance and effect size analysis. PLoS One. 2013; 8:e69843. PMID: 23940529.
Article24. Lautner-Csorba O, Gézsi A, Semsei AF, Antal P, Erdélyi DJ, Schermann G, et al. Candidate gene association study in pediatric acute lymphoblastic leukemia evaluated by Bayesian network based Bayesian multilevel analysis of relevance. BMC Med Genomics. 2012; 5:42. PMID: 23021489.
Article25. Chan TK, Loh XY, Peh HY, Tan WN, Tan WS, Li N, et al. House dust mite-induced asthma causes oxidative damage and DNA double-strand breaks in the lungs. J Allergy Clin Immunol. 2016; 138:84–96. PMID: 27157131.
Article26. Mao B, Gao Y, Bai Y, Yuan Z. Hippo signaling in stress response and homeostasis maintenance. Acta Biochim Biophys Sin (Shanghai). 2015; 47:2–9. PMID: 25476206.
Article27. Piccolo S, Dupont S, Cordenonsi M. The biology of YAP/TAZ: hippo signaling and beyond. Physiol Rev. 2014; 94:1287–1312. PMID: 25287865.
Article28. Yin MX, Zhang L. Hippo signaling in epithelial stem cells. Acta Biochim Biophys Sin (Shanghai). 2015; 47:39–45. PMID: 25476205.
Article29. Moleirinho S, Patrick C, Tilston-Lünel AM, Higginson JR, Angus L, Antkowiak M, et al. Willin, an upstream component of the hippo signaling pathway, orchestrates mammalian peripheral nerve fibroblasts. PLoS One. 2013; 8:e60028. PMID: 23593160.
Article30. Jeffery PK, Wardlaw AJ, Nelson FC, Collins JV, Kay AB. Bronchial biopsies in asthma. An ultrastructural, quantitative study and correlation with hyperreactivity. Am Rev Respir Dis. 1989; 140:1745–1753. PMID: 2690708.
Article31. Beasley R, Roche WR, Roberts JA, Holgate ST. Cellular events in the bronchi in mild asthma and after bronchial provocation. Am Rev Respir Dis. 1989; 139:806–817. PMID: 2923380.
Article32. Laitinen LA, Heino M, Laitinen A, Kava T, Haahtela T. Damage of the airway epithelium and bronchial reactivity in patients with asthma. Am Rev Respir Dis. 1985; 131:599–606. PMID: 3994155.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Exhaled NO: Determinants and Clinical Application in Children With Allergic Airway Disease
- Fractional exhaled nitric oxide in Korean children with allergic rhinitis
- Prevalence and comorbidity of allergic diseases in preschool children
- Role of Fractional Exhaled Nitric Oxide in Predicting Development of Allergic Rhinits in Children with Bronchial Asthma
- Clinical characteristics of allergic rhinitis and nonallergic rhinitis in Korean children