J Korean Orthop Assoc.
2016 Aug;51(4):287-293. 10.4055/jkoa.2016.51.4.287.
Hemodynamic Analysis in Patients Who Underwent Lumbar Spine Fusion Surgery without Blood Transfusion
- Affiliations
-
- 1Department of Orthopedic Surgery, Dong-A University College of Medicine, Busan, Korea. gylee@dau.ac.kr
- KMID: 2348634
- DOI: http://doi.org/10.4055/jkoa.2016.51.4.287
Abstract
- PURPOSE
Lumbar fusion surgery was performed on transfusion-free patients and hemodynamic changes were analyzed.
MATERIALS AND METHODS
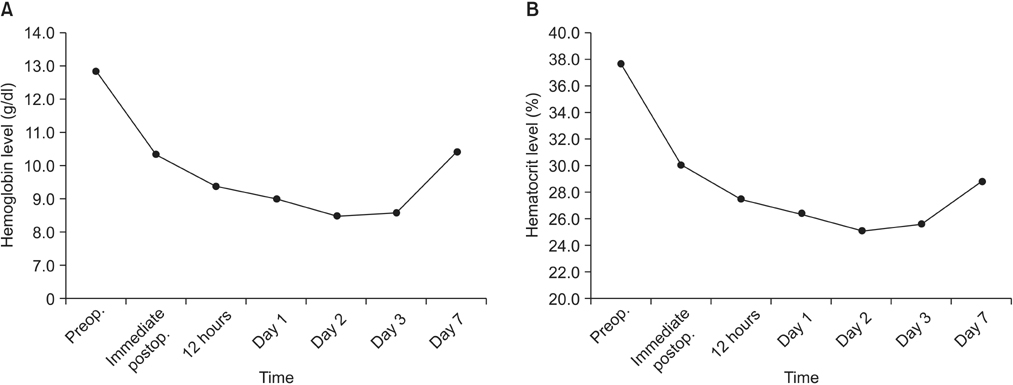
A total of 36 transfusion-free patients who had undergone lumbar fusion surgery using recombinant human erythropoietin (rHuEPO) before surgery from April 2007 to March 2014 were included in the study. Hemoglobin and hematocrit levels were measured before surgery, immediately after surgery, 12 hours after surgery, on day 1, day 2, day 3, and day 7. Changes in levels were investigated and the factors affecting the changes in hemoglobin levels were analyzed.
RESULTS
Changes in hemoglobin and hematocrit were -18.11% before surgery and -22.92% on day 7, respectively, and they tended to recover from day 2 after surgery. Depending on the patient's age, gender, body mass index, blood loss, and surgery method (presence of lumbar interbody fusion), the changes in hemoglobin level did not show statistically significant differences; however, significant differences were observed in the surgical time and extent of the operation.
CONCLUSION
Transfusion alternatives during lumbar spinal fusion are deemed safe methods, leading to good, postoperative hemodynamic outcomes. However, the surgical time and extent of the operation must be determined before surgery.
MeSH Terms
Figure
Reference
-
1. Urban MK, Beckman J, Gordon M, Urquhart B, Boachie-Adjei O. The efficacy of antifibrinolytics in the reduction of blood loss during complex adult reconstructive spine surgery. Spine (Phila Pa 1976). 2001; 26:1152–1156.
Article2. Vitale MG, Levy DE, Park MC, Choi H, Choe JC, Roye DP Jr. Quantifying risk of transfusion in children undergoing spine surgery. Spine J. 2002; 2:166–172.
Article3. Roye DP Jr. Recombinant human erythropoietin and blood management in pediatric spine surgery. Orthopedics. 1999; 22:s158–s160.
Article4. Shapiro GS, Boachie-Adjei O, Dhawlikar SH, Maier LS. The use of Epoetin alfa in complex spine deformity surgery. Spine (Phila Pa 1976). 2002; 27:2067–2071.
Article5. Stehling L, Luban NL, Anderson KC, et al. Guidelines for blood utilization review. Transfusion. 1994; 34:438–448.
Article6. Siller TA, Dickson JH, Erwin WD. Efficacy and cost considerations of intraoperative autologous transfusion in spinal fusion for idiopathic scoliosis with predeposited blood. Spine (Phila Pa 1976). 1996; 21:848–852.
Article7. Rosencher N, Ozier Y. Peri-operative use of EPO. Transfus Clin Biol. 2003; 10:159–164.8. Colomina MJ, Bagó J, Pellisé F, Godet C, Villanueva C. Preoperative erythropoietin in spine surgery. Eur Spine J. 2004; 13:Suppl 1. S40–S49.
Article9. Lee KY. Spine fusion surgery for the patient refusing allotransfusion. J Korean Soc Spine Surg. 2007; 14:87–95.
Article10. Goodnough LT, Strasburg D, Riddell J 4th, Verbrugge D, Wish J. Has recombinant human erythropoietin therapy minimized red-cell transfusions in hemodialysis patients? Clin Nephrol. 1994; 41:303–307.11. Mercuriali F, Inghilleri G. Blood transfusion in oncologic surgery: the role of recombinant human erythropoietin (rHuEPO). Tumori. 1998; 84:S3–S14.12. Rosencher N, Woimant G, Ozier Y, Conseiller C. Preoperative strategy for homologous blood salvage and peri-operative erythropoietin. Transfus Clin Biol. 1999; 6:370–379.13. Goodnough LT, Monk TG, Andriole GL. Erythropoietin therapy. N Engl J Med. 1997; 336:933–938.
Article14. Goldberg MA. Perioperative epoetin alfa increases red blood cell mass and reduces exposure to transfusions: results of randomized clinical trials. Semin Hematol. 1997; 34:3 Suppl 2. 41–47.
Article15. Stowell CP, Chandler H, Jové M, Guilfoyle M, Wacholtz MC. An open-label, randomized study to compare the safety and efficacy of perioperative epoetin alfa with preoperative autologous blood donation in total joint arthroplasty. Orthopedics. 1999; 22:s105–s112.
Article16. Goodnough LT. The safety of recombinant human erythropoietin therapy in autologous blood donors. J Lab Clin Med. 1994; 124:17–18.17. de Andrade JR, Frei D, Guilfoyle M. Integrated analysis of thrombotic/vascular event occurrence in epoetin alfa-treated patients undergoing major, elective orthopedic surgery. Orthopedics. 1999; 22:s113–s118.
Article18. Keating M, Faris P, Ritter M. Erythropoietin use. Orthopedics. 1998; 21:13–14.19. Nelson CL, Fontenot HJ, Flahiff C, Stewart J. An algorithm to optimize perioperative blood management in surgery. Clin Orthop Relat Res. 1998; 357:36–42.
Article20. Suh YS, Choi HS, Won SH, Kim MH, Chun DI, Nho JH. Hemodynamic analysis of patients who underwent arthroplasty without blood transfusion. J Korean Orthop Assoc. 2010; 45:456–463.
Article21. Nelson CL, Stewart JG. Primary and revision total hip replacement in patients who are Jehovah's Witnesses. Clin Orthop Relat Res. 1999; 369:251–261.
Article22. Lisander B, Jonsson R, Nordwall A. Combination of blood-saving methods decreases homologous blood requirements in scoliosis surgery. Anaesth Intensive Care. 1996; 24:555–558.
Article23. Neilipovitz DT, Murto K, Hall L, Barrowman NJ, Splinter WM. A randomized trial of tranexamic acid to reduce blood transfusion for scoliosis surgery. Anesth Analg. 2001; 93:82–87.
Article24. Olsfanger D, Jedeikin R, Metser U, Nusbacher J, Gepstein R. Acute normovolaemic haemodilution and idiopathic scoliosis surgery: effects on homologous blood requirements. Anaesth Intensive Care. 1993; 21:429–431.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical Experiences of Predeposited and Salvaged Autologous Blood Transfusion in Spine Fusion
- Usefulness of Posterolateral Fusion of Lumbar Spine with Allogeneic Bone (Tutoplast)
- Serotonergic Antidepressants Are Associated with Increased Blood Loss and Risk for Transfusion in Single-Level Lumbar Fusion Surgery
- Principles of Lumbar Spine Stabilization
- Biportal Endoscopic Transforaminal Lumbar Interbody Fusion with Percutaneous Instrumentation: A Technical Note