Korean J Physiol Pharmacol.
2008 Dec;12(6):299-306. 10.4196/kjpp.2008.12.6.299.
Long-term Follow-up of Cutaneous Hypersensitivity in Rats with a Spinal Cord Contusion
- Affiliations
-
- 1Department of Physiology and Neuroscience Research Center, Korea University College of Medicine, Seoul 136-705, Korea. ywyoon@korea.ac.kr
- 2Department of Physical Therapy, Korea University College of Health Science, Seoul 136-705, Korea.
- KMID: 2285356
- DOI: http://doi.org/10.4196/kjpp.2008.12.6.299
Abstract
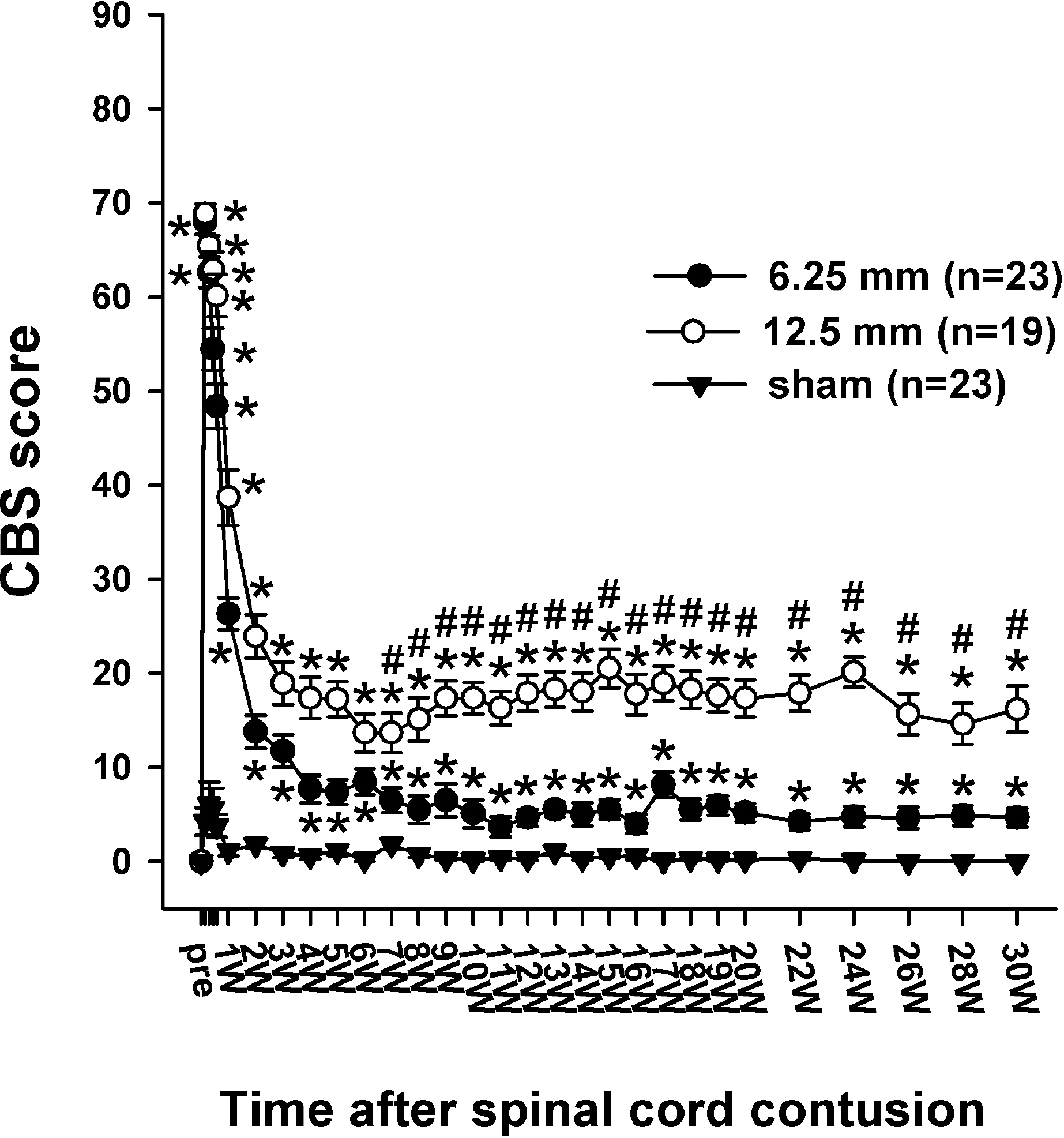
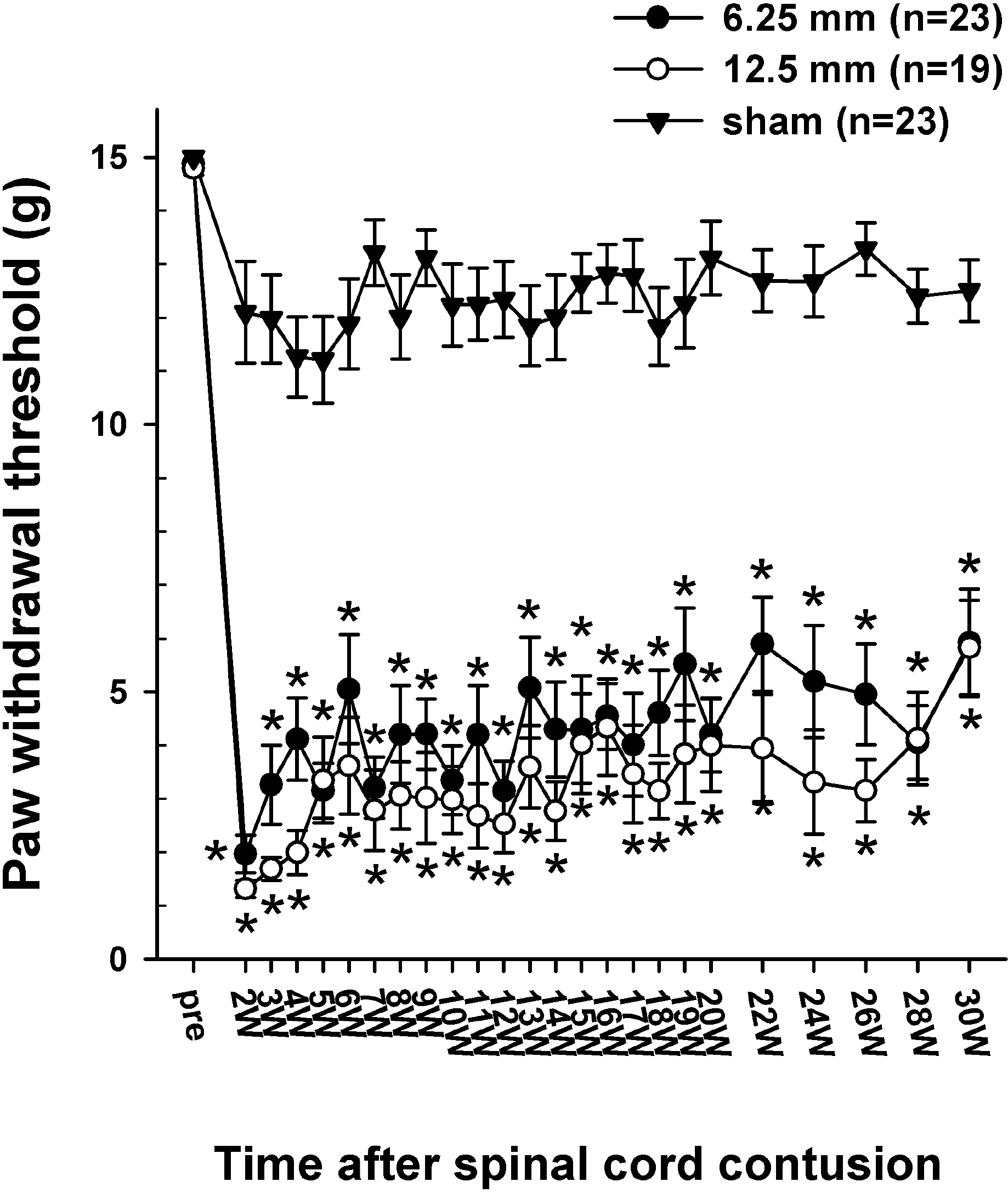
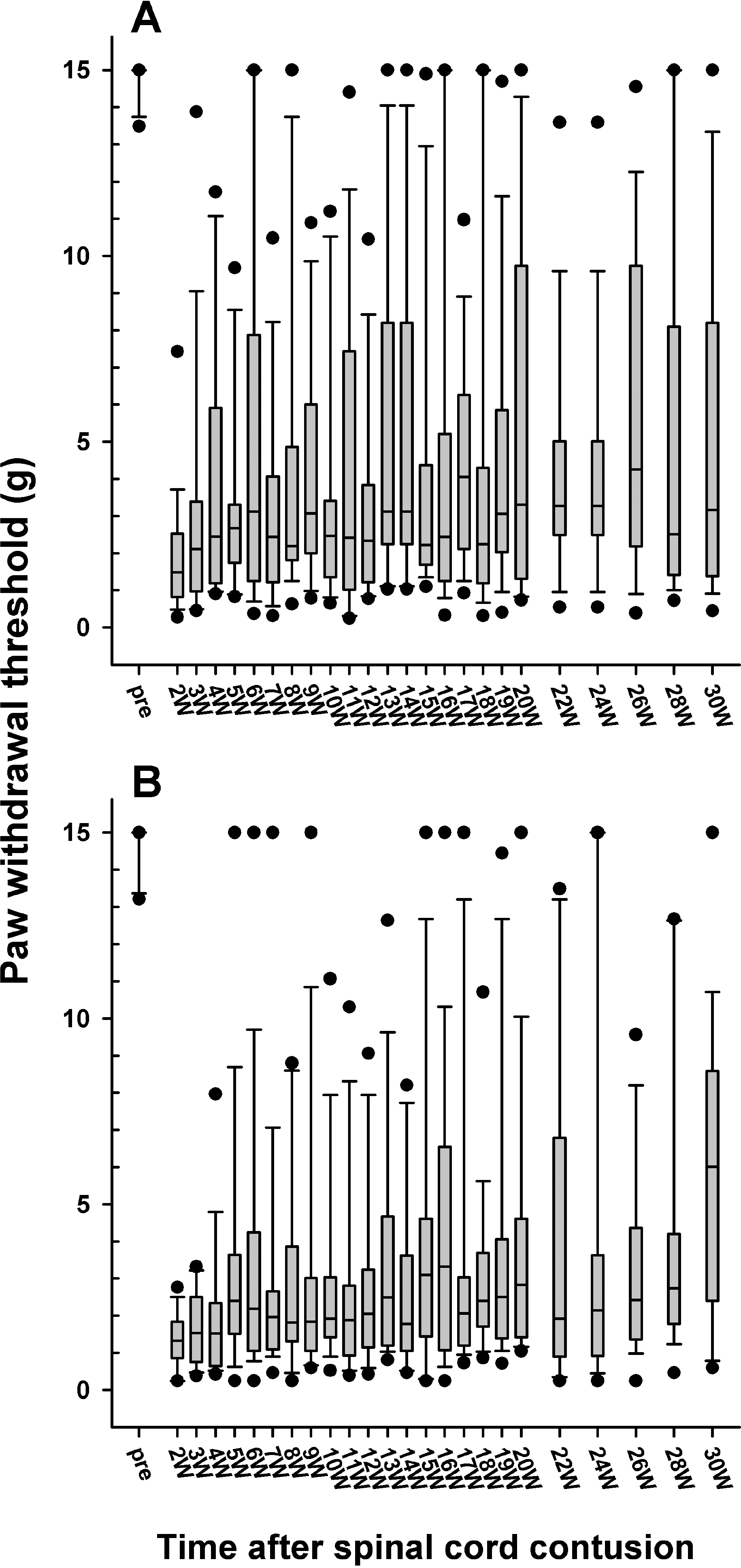
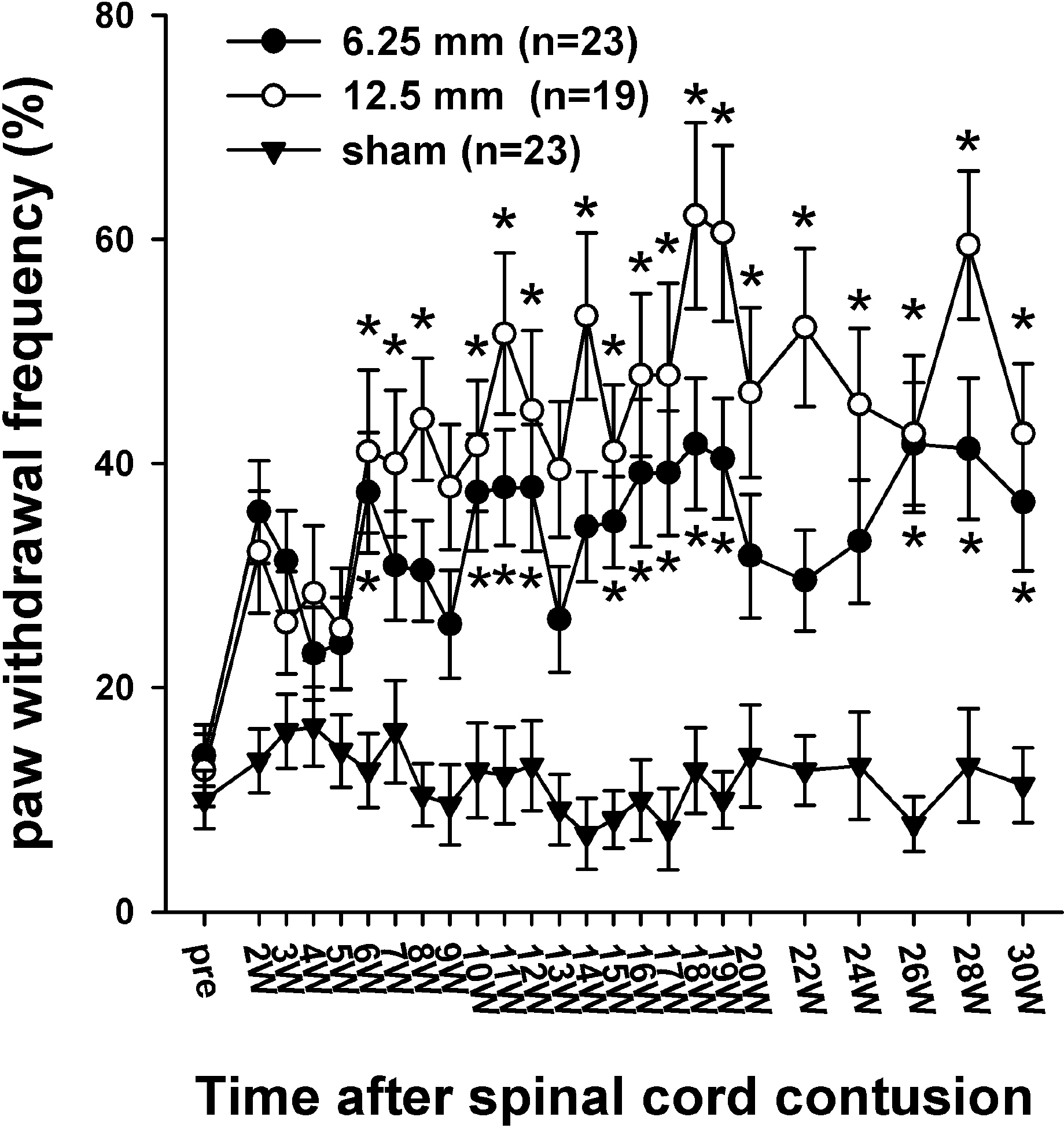
- Sometimes, spinal cord injury (SCI) results in various chronic neuropathic pain syndromes that occur diffusely below the level of the injury. It has been reported that behavioral signs of neuropathic pain are expressed in the animal models of contusive SCI. However, the observation period is relatively short considering the natural course of pain in human SCI patients. Therefore, this study was undertaken to examine the time course of mechanical and cold allodynia in the hindpaw after a spinal cord contusion in rats for a long period of time (30 weeks). The hindpaw withdrawal threshold to mechanical stimulation was applied to the plantar surface of the hindpaw, and the withdrawal frequency to the application of acetone was measured before and after a spinal contusion. The spinal cord contusion was produced by dropping a 10 g weight from a 6.25 and 12.5 mm height using a NYU impactor. After the injury, rats showed a decreased withdrawal threshold to von Frey stimulation, indicating the development of mechanical allodynia which persisted for 30 weeks. The withdrawal threshold between the two experimental groups was similar. The response frequencies to acetone increased after the SCI, but they were developed slowly. Cold allodynia persisted for 30 weeks in 12.5 mm group. The sham animals did not show any significant behavioral changes. These results provide behavioral evidence to indicate that the below-level pain was well developed and maintained in the contusion model for a long time, suggesting a model suitable for pain research, especially in the late stage of SCI or for long term effects of analgesic intervention.
Keyword
MeSH Terms
Figure
Reference
-
Basso DM., Beattie MS., Bresnahan JC. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma. 12:1–21. 1995.
ArticleBasso DM., Beattie MS., Bresnahan JC. Graded histological and locomotor outcomes after spinal cord contusion using the NYU weight-drop device versus transection. Exp Neurol. 139:244–256. 1996a.
ArticleBasso DM., Beattie MS., Bresnahan JC., Anderson DK., Faden AI., Gruner JA., Holford TR., Hsu CY., Noble LJ., Nockels R., Perot PL., Salzman SK., Young W. MASCIS evaluation of open field locomotor scores: effects of experience and teamwork on reliability. Multicenter Animal Spinal Cord Injury Study. J Neurotrauma. 13:343–359. 1996b.Behrmann DL., Bresnahan JC., Beattie MS. Modeling of acute spinal cord injury in the rat: neuroprotection and enhanced recovery with methylprednisolone, U-74006F and YM-14673. Exp Neurol. 126:61–75. 1994.
ArticleBeric A. Central pain: “new” syndromes and their evaluation. Muscle Nerve. 16:1017–1024. 1993.
ArticleBeric A., Dimitrijevic MR., Lindblom U. Central dysesthesia syndrome in spinal cord injury patients. Pain. 34:109–116. 1988.Berrocal Y., Pearse DD., Singh A., Andrade CM., McBroom JS., Puentes R., Eaton MJ. Social and environmental enrichment improves sensory and motor recovery after severe contusive spinal cord injury in the rat. J Neurotrauma. 24:1761–1772. 2007.
ArticleBoivie J. On central pain and central pain mechanisms. Pain. 38:121–122. 1989.
ArticleBrewer KL., Yezierski RP. Effects of adrenal medullary transplants on pain-related behaviors following excitotoxic spinal cord injury. Brain Res. 798:83–92. 1998.
ArticleBunge RP., Puckett WR., Hiester ED. Observations on the pathology of several types of human spinal cord injury, with emphasis on the astrocyte response to penetrating injuries. Adv Neurol. 72:305–315. 1997.Chaplan SR., Bach FW., Pogrel JW., Chung JM., Yaksh TL. Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods. 53:55–63. 1994.
ArticleChoi Y., Yoon YW., Na HS., Kim SH., Chung JM. Behavioral signs of ongoing pain and cold allodynia in a rat model of neuropathic pain. Pain. 59:369–376. 1994.Christensen MD., Everhart AW., Pickelman JT., Hulsebosch CE. Mechanical and thermal allodynia in chronic central pain following spinal cord injury. Pain. 68:97–107. 1996.
ArticleConstantini S., Young W. The effects of methylprednisolone and the ganglioside GM1 on acute spinal cord injury in rats. J Neurosurg. 80:97–111. 1994.
ArticleCruz-Almeida Y., Martinez-Arizala A., Widerstrom-Noga EG. Chronicity of pain associated with spinal cord injury: A longitudinal analysis. J Rehabil Res Dev. 42:585–594. 2005.
ArticleDavidoff G., Roth EJ., Casey KL. Clinical characteristics of central (dysesthetic) pain in spinal cord injury patients. Casey KL, editor. ed,. Pain and central nervous system disease: The central pain syndromes. New York: Raven;p. p. 77–83. 1991.Decosterd I., Woolf CJ. Spared nerve injury: an animal model of persistent peripheral neuropathic pain. Pain. 87:149–158. 2000.
ArticleFinnerup NB., Jensen TS. Spinal cord injury pain-mechanisms and treatment. Eur J Neurol. 11:73–82. 2004.Gale K., Kerasidis H., Wrathall JR. Spinal cord contusion in the rat: behavioral analysis of functional neurologic impairment. Exp Neurol. 88:123–134. 1985.
ArticleGruner JA. A monitored contusion model of spinal cord injury in the rat. J Neurotrauma. 9:123–126. 1992.
ArticleHao JX., Xu XJ., Aldskogius H., Seiger A., Wiesenfeld-Hallin Z. Photochemically induced transient spinal ischemia induces behavioral hypersensitivity to mechanical and cold stimuli, but not to noxious-heat stimuli, in the rat. Exp Neurol. 118:187–194. 1992.
ArticleHao JX., Yu W., Xu XJ., Wiesenfeld-Hallin Z. Capsaicin-sensitive afferents mediate chronic cold, but not mechanical, allodynialike behavior in spinally injured rats. Brain Res. 722:177–180. 1996.
ArticleHook MA., Liu GT., Washburn SN., Ferguson AR., Bopp AC., Huie JR., Grau JW. The impact of morphine after a spinal cord injury. Behav Brain Res. 179:281–293. 2007.
ArticleKim J., Yoon YW., Hong SK., Na HS. Cold and mechanical allodynia in both hindpaws and tail following thoracic spinal cord hemisection in rats: time courses and their correlates. Neurosci Lett. 343:200–204. 2003.
ArticleLee KH., Suh-Kim H., Choi JS., Jeun SS., Kim EJ., Kim SS., Yoon DH., Lee BH. Human mesenchymal stem cell transplantation promotes functional recovery following acute spinal cord injury in rats. Acta Neurobiol Exp (Wars). 67:13–22. 2007.Lee SH., Jung J., Kim J., Kim K., Hong SK., Yoon YW. Behavioral signs of pain in a rat spinal contusion model. Program No. 981.4. 2004. Abstract Viewer/Itinerary Planner. Washington, DC: Society for Neuroscience.Lindsey AE., LoVerso RL., Tovar CA., Hill CE., Beattie MS., Bresnahan JC. An analysis of changes in sensory thresholds to mild tactile and cold stimuli after experimental spinal cord injury in the rat. Neurorehabil Neural Repair. 14:287–300. 2000.Metz GA., Curt A., van de MH., Klusman I., Schwab ME., Dietz V. Validation of the weight-drop contusion model in rats: a comparative study of human spinal cord injury. J Neurotrauma. 17:1–17. 2000.
ArticleRamer MS., Harper GP., Bradbury EJ. Progress in spinal cord research – a refined strategy for the International Spinal Research Trust. Spinal Cord. 38:449–472. 2000.
ArticleRosenzweig ES., McDonald JW. Rodent models for treatment of spinal cord injury: research trends and progress toward useful repair. Curr Opin Neurol. 17:121–131. 2004.
ArticleSiddall PJ., McClelland JM., Rutkowski SB., Cousins MJ. A longitudinal study of the prevalence and characteristics of pain in the first 5 years following spinal cord injury. Pain. 103:249–257. 2003.
ArticleStormer S., Gerner HJ., Gruninger W., Metzmacher K., Follinger S., Wienke C., Aldinger W., Walker N., Zimmermann M., Paeslack V. Chronic pain/dysaesthesiae in spinal cord injury patients: results of a multicentre study. Spinal Cord. 35:446–455. 1997.
ArticleTasker RR., Loeser JD. Central Pain States. Loeser JD, editor. ed,. Bonica's Management of Pain. Philadelphia: Lippincott Williams Wilkins;p. p. 433–457. 2001.Vierck CJ Jr. Siddall P, Yezierski RP. Pain following spinal cord injury: animal models and mechanistic studies. Pain. 89:1–5. 2000.Voda J., Hama A., Sagen J. FK506 reduces the severity of cutaneous hypersensitivity in rats with a spinal cord contusion. Neurosci Res. 58:95–99. 2007.
ArticleWasner G., Lee BB., Engel S., McLachlan E. Residual spinothalamic tract pathways predict development of central pain after spinal cord injury. Brain. 131:2387–2400. 2008.
ArticleYezierski RP. Pain following spinal cord injury: the clinical problem and experimental studies. Pain. 68:185–194. 1996.
ArticleYezierski RP., Liu S., Ruenes GL., Kajander KJ., Brewer KL. Excitotoxic spinal cord injury: behavioral and morphological characteristics of a central pain model. Pain. 75:141–155. 1998.
ArticleYoon YW., Dong H., Arends JJ., Jacquin MF. Mechanical and cold allodynia in a rat spinal cord contusion model. Somatosens Mot Res. 21:25–31. 2004.
ArticleZhao P., Waxman SG., Hains BC. Extracellular signal-regulated kinase-regulated microglia-neuron signaling by prostaglandin E2 contributes to pain after spinal cord injury. J Neurosci. 27:2357–2368. 2007.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Animals models of spinal cord contusion injury
- Paraplegia Following Spinal Cord Contusion from an Indirect Gunshot Injury
- Clinical Features of Central Neuropathic Pain after Contusive Spinal Cord Injury in Rats
- Changes of motor evoked potentials and spinal cord evoked potentials following spinal cord injury in rats
- Therapeutic Effects of Naloxone in Experimental Spinal Cord Injury