Allergy Asthma Immunol Res.
2014 Sep;6(5):415-420. 10.4168/aair.2014.6.5.415.
Clinical Features of Fixed Drug Eruption at a Tertiary Hospital in Korea
- Affiliations
-
- 1Institute of Allergy and Clinical Immunology, Seoul National University Medical Research Center, Seoul, Korea. helenmed@snu.ac.kr
- 2Department of Internal Medicine, Chung-Ang University College of Medicine, Seoul, Korea.
- 3Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea.
- 4Department of Dermatology, Seoul National University College of Medicine, Seoul, Korea.
- KMID: 2181077
- DOI: http://doi.org/10.4168/aair.2014.6.5.415
Abstract
- PURPOSE
Fixed drug eruption (FDE) is characterized by a well-defined erythematous patch, plaque, or bullous eruption that recurs at the same site as the result of systemic exposure to a causative drug, and resolves with or without hyperpigmentation. This study was carried out to identify the common causative drugs and clinical features of FDE in Korea.
METHODS
We reviewed electronic medical records of all patients diagnosed with FDE from January 2000 to December 2010 at a tertiary hospital in Korea.
RESULTS
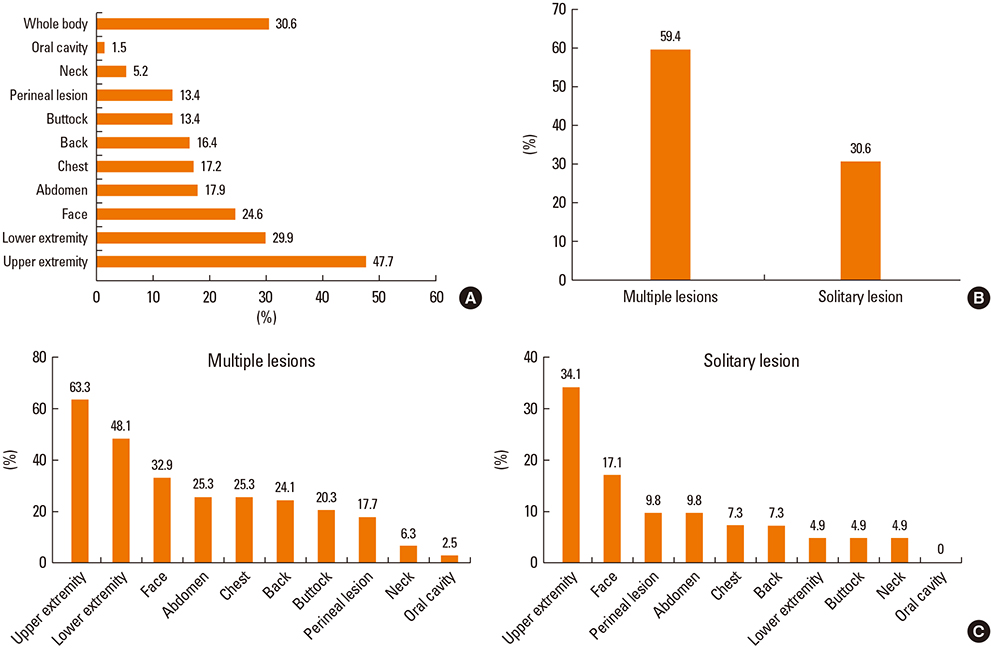
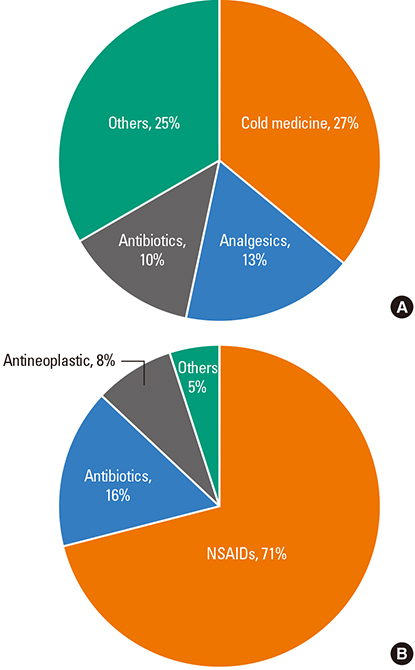
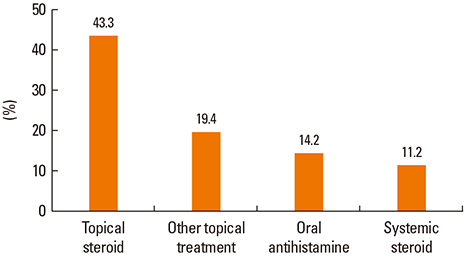
A total of 134 cases were diagnosed as FDE. The mean age was 35.9 years (range, 0-82 years) and 69 (51.5%) of the patients were male. The mean duration from the first event to attending hospital was 1.9 years (range, 1-20 years). The mean number of recurrences was 2.6 (1-10), and 72.6% of patients sought medical care after experiencing symptoms twice or more. Four patients (3.1%) needed hospitalization. The most common sites were the upper extremities (47.7%), followed by the lower extremities, face, abdomen, chest, buttocks and perineum. Clear documentation on the causative drugs was available for 38 patients (28.4%), and among these, non-steroidal anti-inflammatory drugs (NSAIDs) and acetaminophen accounted for 71.1% of cases, and antibiotics accounted for 15.8%. Eighty patients (59.7%) underwent active treatment for FDE, and topical steroids were most frequently prescribed (43.3%), with systemic steroids used in 11.2% of patients.
CONCLUSIONS
NSAIDs and acetaminophen were the main causative agents of FDE, however, the causative agents were not assessed in 25% of patients.
MeSH Terms
-
Abdomen
Acetaminophen
Anti-Bacterial Agents
Anti-Inflammatory Agents, Non-Steroidal
Buttocks
Drug Eruptions*
Electronic Health Records
Hospitalization
Humans
Hyperpigmentation
Korea
Lower Extremity
Male
Perineum
Recurrence
Steroids
Tertiary Care Centers*
Thorax
Upper Extremity
Acetaminophen
Anti-Bacterial Agents
Anti-Inflammatory Agents, Non-Steroidal
Steroids
Figure
Cited by 2 articles
-
Iodinated contrast media-induced fixed drug eruption
Jisu Shim, Soojie Chung, Gun-Woo Kim, Kyoung-Hee Sohn, Ju-Young Kim, Hye-Ryun Kang
Allergy Asthma Respir Dis. 2015;3(5):375-379. doi: 10.4168/aard.2015.3.5.375.Acebrophylline-induced generalized fixed drug eruption confirmed by an oral provocation test
Su Jin Jeong, Heon Sa-Kong, Dong-Hee Park, Sung Geun Lee, So Young Jung, Chan Sun Park
Allergy Asthma Respir Dis. 2017;5(5):298-301. doi: 10.4168/aard.2017.5.5.298.
Reference
-
1. Arndt KA, Jick H. Rates of cutaneous reactions to drugs. A report from the Boston Collaborative Drug Surveillance Program. JAMA. 1976; 235:918–923.2. Bourns DC. Unusual effects of antipyrine. Br Med J. 1889; 2:818–820.3. Brocq L. Eruption erythémato-pigmentée fixé due à l'antipyrine. Ann Dermatol Syphiligr (Paris). 1894; 5:308–313.4. Sehgal VN, Gangwani OP. Fixed drug eruption. Current concepts. Int J Dermatol. 1987; 26:67–74.5. Nigen S, Knowles SR, Shear NH. Drug eruptions: approaching the diagnosis of drug-induced skin diseases. J Drugs Dermatol. 2003; 2:278–299.6. Shelley WB, Shelley ED. Nonpigmenting fixed drug eruption as a distinctive reaction pattern: examples caused by sensitivity to pseudoephedrine hydrochloride and tetrahydrozoline. J Am Acad Dermatol. 1987; 17:403–407.7. Lee AY. Fixed drug eruptions. Incidence, recognition, and avoidance. Am J Clin Dermatol. 2000; 1:277–285.8. Sehgal VN, Srivastava G. Fixed drug eruption (FDE): changing scenario of incriminating drugs. Int J Dermatol. 2006; 45:897–908.9. Bandino JP, Wohltmann WE, Bray DW, Hoover AZ. Naproxen-induced generalized bullous fixed drug eruption. Dermatol Online J. 2009; 15:4.10. Roujeau JC. Clinical heterogeneity of drug hypersensitivity. Toxicology. 2005; 209:123–129.11. Sharma VK, Dhar S, Gill AN. Drug related involvement of specific sites in fixed eruptions: a statistical evaluation. J Dermatol. 1996; 23:530–534.12. Nnoruka EN, Ikeh VO, Mbah AU. Fixed drug eruption in Nigeria. Int J Dermatol. 2006; 45:1062–1065.13. Browne SG. Fixed eruption in deeply pigmented subjects: clinical observations on 350 patients. Br Med J. 1964; 2:1041–1044.14. Sharma VK, Dhar S. Clinical pattern of cutaneous drug eruption among children and adolescents in north India. Pediatr Dermatol. 1995; 12:178–183.15. Mahboob A, Haroon TS. Drugs causing fixed eruptions: a study of 450 cases. Int J Dermatol. 1998; 37:833–838.16. Kanwar AJ, Bharija SC, Singh M, Belhaj MS. Ninety-eight fixed drug eruptions with provocation tests. Dermatologica. 1988; 177:274–279.17. Alanko K, Stubb S, Kauppinen K. Cutaneous drug reactions: clinical types and causative agents. A five-year survey of in-patients (1981-1985). Acta Derm Venereol. 1989; 69:223–226.18. Hayashi H, Shimizu T, Shimizu H. Multiple fixed drug eruption caused by acetaminophen. Clin Exp Dermatol. 2003; 28:455–456.19. Gupta R. Drugs causing fixed drug eruptions: confirmed by provocation tests. Indian J Dermatol Venereol Leprol. 2003; 69:120–121.20. Pudukadan D, Thappa DM. Adverse cutaneous drug reactions: clinical pattern and causative agents in a tertiary care center in South India. Indian J Dermatol Venereol Leprol. 2004; 70:20–24.21. Patel RM, Marfatia YS. Clinical study of cutaneous drug eruptions in 200 patients. Indian J Dermatol Venereol Leprol. 2008; 74:430.22. Nahm DH, Suh CH, Park HS. A case of fixed drug eruption induced by mefenamic acid. J Asthma Allergy Clin Immunol. 1996; 16:346–349.23. Min EJ, Lim DH, Kim JH, Choi SW, Son BK. A case of fixed drug eruption due to acetaminophen. J Korean Pediatr Soc. 2000; 43:1149–1152.24. Hong KB, Jin YM, Kang J, Im IJ, Roh EJ, Son JS, Chung EH. A case of fixed drug eruption caused by acetaminophen in a child. Pediatr Allergy Respir Dis. 2007; 17:314–319.25. Bigby M, Stern R. Cutaneous reactions to nonsteroidal anti-inflammatory drugs. A review. J Am Acad Dermatol. 1985; 12:866–876.26. Bailin PL, Matkaluk RM. Cutaneous reactions to rheumatological drugs. Clin Rheum Dis. 1982; 8:493–516.27. Pasricha JS. Drugs causing fixed eruptions. Br J Dermatol. 1979; 100:183–185.28. Cutaneous reactions to analgesic-antipyretics and nonsteroidal anti-inflammatory drugs. Analysis of reports to the spontaneous reporting system of the Gruppo Italiano Studi Epidemiologici in Dermatologia. Dermatology. 1993; 186:164–169.29. Sehgal VN. Paracetamol-induced bilateral symmetric, multiple fixed drug eruption (MFDE) in a child. Pediatr Dermatol. 1999; 16:165–166.30. Brahimi N, Routier E, Raison-Peyron N, Tronquoy AF, Pouget-Jasson C, Amarger S, Machet L, Amsler E, Claeys A, Sassolas B, Leroy D, Grange A, Dupuy A, Cordel N, Bonnetblanc JM, Milpied B, Doutre MS, Guinnepain MT, Barbaud A, Chosidow O, Roujeau JC, Lebrun-Vignes B, Descamps V. A three-year-analysis of fixed drug eruptions in hospital settings in France. Eur J Dermatol. 2010; 20:461–464.31. Kidon MI, Kang LW, Chin CW, Hoon LS, See Y, Goh A, Lin JT, Chay OM. Early presentation with angioedema and urticaria in cross-reactive hypersensitivity to nonsteroidal antiinflammatory drugs among young, Asian, atopic children. Pediatrics. 2005; 116:e675–e680.32. Barbaud A, Gonçalo M, Bruynzeel D, Bircher A. European Society of Contact Dermatitis. Guidelines for performing skin tests with drugs in the investigation of cutaneous adverse drug reactions. Contact Dermatitis. 2001; 45:321–328.33. Shiohara T. Fixed drug eruption: pathogenesis and diagnostic tests. Curr Opin Allergy Clin Immunol. 2009; 9:316–321.34. Pichler WJ, Tilch J. The lymphocyte transformation test in the diagnosis of drug hypersensitivity. Allergy. 2004; 59:809–820.35. Gimenez-Camarasa JM, Garcia-Calderon P, de Moragas JM. Lymphocyte transformation test in fixed drug eruption. N Engl J Med. 1975; 292:819–821.