Tuberc Respir Dis.
2014 Jun;76(6):284-288.
p53 Expression in a Malignant Mesothelioma Patient during Seven-Year Follow-up
- Affiliations
-
- 1Division of Allergy and Respiratory Medicine, Department of Internal Medicine, Soonchunhyang University Hospital, Soonchunhyang University College of Medicine, Seoul, Korea. uhs@schmc.ac.kr
- 2Department of Pathology, Soonchunhyang University Hospital, Soonchunhyang University College of Medicine, Seoul, Korea.
Abstract
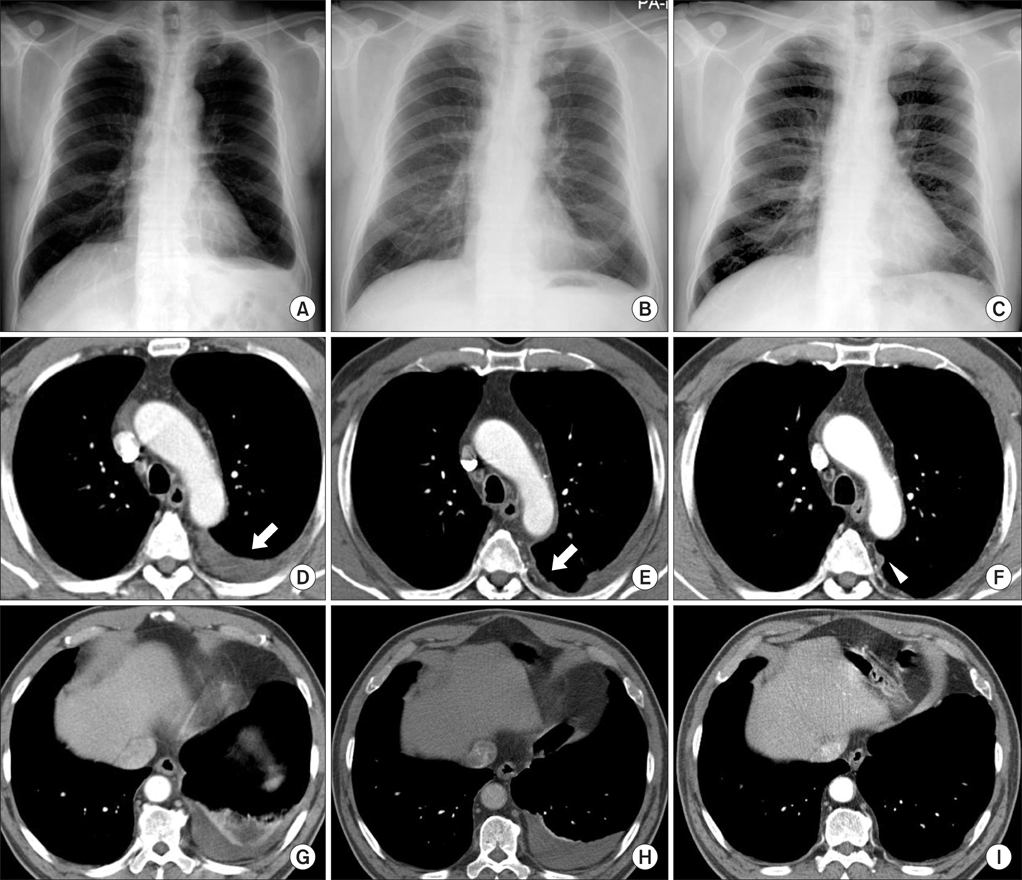
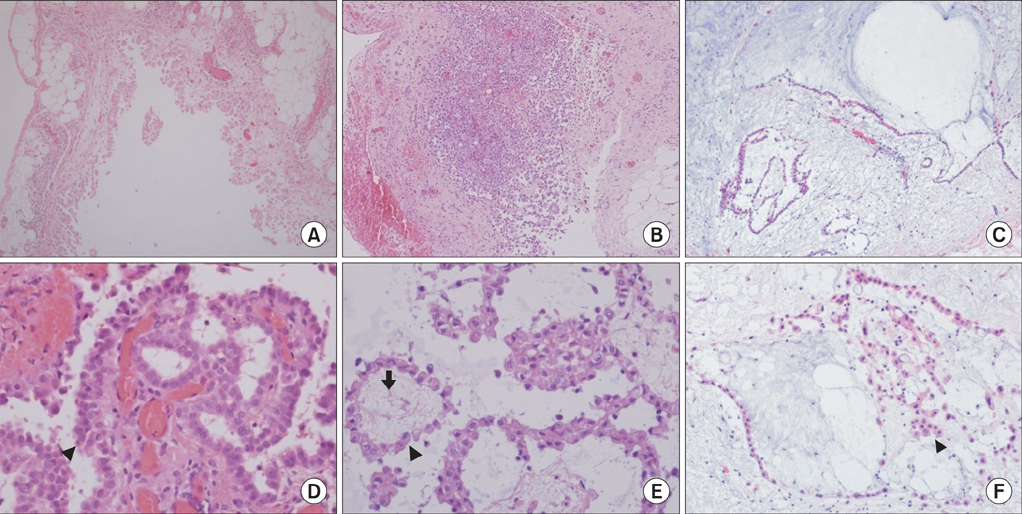
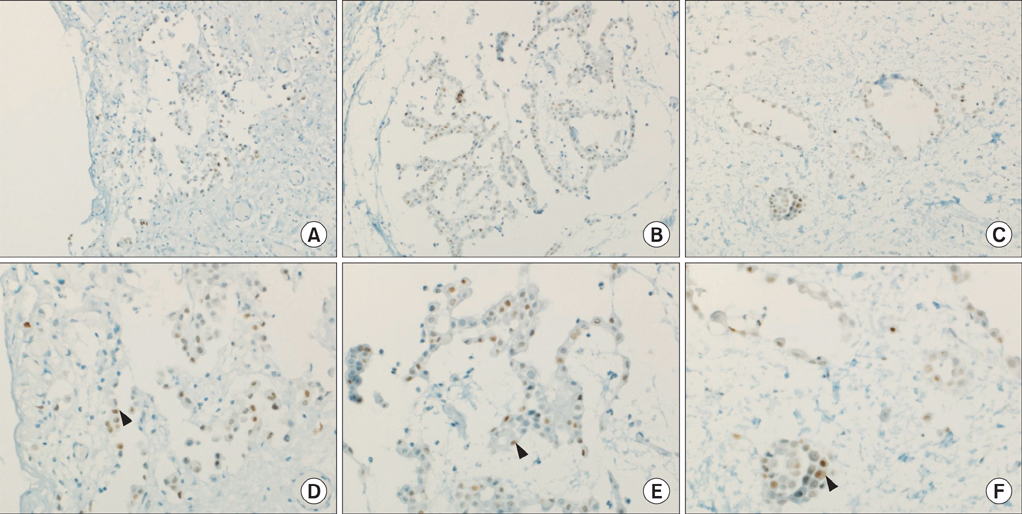
- Malignant mesothelioma (MM) is the aggressive tumor of serosal surfaces. There are crude pathogenetic results regarding the biology of MM. Coordinated upregulations of p53 gene expression are shown in malignancies. We believed that there are changes in the p53 expression with transformation from reactive hyperplasia to MM. A 65-year-old male was admitted the hospital because of left pleuritic chest pains in 2004. Chest computed tomography (CT) results showed left pleural effusions with loculation and pleural thickening. Pathologic findings revealed reactive mesothelial hyperplasia. In 2008, the patient again felt left pleuritic chest pains. Chest CT showed progressive thickening of the left pleura. Pathologic diagnosis was atypical mesothelial hyperplasia. In 2011, chest CT showed progressive thickening of his left pleura. He was diagnosed with well-differentiated papillary mesothelioma. Serial change was analyzed by immunohistochemical staining for p53 of pleural tissues. There were no remarkable changes in p53 expressions during the transformation to MM.
Keyword
MeSH Terms
Figure
Reference
-
1. Robinson BW, Lake RA. Advances in malignant mesothelioma. N Engl J Med. 2005; 353:1591–1603.2. Carbone M, Pass HI, Rizzo P, Marinetti M, Di Muzio M, Mew DJ, et al. Simian virus 40-like DNA sequences in human pleural mesothelioma. Oncogene. 1994; 9:1781–1790.3. Hasteh F, Lin GY, Weidner N, Michael CW. The use of immunohistochemistry to distinguish reactive mesothelial cells from malignant mesothelioma in cytologic effusions. Cancer Cytopathol. 2010; 118:90–96.4. Husain AN, Colby TV, Ordonez NG, Krausz T, Borczuk A, Cagle PT, et al. Guidelines for pathologic diagnosis of malignant mesothelioma: a consensus statement from the International Mesothelioma Interest Group. Arch Pathol Lab Med. 2009; 133:1317–1331.5. British Thoracic Society Standards of Care Committee. BTS statement on malignant mesothelioma in the UK, 2007. Thorax. 2007; 62:Suppl 2. ii1–ii19.6. Attanoos RL, Griffin A, Gibbs AR. The use of immunohistochemistry in distinguishing reactive from neoplastic mesothelium: a novel use for desmin and comparative evaluation with epithelial membrane antigen, p53, platelet-derived growth factor-receptor, P-glycoprotein and Bcl-2. Histopathology. 2003; 43:231–238.7. King J, Thatcher N, Pickering C, Hasleton P. Sensitivity and specificity of immunohistochemical antibodies used to distinguish between benign and malignant pleural disease: a systematic review of published reports. Histopathology. 2006; 49:561–568.8. O'Kane SL, Pound RJ, Campbell A, Chaudhuri N, Lind MJ, Cawkwell L. Expression of bcl-2 family members in malignant pleural mesothelioma. Acta Oncol. 2006; 45:449–453.9. Cagle PT, Brown RW, Lebovitz RM. p53 immunostaining in the differentiation of reactive processes from malignancy in pleural biopsy specimens. Hum Pathol. 1994; 25:443–448.10. Attanoos RL, Gibbs AR. Pathology of malignant mesothelioma. Histopathology. 1997; 30:403–418.11. Scurry J, Duggan MA. Malignant mesothelioma eight years after a diagnosis of atypical mesothelial hyperplasia. J Clin Pathol. 1999; 52:535–537.12. Angelopoulou K, Diamandis EP, Sutherland DJ, Kellen JA, Bunting PS. Prevalence of serum antibodies against the p53 tumor suppressor gene protein in various cancers. Int J Cancer. 1994; 58:480–487.13. Lubin R, Schlichtholz B, Teillaud JL, Garay E, Bussel A, Wild CP. p53 antibodies in patients with various types of cancer: assay, identification, and characterization. Clin Cancer Res. 1995; 1:1463–1469.14. Esposito V, Baldi A, De Luca A, Claudio PP, Signoriello G, Bolognese A, et al. p53 immunostaining in differential diagnosis of pleural mesothelial proliferations. Anticancer Res. 1997; 17:733–736.15. Zekri AR, Bahnassy AA, Mohamed WS, Hassan N, Abdel-Rahman AR, El-Kassem FA, et al. Evaluation of simian virus-40 as a biological prognostic factor in Egyptian patients with malignant pleural mesothelioma. Pathol Int. 2007; 57:493–501.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A case of primary malignant mesothelioma of tunica vaginalis testis
- Patterns of p53 expression in phyllodes tumors of the breast: an immunohistochemical study
- A Case of Diffuse Malignant Mesothelioma of the Peritoneum
- Case of Malignant Mesothelioma of the Peritoneum and Pleura
- Two cases of malignant mesothelioma of the peritoneum and pericardium