Korean J Gastroenterol.
2015 Apr;65(4):222-228. 10.4166/kjg.2015.65.4.222.
Enhanced Resolution of Eosinophilic Liver Abscess Associated with Toxocariasis by Albendazole Treatment
- Affiliations
-
- 1Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. drsmchoi@gmail.com
- KMID: 2373213
- DOI: http://doi.org/10.4166/kjg.2015.65.4.222
Abstract
- BACKGROUND/AIMS
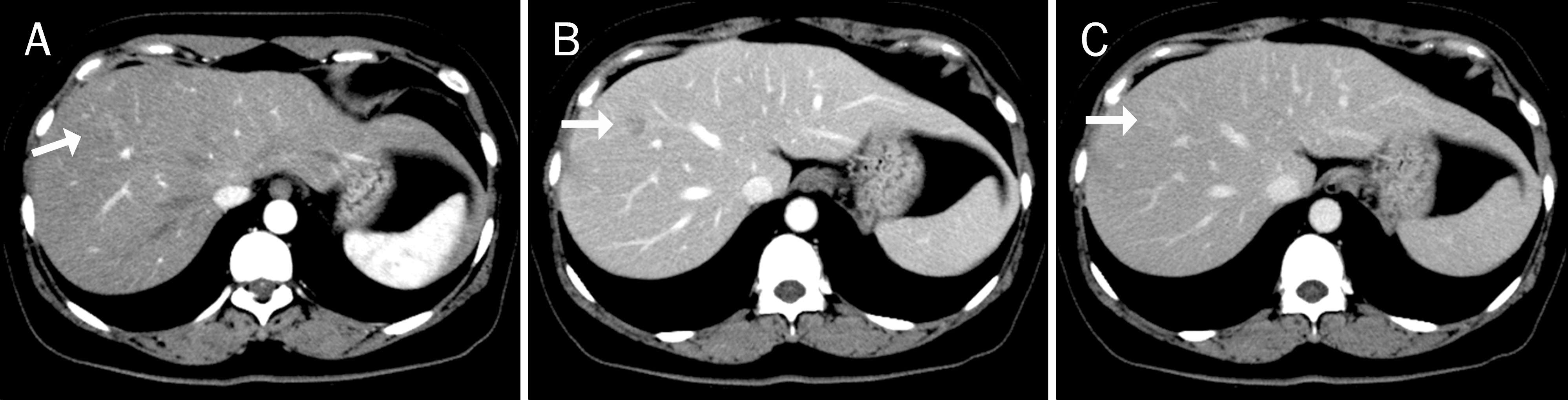
Visceral larva migrans, caused by Toxocara canis and Toxocara cati, has emerged as a significant cause of eosinophilic liver abscess (ELA). Differentiation of ELA associated with toxocariasis (ELA-T) from metastasis or primary liver malignancy is sometimes difficult. However, the role of albendazole treatment remains uncertain in this condition. The aim of this study was to evaluate whether albendazole can enhance the radiologic resolution of ELA-T.
METHODS
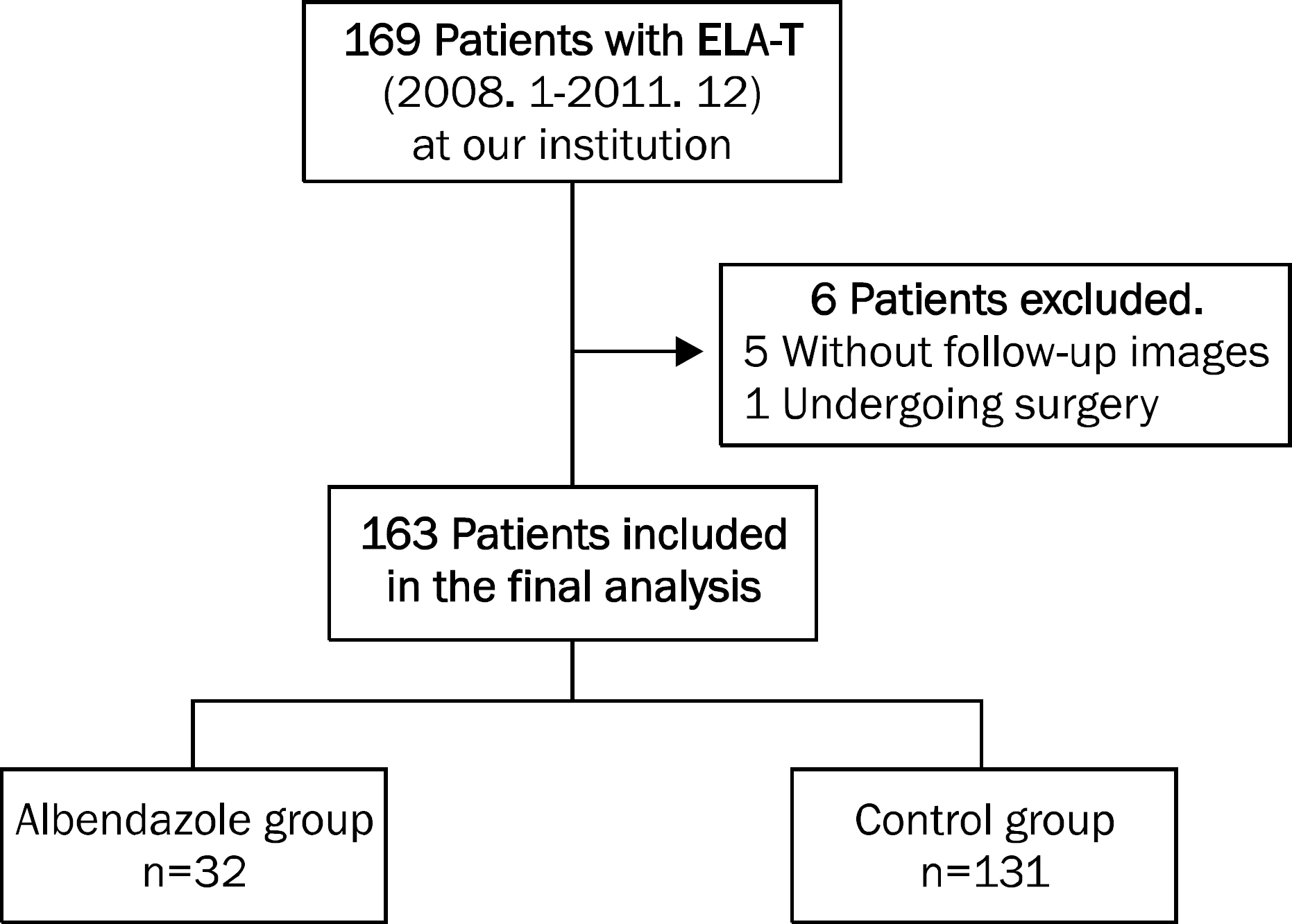
We retrospectively reviewed the medical records of the patients diagnosed with ELA-T at our institution between January 2008 and December 2011. ELA-T was diagnosed based on the imaging findings on computed tomography or magnetic resonance imaging and the presence of positive serum IgG antibody for Toxocara canis. Among a total of 163 patients, 32 patients received albendazole (albendazole group) and 131 did not (control group). Baseline characteristics and fate of liver nodules were compared between the two groups.
RESULTS
Baseline characteristics (age, sex, number and maximal size of lesions, eosinophil count) were similar between the two groups. Median duration for achieving radiologic resolution in the albendazole group was significantly shorter than in the control group (207 days [range 186-228] vs. 302 days [range 224-380], p=0.023). In Cox regression analysis of the cumulative rates of radiologic resolution, the hazard ratio for albendazole treatment was 1.99 (95% confidence interval, 1.22-3.23).
CONCLUSIONS
Radiologic resolution of ELA-T can be accelerated with albendazole treatment. Hence, inconvenience associated with long-term follow-up and unnecessary worries among patients can be eliminated with albendazole treatment.
Keyword
MeSH Terms
-
Adult
Aged
Aged, 80 and over
Albendazole/*therapeutic use
Animals
Antiprotozoal Agents/*therapeutic use
Eosinophils/*immunology
Female
Humans
Immunoglobulin G/blood
Larva Migrans, Visceral/*drug therapy/parasitology
Liver/enzymology/metabolism
Liver Abscess/*etiology
Magnetic Resonance Imaging
Male
Middle Aged
Proportional Hazards Models
Retrospective Studies
Tomography, X-Ray Computed
Toxocara canis/immunology/isolation & purification
Albendazole
Antiprotozoal Agents
Immunoglobulin G
Figure
Reference
-
References
1. Jang JK, Paik SW, Choi MS, et al. Clinical characteristics of 19 cases of biopsy-proven eosinophilic liver abscess: single center experience. Korean J Gastroenterol. 2001; 38:37–41.2. Jang HJ, Lee WJ, Lee SJ, Kim SH, Lim HK, Lim JH. Focal eosinophilic necrosis of the liver in patients with underlying gastric or colorectal cancer: CT differentiation from metastasis. Korean J Radiol. 2002; 3:240–244.
Article3. Yoo SY, Han JK, Kim YH, Kim TK, Choi BI, Han MC. Focal eosinophilic infiltration in the liver: radiologic findings and clinical course. Abdom Imaging. 2003; 28:326–332.
Article4. Kim YK, Kim CS, Moon WS, Cho BH, Lee SY, Lee JM. MRI findings of focal eosinophilic liver diseases. Am J Roentgenol. 2005; 184:1541–1548.
Article5. Chang S, Lim JH, Choi D, et al. Hepatic visceral larva migrans of Toxocara canis: CT and sonographic findings. Am J Roentgenol. 2006; 187:W622–W629.6. Lim JH, Lee KS. Eosinophilic infiltration in Korea: idiopathic? Korean J Radiol. 2006; 7:4–6.
Article7. Kim WH, Kim SH, Kim YH, et al. Fluorine-18-FDG PET findings of focal eosinophilic liver disease: correlation with CT and/or MRI, laboratory, and pathologic findings. Abdom Imaging. 2010; 35:437–446.
Article8. Lim JH. Toxocariasis of the liver: visceral larva migrans. Abdom Imaging. 2008; 33:151–156.
Article9. Hartleb M, Januszewski K. Severe hepatic involvement in visceral larva migrans. Eur J Gastroenterol Hepatol. 2001; 13:1245–1249.
Article10. Leone N, Baronio M, Todros L, et al. Hepatic involvement in larva migrans of Toxocara canis: report of a case with pathological and radiological findings. Dig Liver Dis. 2006; 38:511–514.
Article11. Hossack J, Ricketts P, Te HS, Hart J. A case of adult hepatic toxocariasis. Nat Clin Pract Gastroenterol Hepatol. 2008; 5:344–348.
Article12. Kaplan KJ, Goodman ZD, Ishak KG. Eosinophilic granuloma of the liver: a characteristic lesion with relationship to visceral larva migrans. Am J Surg Pathol. 2001; 25:1316–1321.13. Pawlowski Z. Toxocariasis in humans: clinical expression and treatment dilemma. J Helminthol. 2001; 75:299–305.
Article14. Kim YS, Park SJ, Kim HK, Park JM. A case of eosinophilic abscess mistaken for metastasis due to FDG uptake in PET-CT. Korean J Gastroenterol. 2009; 54:349–354.
Article15. Won JH, Kim MJ, Kim BM, et al. Focal eosinophilic infiltration of the liver: a mimick of hepatic metastasis. Abdom Imaging. 1999; 24:369–372.
Article16. Park S, Kim YS, Kim YJ, et al. Toxocariasis masquerading as liver and lung metastatic nodules in patents with gastrointestinal cancer: clinicopathologic study of five cases. Dig Dis Sci. 2012; 57:155–160.
Article17. Jin SY. Eosinophilic Liver Abscess. Korean J Hepatol. 2005; 11:176–179.18. Shin YM. Hepatic eosinophilic abscess presenting as a single nodular mass. Korean J Hepatol. 2010; 16:95–99.
Article19. Jung JK, Jung JT, Lee CH, Kim EY, Kwon JG, Kim BS. A case of hepatic abscess caused by toxocara. Korean J Hepatol. 2007; 13:409–413.
Article20. Despommier D. Toxocariasis: clinical aspects, epidemiology, medical ecology, and molecular aspects. Clin Microbiol Rev. 2003; 16:265–272.
Article21. Kwon NH, Oh MJ, Lee SP, Lee BJ, Choi DC. The prevalence and diagnostic value of toxocariasis in unknown eosinophilia. Ann Hematol. 2006; 85:233–238.
Article22. Magnaval JF, Glickman LT, Dorchies P, Morassin B. Highlights of human toxocariasis. Korean J Parasitol. 2001; 39:1–11.
Article23. Othman AA. Therapeutic battle against larval toxocariasis: are we still far behind? Acta Trop. 2012; 124:171–178.
Article24. Moore TA, McCarthy JS. Toxocariasis and larva migrans syndromes. Guerrant RL, Walker DH, Weller PF, editors. Tropical infectious diseases: principles, pathogens and practice. 3rd ed.Philadelphia: Elsevier;2011. p. 1209–1216.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical Evaluation of Toxocariasis Presenting as a Liver Abscess
- Cerebral Toxocariasis Presented With Seizure and Memory Disturbance
- A Case of Toxocariasis in a Patient with Cecal Cancer Mimicking Hepatic Metastasis
- A Case of Recurrent Toxocariasis Presenting With Urticaria
- Eosinophilic Liver Abscess Caused by Toxocara Canis