Allergy Asthma Immunol Res.
2014 May;6(3):242-251. 10.4168/aair.2014.6.3.242.
Small Airway Impairment and Bronchial Hyperresponsiveness in Asthma Onset
- Affiliations
-
- 1Unit of Pneumology, "Misericordia" Hospital, Grosseto, Italy. bsposat@tin.it
- 2Institute of Clinical Physiology, National Research Council (CNR), Pisa, Italy.
- 3Unit of Pneumology and UTIP, "S.Donato" Hospital, Arezzo, Italy.
- KMID: 2260687
- DOI: http://doi.org/10.4168/aair.2014.6.3.242
Abstract
- PURPOSE
Our study tried to find a relationship between baseline FEF25-75% and airway hyperresponsiveness (AHR) and whether a greater FEF25-75% impairment may be a marker of a more severe hyperresponsiveness in subjects with normal FEV1 and FEV1/FVC and suggestive asthma symptoms. Besides, we tried to asses a FEF25-75% cut-off value to identify hyper-reactive subjects.
METHODS
4,172 subjects (2,042 M; mean age: 38.3+/-14.9; mean FEV1 % predicted: 100.5+/-12.7 and FEV1/FVC: 85.4+/-6.8) were examined after performing a methacholine (Mch) test. All subjects reported a symptom onset within 3 years before the test. Subjects with PD20<400 or >400 microg were arbitrarily considered affected by moderate/severe and borderline AHR, respectively.
RESULTS
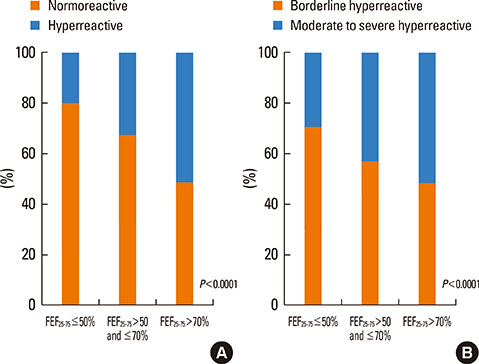
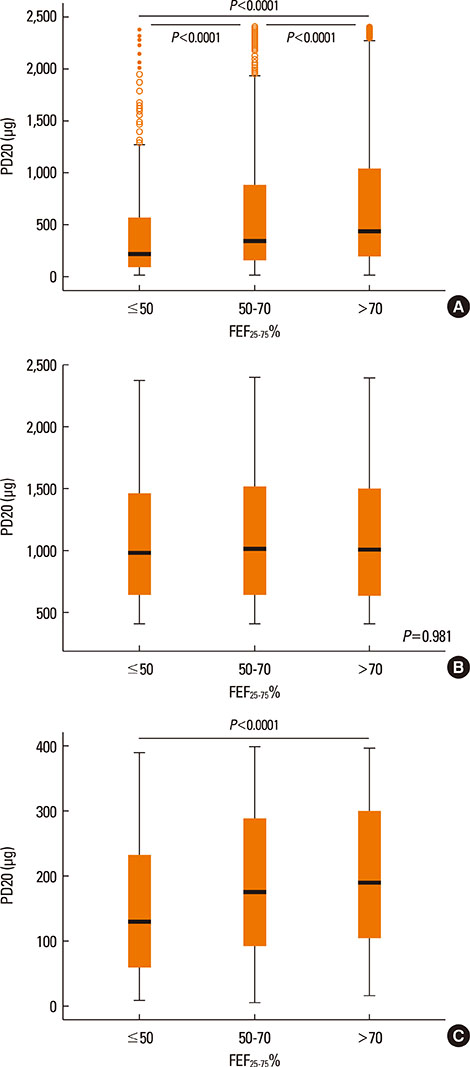
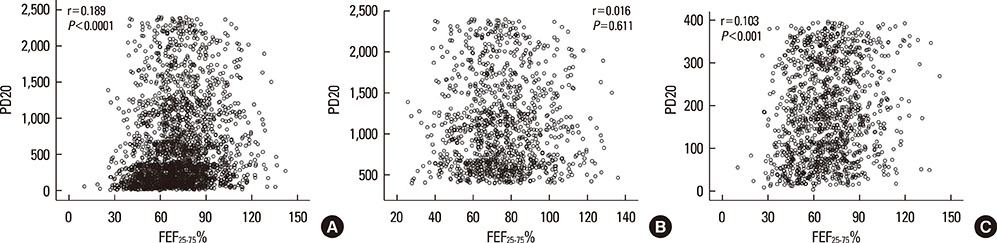
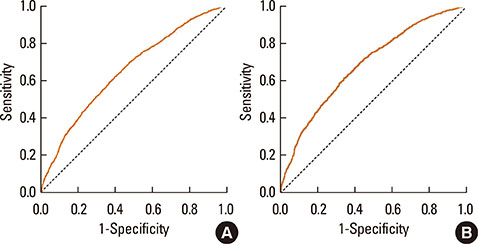
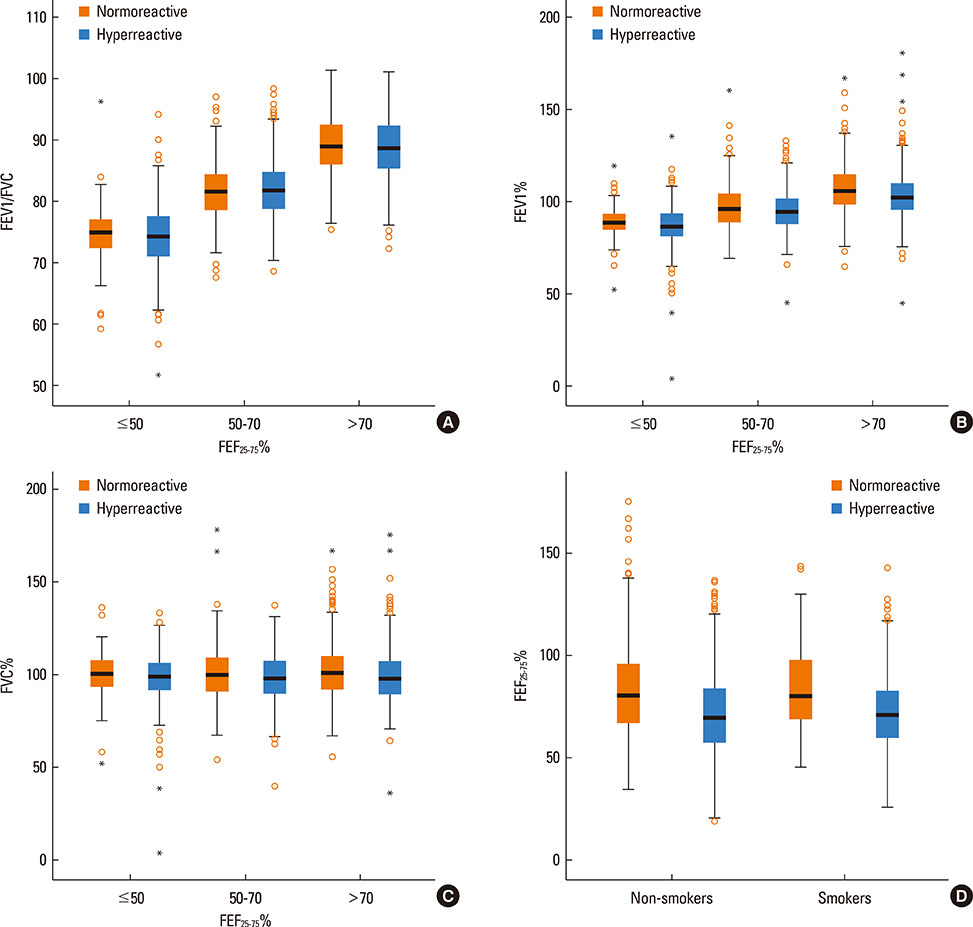
PD20 values were 213 (IQR:86-557), 340 (IQR:157-872) and 433 (IQR:196-1032) microg in subjects with baseline FEF25-75< or =50%, FEF25-75 between 50 and 70% and FEF25-75>70% respectively (P<0.0001). Only in moderate/severe hyper-reactive subjects (excluded borderlines), PD20 was lower in the FEF25-75< or =50% subgroup than in the 1 with FEF25-75>70%. The hyperreactive subjects percentage, was higher in those with FEF25-75< or =50% and lower in those with FEF25-75>70% (P<0.0001). FEF25-75<50% (compared to FEF25-75>70%) was a higher AHR risk factor, especially in subjects with moderate/severe AHR (OR: 2.18 [IQR:1.41-3.37]; P<0.0001). Thresholds yielding the highest combined sensitivity/specificity for FEF25-75% were 75.19 (area under curve [AUC]: 0.653) and 74.95 (AUC:0.688) in subjects with PD20<2,400 and <400 microg respectively. FEV1, FVC, and FEV1/FVC measured in subjects with different FEF25-75< or =50%, FEF25-75>50 and < or =70% or FEF25-75>70% levels were similar both in normoreactive and hyperreactive subjects.
CONCLUSIONS
At asthma onset, reduced baseline FEF25-75 values with normal FEV1 and FEV1/FVC may predict AHR. Detectable predictive cut-off values do not exist because even normoreactive subjects can show lower FEF25-75 values. Furthermore, a greater FEF25-75 reduction may be associated to a more severe AHR, suggesting a possible FEF25-75 role in the management of asthma when FEV1 and FEV1/FVC are normal.
Figure
Cited by 1 articles
-
Value of Exhaled Nitric Oxide and FEF25–75 in Identifying Factors Associated With Chronic Cough in Allergic Rhinitis
Xiaofang Liu, Xiangdong Wang, Xiujuan Yao, Yuhong Wang, Yongchang Sun, Luo Zhang
Allergy Asthma Immunol Res. 2019;11(6):830-845. doi: 10.4168/aair.2019.11.6.830.
Reference
-
1. Global Initiative for Asthma (US). Global strategy for asthma management and prevention 2011 [Internet]. Global Initiative for Asthma;2011. cited 2012 Jun 10. Available from: http://www.ginasthma.org/GINA-Report,-Global-Strategy-for-Asthma-Management-and-Prevention.2. van den Berge M, ten Hacken NH, Cohen J, Douma WR, Postma DS. Small airway disease in asthma and COPD: clinical implications. Chest. 2011; 139:412–423.3. Burgel PR. The role of small airways in obstructive airway diseases. Eur Respir Rev. 2011; 20:23–33.4. Ohwada A, Takahashi K. Concave pattern of a maximal expiratory flow-volume curve: a sign of airflow limitation in adult bronchial asthma. Pulm Med. 2012; 2012:797495.5. Zapletal A, Hladíková M, Chalupová J, Svobodová T, Vávrová V. Area under the maximum expiratory flow-volume curve--a sensitive parameter in the evaluation of airway patency. Respiration. 2008; 75:40–47.6. Ciprandi G, Cirillo I, Vizzaccaro A, Monardo M, Tosca MA. Early bronchial airflow impairment in patients with persistent allergic rhinitis and bronchial hyperreactivity. Respir Med. 2005; 99:1606–1612.7. Mariotta S, Sposato B, Ricci A, Bruno P, Aquilini M, Mannino F. Reversibility test in the early stages of bronchial asthma. J Asthma. 2005; 42:487–491.8. Sposato B, Mariotta S, Ricci A. When should a reversibility test be performed on patients with early stages of asthma and normal spirometry? J Asthma. 2008; 45:479–483.9. Park SH, Lee SY, Kang SM, Seon CS, Kim HK, Lee BH, Lee JH, Kim SH. Prediction of brobchodilator response by using FEF25~75% in adult patient with a normal spirometry result. Tuberc Respir Dis. 2011; 71:188–194.10. Ciprandi G, Capasso M, Leonardi S, Lionetti E, La Rosa M, Salpietro C, Miraglia Del Giudice M, Cirillo I, Tosca M, Marseglia GL. Impaired FEF25-75 values may predict bronchial reversibility in allergic children with rhinitis or asthma. J Biol Regul Homeost Agents. 2012; 26:S19–S25.11. U.S. Department of Health and Human Services. National Heart, Lung and Blood Institute (US). Expert panel report 3: guidelines for the diagnosis and mangement of asthma [Internet]. Bethesda (MD): National Heart, Lung and Blood Institute Health Information Center;2007. cited 2012 Jun. Available from: http://www.nhlbi.nih.gov/guidelines/asthma/asthgdln.pdf.12. Pellegrino R, Viegi G, Brusasco V, Crapo RO, Burgos F, Casaburi R, Coates A, van der Grinten CP, Gustafsson P, Hankinson J, Jensen R, Johnson DC, MacIntyre N, McKay R, Miller MR, Navajas D, Pedersen OF, Wanger J. Interpretative strategies for lung function tests. Eur Respir J. 2005; 26:948–968.13. Standardized lung function testing. Official statement of the European Respiratory Society. Eur Respir J Suppl. 1993; 16:1–100.14. Alberts WM, Ferris MC, Brooks SM, Goldman AL. The FEF25-75% and the clinical diagnosis of asthma. Ann Allergy. 1994; 73:221–225.15. Cirillo I, Klersy C, Marseglia GL, Vizzaccaro A, Pallestrini E, Tosca M, Ciprandi G. Role of FEF25%-75% as a predictor of bronchial hyperreactivity in allergic patients. Ann Allergy Asthma Immunol. 2006; 96:692–700.16. Ciprandi G, Tosca MA, Capasso M. Forced expiratory flow between 25 and 75% of vital capacity might be a predictive factor for bronchial hyperreactivity in children with allergic rhinitis, asthma, or both. Allergy Asthma Proc. 2011; 32:e22–e28.17. Ciprandi G, Tosca MA, Castellazzi AM, Cairello F, Salpietro C, Arrigo T, Miraglia Del Giudice M. FEF(25-75) might be a predictive factor for bronchial inflammation and bronchial hyperreactivity in adolescents with allergic rhinitis. Int J Immunopathol Pharmacol. 2011; 24:17–20.18. Mendonça NT, Kenyon J, LaPrad AS, Syeda SN, O'Connor GT, Lutchen KR. Airway resistance at maximum inhalation as a marker of asthma and airway hyperresponsiveness. Respir Res. 2011; 12:96.19. Parker AL, Abu-Hijleh M, McCool FD. Ratio between forced expiratory flow between 25% and 75% of vital capacity and FVC is a determinant of airway reactivity and sensitivity to methacholine. Chest. 2003; 124:63–69.20. Rao DR, Gaffin JM, Baxi SN, Sheehan WJ, Hoffman EB, Phipatanakul W. The utility of forced expiratory flow between 25% and 75% of vital capacity in predicting childhood asthma morbidity and severity. J Asthma. 2012; 49:586–592.21. del Giudice MM, Brunese FP, Piacentini GL, Pedullà M, Capristo C, Decimo F, Capristo AF. Fractional exhaled nitric oxide (FENO), lung function and airway hyperresponsiveness in naive atopic asthmatic children. J Asthma. 2004; 41:759–765.22. Spallarossa D, Battistini E, Silvestri M, Sabatini F, Fregonese L, Brazzola G, Rossi GA. Steroid-naive adolescents with mild intermittent allergic asthma have airway hyperresponsiveness and elevated exhaled nitric oxide levels. J Asthma. 2003; 40:301–310.23. Nishio K, Odajima H, Motomura C, Nakao F, Nishima S. Exhaled nitric oxide and exercise-induced bronchospasm assessed by FEV1, FEF25-75% in childhood asthma. J Asthma. 2007; 44:475–478.24. Yoon JY, Woo SI, Kim H, Sun YH, Hahn YS. Fractional exhaled nitric oxide and forced expiratory flow between 25% and 75% of vital capacity in children with controlled asthma. Korean J Pediatr. 2012; 55:330–336.25. Cole TJ, Bellizzi MC, Flegal KM, Dietz WH. Establishing a standard definition for child overweight and obesity worldwide: international survey. BMJ. 2000; 320:1240–1243.26. Crapo RO, Casaburi R, Coates AL, Enright PL, Hankinson JL, Irvin CG, MacIntyre NR, McKay RT, Wanger JS, Anderson SD, Cockcroft DW, Fish JE, Sterk PJ. Guidelines for methacholine and exercise challenge testing-1999. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. Am J Respir Crit Care Med. 2000; 161:309–329.27. Sposato B, Scalese M, Pammolli A, Scala R, Naldi M. Seasons can influence the results of the methacholine challenge test. Ann Thorac Med. 2012; 7:61–68.28. Marcon A, Cerveri I, Wjst M, Antó J, Heinrich J, Janson C, Jarvis D, Leynaert B, Probst-Hensch N, Svanes C, Toren K, Burney P, de Marco R. Can an airway challenge test predict respiratory diseases? A population-based international study. J Allergy Clin Immunol. 2014; 133:104–110.e4.29. De Marco R, Accordini S, Marcon A, Cerveri I, Antó JM, Gislason T, Heinrich J, Janson C, Jarvis D, Kuenzli N, Leynaert B, Sunyer J, Svanes C, Wjst M, Burney P. European Community Respiratory Health Survey (ECRHS). Risk factors for chronic obstructive pulmonary disease in a European cohort of young adults. Am J Respir Crit Care Med. 2011; 183:891–897.30. De Marco R, Accordini S, Cerveri I, Corsico A, Antó JM, Künzli N, Janson C, Sunyer J, Jarvis D, Chinn S, Vermeire P, Svanes C, Ackermann-Liebrich U, Gislason T, Heinrich J, Leynaert B, Neukirch F, Schouten JP, Wjst M, Burney P. Incidence of chronic obstructive pulmonary disease in a cohort of young adults according to the presence of chronic cough and phlegm. Am J Respir Crit Care Med. 2007; 175:32–39.31. Løkke A, Lange P, Scharling H, Fabricius P, Vestbo J. Developing COPD: a 25 year follow up study of the general population. Thorax. 2006; 61:935–939.32. Gibson PG, Simpson JL. The overlap syndrome of asthma and COPD: what are its features and how important is it? Thorax. 2009; 64:728–735.33. Bjermer L. History and future perspectives of treating asthma as a systemic and small airways disease. Respir Med. 2001; 95:703–719.34. Hamid Q. Pathogenesis of small airways in asthma. Respiration. 2012; 84:4–11.35. Hamid QA. Peripheral inflammation is more important than central inflammation. Respir Med. 1997; 91:Suppl A. 11–12.36. Fujisawa T, Yasui H, Akamatsu T, Hashimoto D, Enomoto N, Inui N, Nakamura Y, Maekawa M, Suda T, Chida K. Alveolar nitric oxide concentration reflects peripheral airway obstruction in stable asthma. Respirology. 2013; 18:522–527.37. Dupont LJ, Rochette F, Demedts MG, Verleden GM. Exhaled nitric oxide correlates with airway hyperresponsiveness in steroid-naive patients with mild asthma. Am J Respir Crit Care Med. 1998; 157:894–898.38. Reid DW, Johns DP, Feltis B, Ward C, Walters EH. Exhaled nitric oxide continues to reflect airway hyperresponsiveness and disease activity in inhaled corticosteroid-treated adult asthmatic patients. Respirology. 2003; 8:479–486.39. Langley SJ, Goldthorpe S, Custovic A, Woodcock A. Relationship among pulmonary function, bronchial reactivity, and exhaled nitric oxide in a large group of asthmatic patients. Ann Allergy Asthma Immunol. 2003; 91:398–404.40. Ciprandi G, Tosca MA, Capasso M. Exhaled nitric oxide in children with allergic rhinitis and/or asthma: a relationship with bronchial hyperreactivity. J Asthma. 2010; 47:1142–1147.41. Jatakanon A, Lim S, Kharitonov SA, Chung KF, Barnes PJ. Correlation between exhaled nitric oxide, sputum eosinophils, and methacholine responsiveness in patients with mild asthma. Thorax. 1998; 53:91–95.42. Currie GP, Jackson CM, Lee DK, Lipworth BJ. Determinants of airway hyperresponsiveness in mild asthma. Ann Allergy Asthma Immunol. 2003; 90:560–563.43. van der Wiel E, ten Hacken NH, Postma DS, van den Berge M. Small-airways dysfunction associates with respiratory symptoms and clinical features of asthma: a systematic review. J Allergy Clin Immunol. 2013; 131:646–657.44. Brannan JD, Lougheed MD. Airway hyperresponsiveness in asthma: mechanisms, clinical significance, and treatment. Front Physiol. 2012; 3:460.45. Allen ND, Davis BE, Hurst TS, Cockcroft DW. Difference between dosimeter and tidal breathing methacholine challenge: contributions of dose and deep inspiration bronchoprotection. Chest. 2005; 128:4018–4023.46. Cockcroft DW, Davis BE. The bronchoprotective effect of inhaling methacholine by using total lung capacity inspirations has a marked influence on the interpretation of the test result. J Allergy Clin Immunol. 2006; 117:1244–1248.47. DeMeo DL, Carey VJ, Chapman HA, Reilly JJ, Ginns LC, Speizer FE, Weiss ST, Silverman EK. Familial aggregation of FEF(25-75) and FEF(25-75)/FVC in families with severe, early onset COPD. Thorax. 2004; 59:396–400.48. Kornmann O, Beeh KM, Beier J, Geis UP, Ksoll M, Buhl R. Global Initiative for Obstructive Lung Disease. Newly diagnosed chronic obstructive pulmonary disease. Clinical features and distribution of the novel stages of the Global Initiative for Obstructive Lung Disease. Respiration. 2003; 70:67–75.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effect of Lower Airway Disease on the Severity of Chronic Rhinosinusitis
- The Effectiveness of Pimecrolimus in Airway Inflammation and Bronchial Hyperresponsiveness in Murine Asthma Model
- Characteristics and Follow-Up of Bronchial Hyperresponsiveness in Cough Variant Asthma
- Relationship Between Atopy and Bronchial Hyperresponsiveness
- Bronchial Structural Changes in Childhood Asthma