J Gynecol Oncol.
2008 Dec;19(4):223-228. 10.3802/jgo.2008.19.4.223.
Stage IIIC epithelial ovarian cancer classified solely by lymph node metastasis has a more favorable prognosis than other types of stage IIIC epithelial ovarian cancer
- Affiliations
-
- 1Department of Obstetrics and Gynecology, University of Ulsan College of Medicine, Asan Medical Center, Seoul, Korea. jhnam@amc.seoul.kr
- KMID: 2173410
- DOI: http://doi.org/10.3802/jgo.2008.19.4.223
Abstract
OBJECTIVE
To verify whether it can be justified to classify patients to stage IIIC epithelial ovarian cancer based on nodal involvement only.
METHODS
This study included all consecutive patients with stage IIIC epithelial ovarian cancer who underwent upfront cytoreductive surgery according to the FIGO guideline followed by platinum based chemotherapy from September 1989 to September 2006 at Asan Medical Center.
RESULTS
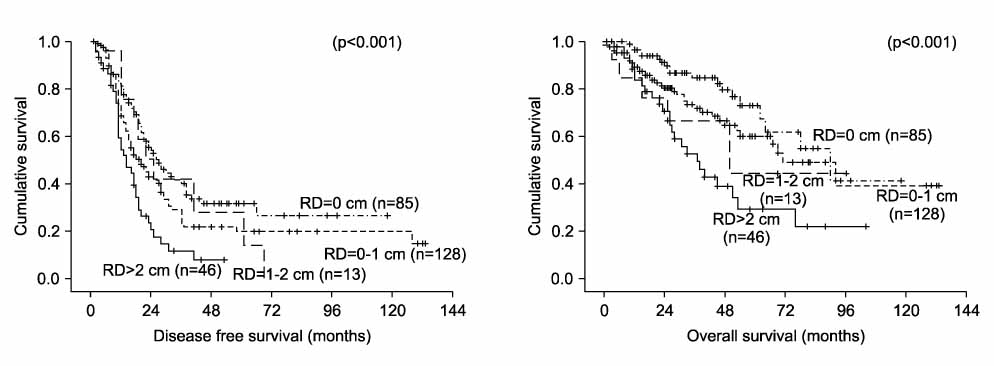
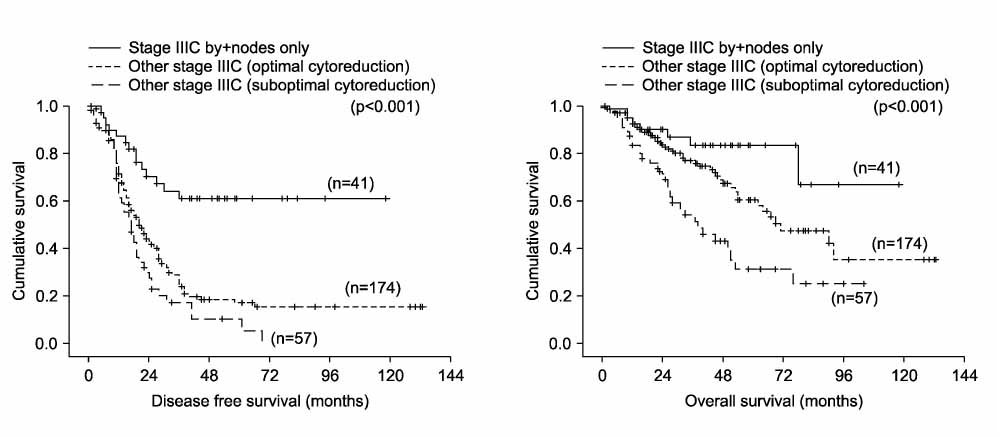
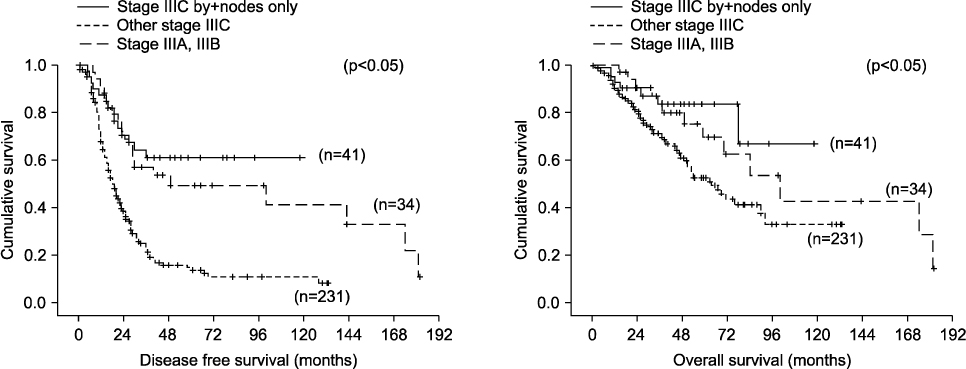
During the study period, a total of 272 patients met the inclusion criteria. Optimal cytoreduction was achieved in 213 patients, and complete cytoreduction was achieved in 85 patients. Median follow-up time was 37 months (range, 6-181 months). The 5-year disease free survival (DFS) and overall survival (OS) rate of all patients were 23% and 57%, respectively. Forty-one patients were allocated to stage IIIC by positive nodes only. Patients with stage IIIC disease due to positive nodes only had significantly longer DFS and OS compared to other stage IIIC patients (p<0.001 and p<0.001). The DFS and OS of these patients was significantly better than those of other stage IIIC patients who achieved complete or optimal cytoreduction (p<0.001 and p<0.001). The outcome was even better than that of stage IIIA and IIIB patients (p<0.05 and p<0.05).
CONCLUSION
Patients with stage IIIC epithelial ovarian cancer due to positive nodes only had a more favorable prognosis compared to other stage IIIC patients. Therefore, reevaluation of the current FIGO staging system for stage IIIC epithelial ovarian cancer is required.
MeSH Terms
Figure
Cited by 1 articles
-
Survival analysis of revised 2013 FIGO staging classification of epithelial ovarian cancer and comparison with previous FIGO staging classification
E Sun Paik, Yoo-Young Lee, Eun-Jung Lee, Chel Hun Choi, Tae-Joong Kim, Jeong-Won Lee, Duk-Soo Bae, Byoung-Gie Kim
Obstet Gynecol Sci. 2015;58(2):124-134. doi: 10.5468/ogs.2015.58.2.124.
Reference
-
1. Jemal A, Siegel R, Ward E, Murray T, Xu J, Smigal C, et al. Cancer statistics, 2006. CA Cancer J Clin. 2006. 56:106–130.2. Korean Society of Obstetrics and Gynecology. Annual report of gynecologic cancer reistry program in Korea for 2004 (2004.1.1-2004.12.31). Korean J Obstet Gynecol. 2007. 50:28–78.3. Young RC, Fuks Z, Hoskins WJ. Devita VT, Hellman S, Rosenberg SA, editors. Cancer of the ovary. Cancer: Principles and practice of oncology. 1989. 3rd ed. Philadelphia: JB Lippincott Co;1162–1196.4. FIGO cancer committee. Staging announcement. Gynecol Oncol. 1986. 25:383–385.5. McGuire WP, Hoskins WJ, Brady MF, Kucera PR, Partridge EE, Look KY, et al. Cyclophosphamide and cisplatin compared with paclitaxel and cisplatin in patients with stage III and stage IV ovarian cancer. N Engl J Med. 1996. 334:1–6.6. Ozols RF. Recurrent ovarian cancer: evidence-based treatment. J Clin Oncol. 2002. 20:1161–1163.7. Knapp RC, Friedman EA. Aortic lymph node metastases in early ovarian cancer. Am J Obstet Gynecol. 1974. 119:1013–1017.8. Chen SS. Survival of ovarian carcinoma with or without lymph node metastasis. Gynecol Oncol. 1987. 27:368–372.9. Spirtos NM, Gross GM, Freddo JL, Ballon SC. Cytoreductive surgery in advanced epithelial cancer of the ovary: the impact of aortic and pelvic lymphadenectomy. Gynecol Oncol. 1995. 56:345–352.10. The oncology committee of the International Federation of Gynecology and Obstetrics. FIGO news: changes to the 1985 FIGO report on the result of treatment in gynaecological cancer. Int J Gynecol Obstet. 1987. 25:87–88.11. Hacker NF, Berek JS, Lagasse LD, Nieberg RK, Elashoff RM. Primary cytoreductive surgery for epithelial ovarian cancer. Obstet Gynecol. 1983. 61:413–420.12. Einhorn N, Nilsson B, Sjöall K. Factors influencing survival in carcinoma of the ovary. Study from a well-defined Swedish population. Cancer. 1985. 55:2019–2025.13. Hoskins WJ, Bundy BN, Thigpen JT, Omura GA. The influence of cytoreductive surgery on recurrence-free interval and survival in small-volume stage III epithelial ovarian cancer: a Gynecologic Oncology Group study. Gynecol Oncol. 1992. 47:159–166.14. Farias-Eisner R, Teng F, Oliveira M, Leuchter R, Karlan B, Lagasse LD, et al. The influence of tumor grade, distribution, and extent of carcinomatosis in minimal residual stage III epithelial ovarian cancer after optimal primary cytoreductive surgery. Gynecol Oncol. 1994. 55:108–110.15. Kaplan EL, Meier P. Nonparametric extimator from incomplete observations. J Am Stat Assoc. 1958. 53:457–481.16. Mantel N. Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemother Rep. 1966. 50:163–170.17. Cox DR. Regression models and life tables. R Stat Soc Bull. 1972. 34:187–220.18. Meier WJ. Lymphadenectomy in stage I ovarian cancer. Proc Am Soc Clin Oncol. 2000. 19:1541.19. Benedetti-Panici P, Greggi S, Maneschi F, Scambia G, Amoroso M, Rabitti C, et al. Anatomical and pathological study of retroperitoneal nodes in epithelial ovarian cancer. Gynecol Oncol. 1993. 51:150–154.20. Petru E, Lahousen M, Tamussino K, Pickel H, Stranzl H, Stettner H, et al. Lymphadenectomy in stage I ovarian cancer. Am J Obstet Gynecol. 1994. 170:656–662.21. Baiocchi G, Raspagliesi F, Grosso G, Fontanelli R, Cobellis L, di Re E, et al. Early ovarian cancer: Is there a role for systematic pelvis and para-aortic lymphadenectomy? Int J Gynecol Cancer. 1998. 8:103–108.22. Burghardt E, Girardi F, Lahousen M, Tamussino K, Stettner H. Patterns of pelvic and paraaortic lymph node involvement in ovarian cancer. Gynecol Oncol. 1991. 40:103–106.23. Lanza A, D'addato F, Valli M, Bussone R, Caldarola B, Re A, et al. Pelvic and para-aortic lymph nodal positivity in the ovarian carcinoma: its prognostic significance. Eur J Gynaecol Oncol. 1988. 9:36–39.24. Carnino F, Fuda G, Ciccone G, Iskra L, Guercio E, Dadone D, et al. Significance of lymph node sampling in epithelial carcinoma of the ovary. Gynecol Oncol. 1997. 65:467–472.25. Kanazawa K, Suzuki T, Tokashiki M. The validity and significance of substage IIIC by node involvement in epithelial ovarian cancer: impact of nodal metastasis on patient survival. Gynecol Oncol. 1999. 73:237–241.26. Cliby WA, Aletti GD, Wilson TO, Podratz KC. Is it justified to classify patients to Stage IIIC epithelial ovarian cancer based on nodal involvement only? Gynecol Oncol. 2006. 103:797–801.27. Onda T, Yoshikawa H, Yasugi T, Mishima M, Nakagawa S, Yamada M, et al. Patients with ovarian carcinoma upstaged to stage III after systematic lymphadenctomy have similar survival to Stage I/II patients and superior survival to other Stage III patients. Cancer. 1998. 83:1555–1560.28. Herzog TJ. Assessing the adequacy of surgical staging for ovarian cancer. Gynecol Oncol. 2006. 103:781–782.29. Ferrandina G, Scambia G, Legge F, Petrillo M, Salutari V. Ovarian cancer patients with "node-positive-only" Stage IIIC disease have a more favorable outcome than Stage IIIA/B. Gynecol Oncol. 2007. 107:154–156.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Analysis of para-aortic lymphadenectomy up to the level of the renal vessels in apparent early-stage ovarian cancer
- A Case of Recurrent Early-stage Epithelial Ovarian Cancer Presenting as Bone Metastasis
- Metastatic axillary node ratio predicts recurrence and poor long-term prognosis in patients with advanced stage IIIC (pN3) breast cancer
- Selective adjuvant radiation therapy for distant lymph node metastasis in patients with stage 4B epithelial ovarian cancer: a case series
- Survival analysis of revised 2013 FIGO staging classification of epithelial ovarian cancer and comparison with previous FIGO staging classification