J Korean Ophthalmol Soc.
2011 May;52(5):574-581. 10.3341/jkos.2011.52.5.574.
The Long-Term Observation of Intraocular Pressure after Intravitreal Injections of Bevacizumab, Triamcinolone, and Combination of Both
- Affiliations
-
- 1Department of Ophthalmology, Hanyang University College of Medicine, Seoul, Korea. goddns76@hanmail.net
- KMID: 2214577
- DOI: http://doi.org/10.3341/jkos.2011.52.5.574
Abstract
- PURPOSE
The present study investigates the onset and frequency of increased intraocular pressure (IOP) after injections of intravitreal Bevacizumab, Triamcinolone, and a combination of both drugs.
METHODS
Patients were classified into three groups: Bevacizumab group (group A), Triamcinolone group (group B), and combined drug group (group C), irrespective of the underlying causes. The IOP was measured 30 minutes prior to followed by one day, one week, one month, two months, three months, six months, and one year after injection. The measured IOP at each time point was compared with the pre-injection IOP and the differences in IOP among the three groups were statistically analyzed. The relationships between various factors possible of increasing IOP were also analyzed.
RESULTS
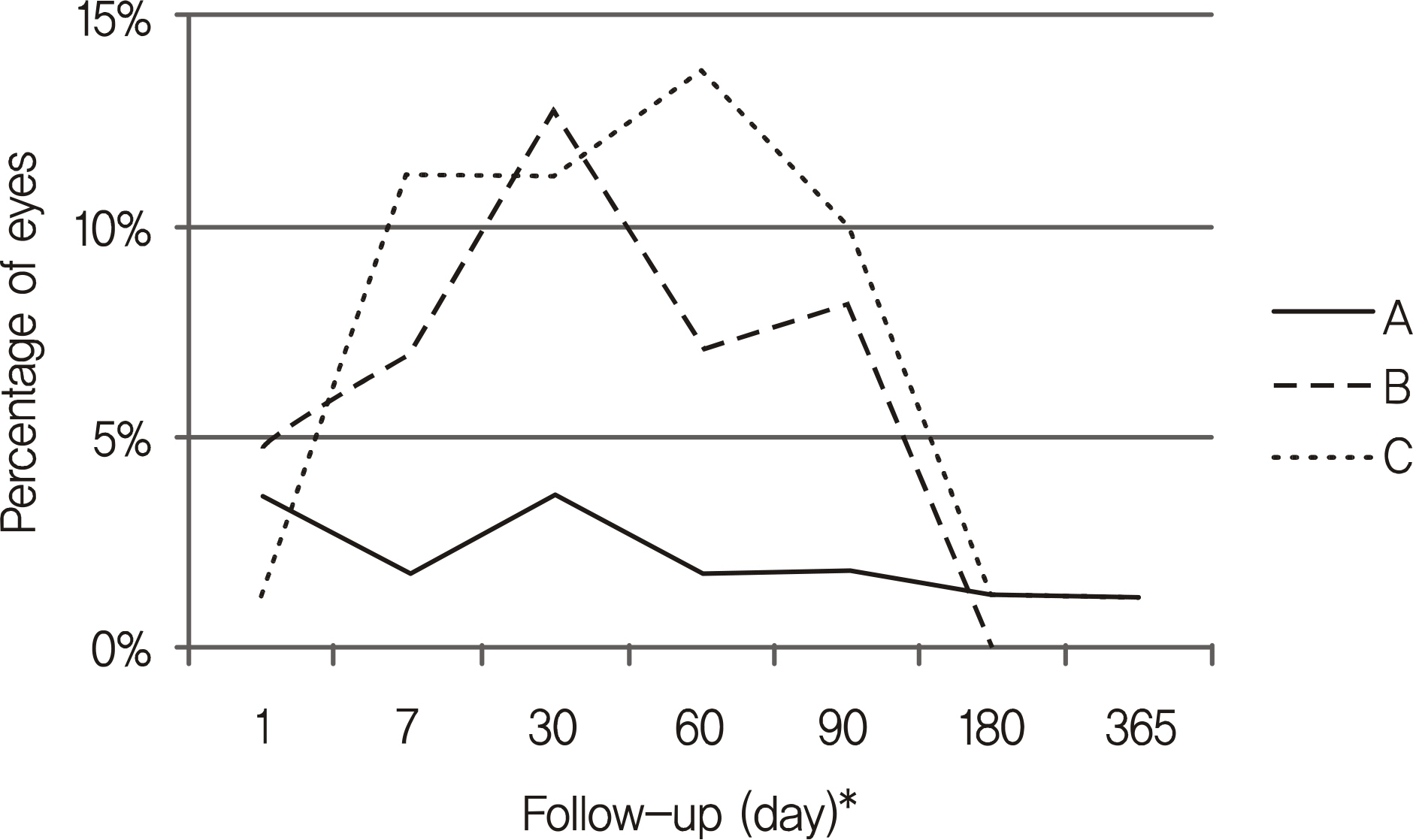
A total of 259 subjects were enrolled in the present study. An IOP increase of more than 5 mm Hg was observed in 25 eyes (15%) in group A, 34 eyes (40%) in group B, and 40 eyes (50%) in group C. There was no statistically significant mean IOP change after injection in group A, but in groups B and C there was an increase in mean IOP up until two and three months after injection, respectively. However, eyes in group A with a history of glaucoma showed statistically significant increases in IOP.
CONCLUSIONS
The onsets and frequencies of increased IOP were different with the different drugs. Proper follow-up for increased IOP after injection is necessary based on the type of drug used.
MeSH Terms
Figure
Reference
-
References
1. Chang MW, Kim SW, Oh IK, et al. Intravitreal triamcinolone injection with or without bevacizumab for diabetic macular edema. J Korean Ophthalmol Soc. 2008; 49:1269–74.
Article2. Ahmadieh H, Ramezani A, Shoeibi N, et al. Intravitreal bevacizumab with or without triamcinolone for refractory diabetic macular edema; a placebo-controlled, randomized clinical trial. Graefes Arch Clin Exp Ophthalmol. 2008; 246:483–9.
Article3. Ehrlich R, Ciulla TA, Moss AM, Harris A. Combined treatment of intravitreal bevacizumab and intravitreal triamcinolone in patients with retinal vein occlusion: 6 months of follow-up. Graefes Arch Clin Exp Ophthalmol. 2010; 248:375–80.
Article4. Faghihi H, Roohipoor R, Mohammadi SF, et al. Intravitreal bevacizumab versus combined bevacizumab-triamcinolone versus macular laser photocoagulation in diabetic macular edema. Eur J Ophthalmol. 2008; 18:941–8.
Article5. Folgosa MS, Messias A, Takata C, et al. Single intravitreal injection of triamcinolone combined with bevacizumab for the treatment of diffuse diabetic macular oedema refractory to grid photocoagulation. Acta Ophthalmol. 2010; 88:e297–8.
Article6. Jonas JB, Hayler JK, Söfker A, Panda-Jonas S. Intravitreal injection of crystalline cortisone as adjunctive treatment of proliferative diabetic retinopathy. Am J Ophthalmol. 2001; 131:468–71.
Article7. Park CH, Jaffe GJ, Fekrat S. Intravitreal triamcinolone acetonide in eyes with cystoid macular edema associated with central retinal vein occlusion. Am J Ophthalmol. 2003; 136:419–25.
Article8. Antcliff RJ, Spalton DJ, Stanford MR, et al. Intravitreal triamcinolone for uveitic cystoid macular edema: an optical coherence tomography study. Ophthalmology. 2001; 108:765–72.
Article9. Danis RP, Ciulla TA, Pratt LM, Anliker W. Intravitreal triamcinolone acetonide in exudative age-related macular degeneration. Retina. 2000; 20:244–50.
Article10. Jonas JB, Kreissig I, Degenring R. Intraocular pressure after intravitreal injection of triamcinolone acetonide. Br J Ophthalmol. 2003; 87:24–7.
Article11. Jonas JB, Kreissig I, Degenring R, et al. Secondary chronic open-angle glaucoma after intravitreal triamcinolone acetonide. Arch Ophthalmol. 2003; 121:729–30.
Article12. Roth DB, Realini T, Feuer WJ, et al. Short-term complications of intravitreal injection of triamcinolone acetonide. Retina. 2008; 28:66–70.
Article13. Smithen LM, Ober MD, Maranan L, Spaide RF. Intravitreal triamcinolone acetonide and intraocular pressure. Am J Ophthalmol. 2004; 138:740–3.
Article14. Jonas JB, Degenring RF, Kreissig I, et al. Intraocular pressure elevation after intravitreal triamcinolone acetonide injection. Ophthalmology. 2005; 112:593–8.
Article15. Roth DB, Verma V, Realini T, et al. Long-term incidence and timing of intraocular hypertension after intravitreal triamcinolone acetonide injection. Ophthalmology. 2009; 116:455–60.
Article16. Jung JW, Nam DH, Shyn KH. The complications after intravitreal injection of triamcinolone acetonide. J Korean Ophthalmol Soc. 2007; 48:55–62.17. Kim SJ, Park YM, Lee SU, et al. Short-term safety and efficacy of intravitreal bavacizumab injection. J Korean Ophthalmol Soc. 2009; 50:219–26.
Article18. Falkenstein IA, Cheng L, Freeman WR. Changes of intraocular pressure after intravitreal injection of bevacizumab (avastin). Retina. 2007; 27:1044–7.
Article19. Bakri SJ, Larson TA, Edwards AO. Intraocular inflammation following intravitreal injection of bevacizumab. Graefes Arch Clin Exp Ophthalmol. 2008; 246:779–81.
Article20. Shima C, Sakaguchi H, Gomi F, et al. Complications in patients after intravitreal injection of bevacizumab. Acta Ophthalmologica. 2008; 86:372–6.
Article21. Ozkiriş A, Erkiliç K. Complications of intravitreal injection of triamcinolone acetonide. Can J Ophthalmol. 2005; 40:63–8.22. Yang YH, Kim KR, Yang SW, Yim HB. The effect of intravitreal triamcinolone acetonide on intraocular pressure. J Korean Ophthalmol Soc. 2004; 45:1081–5.23. Smithen LM, Ober MD, Maranan L, Spaide RF. Intravitreal triamcinolone acetonide and intraocular pressure. Am J Ophthalmol. 2004; 138:740–3.
Article24. Bakri SJ, Beer PM. The effect of intravitreal triamcinolone acetonide on intraocular pressure. Ophthalmic Surg Lasers Imaging. 2003; 34:386–90.
Article25. Park HY, Yi K, Kim HK. Intraocular pressure elevation after intravitreal triamcinolone acetonide injection. Korean J Ophthalmol. 2005; 19:122–7.
Article26. Hattenbach LO, Klais C, Koch FH, Gümbel HO. Intravitreous injection of tissue plasminogen activator and gas in the treatment of submacular hemorrhage under various conditions. Ophthalmology. 2001; 108:1485–92.
Article27. Shin JH, Lee SW, Kim DK, et al. Intravitreal injection of triamcinolone acetonide for macular edema: long-term safety and efficacy after two years. J Korean Ophthalmol Soc. 2007; 48:1670–4.28. Chung MS, Chang WH. Efficacy of anterior chamber paracentesis in intravitreal triamcinolone injection. J Korean Ophthalmol Soc. 2005; 46:1328–32.29. Lee MW, Kyung SE, Chang MH. Prophylactic effect of brimoni-dine 0.15% on IOP elevation after intravitreal triamcinolone acetonide injection. J Korean Ophthalmol Soc. 2008; 49:743–52.
Article30. Seong HK, Lee JM, Park YS, Lee BR. Immediate natural course of IOP after IVTA and the effect of preoperative ocular massage. J Korean Ophthalmol Soc. 2007; 48:808–14.31. Kahook MY, Kimura AE, Wong LJ, et al. Sustained elevation in intraocular pressure associated with intravitreal bevacizumab injections. Ophthalmic Surg Lasers Imaging. 2009; 40:293–5.
Article32. Jalil A, Fenerty C, Charles S. Intravitreal bevacizumab (Avastin) causing acute glaucoma: an unreported complication. Eye. 2007; 21:1541.
Article33. Kubota T, Okabe H, Hisatomi T, et al. Ultrastructure of the trabecular meshwork in secondary glaucoma eyes after intravitreal triamcinolone acetonide. J Glaucoma. 2006; 15:117–9.
Article34. Cho P, Lui T. Comparison of the performance of the Nidek NT-2000 noncontact tonometer with the Keeler Pulsair 2000 and the Goldmann applanation tonometer. Optom Vis Sci. 1997; 74:51–8.
Article35. Audren F, Lecleire-Collet A, Erginay A, et al. Intravitreal triamcinolone acetonide for diffuse diabetic macular edema: phase 2 trial comparing 4 mg vs 2 mg. Am J Ophthalmol. 2006; 142:794–9.
Article36. Lee JM, Kim SJ, Yi KY, Kim HK. Intraocular pressure change after secondary intravitreal triamcinolone acetonide injection. J Korean Ophthalmol Soc. 2007; 48:97–102.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effect of Intravitreal Triamcinolone Acetonide and Bevacizumab Injection on Intraocular Pressure
- Comparison of Intravitreal Bevacizumab Alone Injection and Intravitreal Combination Low-Dose Bevacizumab-Triamcinolone Injection or Diabetic Macular Edema
- Intravitreal Triamcinolone Injection with or Without Bevacizumab for Diabetic Macular Edema
- Comparison of Bevacizumab and Combined Low-dose Bevacizumab and Triamcinolone in Central Retinal Vein Occlusion
- Comparison of Short-term Effects of Intravitreal Injection of Three Modalities on Central Retinal Vein Occlusion