Cancer Res Treat.
2014 Apr;46(2):178-185.
Diagnostic Value of Circulating Extracellular miR-134, miR-185, and miR-22 Levels in Lung Adenocarcinoma-Associated Malignant Pleural Effusion
- Affiliations
-
- 1Department of Internal Medicine, Cheongju St. Mary Hospital, Cheongju, Korea.
- 2Bioevaluation Center, Korea Research Institute of Bioscience and Biotechnology, Cheongwon, Korea.
- 3Department of Pathology, Chungbuk National University College of Medicine, Cheongju, Korea.
- 4Department of Internal Medicine, Chungbuk National University College of Medicine, Cheongju, Korea. sook3529@hanmail.net
Abstract
- PURPOSE
The accurate and timely diagnosis of malignant pleural effusion (MPE) in lung cancer patients is important because MPE has a poor prognosis and is classified as stage IV disease. Molecular biomarkers for pleural effusion, such as circulating extracellular microRNAs (miRNAs) isolated from pleural fluid, may help in the diagnosis of MPE. The present study examined whether miRNAs that are deregulated in lung cancer (miR-134, miR-185, and miR-22) can serve as diagnostic markers for lung adenocarcinoma-associated MPE (LA-MPE).
MATERIALS AND METHODS
Real-time reverse transcription quantitative polymerase chain reaction was used to measure the expression of the three miRNAs in samples from 87 patients with pleural effusion comprising 45 LA-MPEs and 42 benign pleural effusions (BPEs). The area under the receiver operating characteristic curve (AUC) was then used to evaluate the diagnostic performance of each of the three miRNAs and compare it with that of the common tumor marker, carcinoembryonic antigen (CEA).
RESULTS
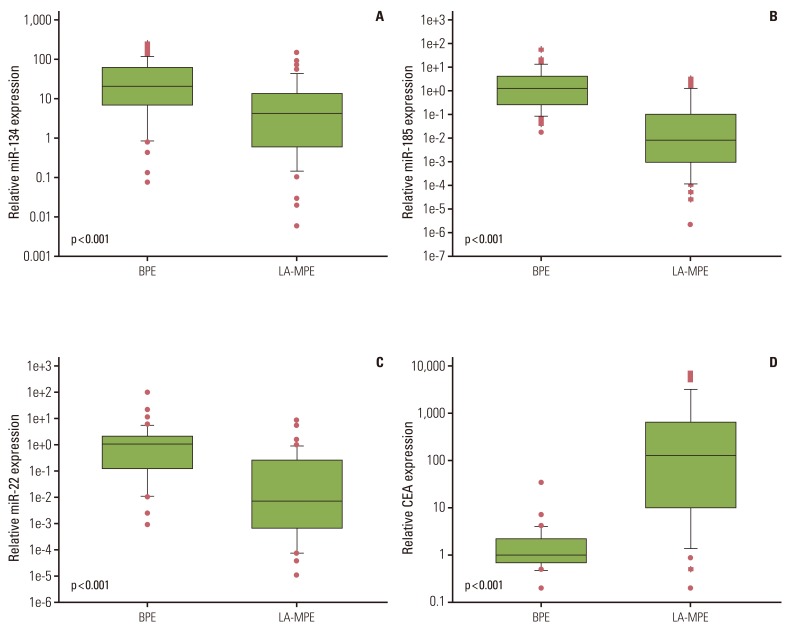
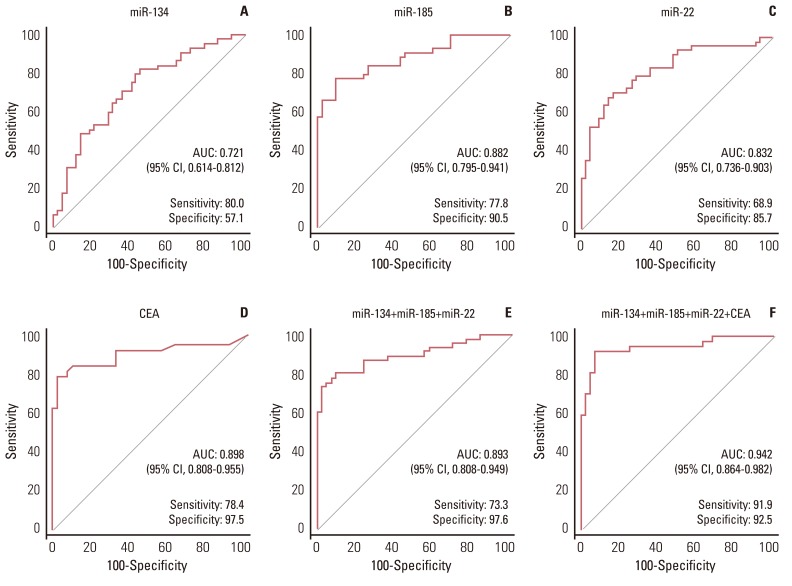
The expression of all three miRNAs was significantly lower in LA-MPE than in BPE (p <0.001). The AUCs for miR-134, miR-185, miR-22, and CEA were 0.721, 0.882, 0.832, and 0.898, respectively. Combining CEA with the three miRNAs increased the diagnostic performance, yielding an AUC of 0.942 (95% confidence interval, 0.864 to 0.982), with a sensitivity of 91.9% and a specificity of 92.5%.
CONCLUSION
The present study suggests that the expression levels of circulating extracellular miR-134, miR-185, and miR-22 in patients with pleural effusion may have diagnostic value when differentiating between LA-MPE and BPE.
Keyword
MeSH Terms
Figure
Reference
-
1. Memon A, Zawadzki ZA. Malignant effusions: diagnostic evaluation and therapeutic strategy. Curr Probl Cancer. 1981; 5:1–30. PMID: 6263549.
Article2. Johnston WW. The malignant pleural effusion: a review of cytopathologic diagnoses of 584 specimens from 472 consecutive patients. Cancer. 1985; 56:905–909. PMID: 4016683.
Article3. Goldstraw P, Crowley J, Chansky K, Giroux DJ, Groome PA, Rami-Porta R, et al. The IASLC Lung Cancer Staging Project: proposals for the revision of the TNM stage groupings in the forthcoming (seventh) edition of the TNM classification of malignant tumours. J Thorac Oncol. 2007; 2:706–714. PMID: 17762336.
Article4. See KC, Lee P. Advances in the diagnosis of pleural disease in lung cancer. Ther Adv Respir Dis. 2011; 5:409–418. PMID: 21697244.
Article5. Nance KV, Shermer RW, Askin FB. Diagnostic efficacy of pleural biopsy as compared with that of pleural fluid examination. Mod Pathol. 1991; 4:320–324. PMID: 2068057.6. Gu P, Huang G, Chen Y, Zhu C, Yuan J, Sheng S. Diagnostic utility of pleural fluid carcinoembryonic antigen and CYFRA 21-1 in patients with pleural effusion: a systematic review and meta-analysis. J Clin Lab Anal. 2007; 21:398–405. PMID: 18022924.
Article7. Bartels CL, Tsongalis GJ. MicroRNAs: novel biomarkers for human cancer. Clin Chem. 2009; 55:623–631. PMID: 19246618.
Article8. Cho WC. MicroRNAs: potential biomarkers for cancer diagnosis, prognosis and targets for therapy. Int J Biochem Cell Biol. 2010; 42:1273–1281. PMID: 20026422.
Article9. Zhou L, Zhao YP, Liu WJ, Dong J, Chen WY, Zhang TP, et al. Circulating microRNAs in cancer: diagnostic and prognostic significance. Expert Rev Anticancer Ther. 2012; 12:283–288. PMID: 22316375.
Article10. Cortez MA, Bueso-Ramos C, Ferdin J, Lopez-Berestein G, Sood AK, Calin GA. MicroRNAs in body fluids: the mix of hormones and biomarkers. Nat Rev Clin Oncol. 2011; 8:467–477. PMID: 21647195.11. Zhang X, Wang H, Zhang S, Song J, Zhang Y, Wei X, et al. MiR-134 functions as a regulator of cell proliferation, apoptosis, and migration involving lung septation. In Vitro Cell Dev Biol Anim. 2012; 48:131–136. PMID: 22259016.
Article12. Li J, Wang Y, Luo J, Fu Z, Ying J, Yu Y, et al. miR-134 inhibits epithelial to mesenchymal transition by targeting FOXM1 in non-small cell lung cancer cells. FEBS Lett. 2012; 586:3761–3765. PMID: 23010597.
Article13. Takahashi Y, Forrest AR, Maeno E, Hashimoto T, Daub CO, Yasuda J. MiR-107 and MiR-185 can induce cell cycle arrest in human non small cell lung cancer cell lines. PLoS One. 2009; 4:e6677. PMID: 19688090.
Article14. Ling B, Wang GX, Long G, Qiu JH, Hu ZL. Tumor suppressor miR-22 suppresses lung cancer cell progression through post-transcriptional regulation of ErbB3. J Cancer Res Clin Oncol. 2012; 138:1355–1361. PMID: 22484852.
Article15. Han HS, Yun J, Lim SN, Han JH, Lee KH, Kim ST, et al. Downregulation of cell-free miR-198 as a diagnostic biomarker for lung adenocarcinoma-associated malignant pleural effusion. Int J Cancer. 2013; 133:645–652. PMID: 23354517.
Article16. Rabinowits G, Gercel-Taylor C, Day JM, Taylor DD, Kloecker GH. Exosomal microRNA: a diagnostic marker for lung cancer. Clin Lung Cancer. 2009; 10:42–46. PMID: 19289371.
Article17. Xie L, Wang T, Yu S, Chen X, Wang L, Qian X, et al. Cell-free miR-24 and miR-30d, potential diagnostic biomarkers in malignant effusions. Clin Biochem. 2011; 44:216–220. PMID: 21093424.
Article18. Wang T, Lv M, Shen S, Zhou S, Wang P, Chen Y, et al. Cell-free microRNA expression profiles in malignant effusion associated with patient survival in non-small cell lung cancer. PLoS One. 2012; 7:e43268. PMID: 22937028.
Article19. Johnson CD, Esquela-Kerscher A, Stefani G, Byrom M, Kelnar K, Ovcharenko D, et al. The let-7 microRNA represses cell proliferation pathways in human cells. Cancer Res. 2007; 67:7713–7722. PMID: 17699775.
Article20. Liu H, Brannon AR, Reddy AR, Alexe G, Seiler MW, Arreola A, et al. Identifying mRNA targets of microRNA dysregulated in cancer: with application to clear cell renal cell carcinoma. BMC Syst Biol. 2010; 4:51. PMID: 20420713.
Article21. Akcakaya P, Ekelund S, Kolosenko I, Caramuta S, Ozata DM, Xie H, et al. miR-185 and miR-133b deregulation is associated with overall survival and metastasis in colorectal cancer. Int J Oncol. 2011; 39:311–318. PMID: 21573504.
Article22. Yamakuchi M, Yagi S, Ito T, Lowenstein CJ. MicroRNA-22 regulates hypoxia signaling in colon cancer cells. PLoS One. 2011; 6:e20291. PMID: 21629773.
Article23. Zhang J, Yang Y, Yang T, Liu Y, Li A, Fu S, et al. microRNA-22, downregulated in hepatocellular carcinoma and correlated with prognosis, suppresses cell proliferation and tumourigenicity. Br J Cancer. 2010; 103:1215–1220. PMID: 20842113.
Article24. Xu D, Takeshita F, Hino Y, Fukunaga S, Kudo Y, Tamaki A, et al. miR-22 represses cancer progression by inducing cellular senescence. J Cell Biol. 2011; 193:409–424. PMID: 21502362.
Article25. Liu L, Jiang Y, Zhang H, Greenlee AR, Yu R, Yang Q. miR-22 functions as a micro-oncogene in transformed human bronchial epithelial cells induced by anti-benzo[a]pyrene-7, 8-diol-9,10-epoxide. Toxicol In Vitro. 2010; 24:1168–1175. PMID: 20170724.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- miR-205 and miR-200c: Predictive Micro RNAs for Lymph Node Metastasis in Triple Negative Breast Cancer
- MicroRNA-374a Expression as a Prognostic Biomarker in Lung Adenocarcinoma
- Serum miR-3620-3p as a Novel Biomarker for Ankylosing Spondylitis
- Circulating microRNA expression profiling in young obese Korean women
- Hsa_circ_0129047 sponges miR-665 to attenuate lung adenocarcinoma progression by upregulating protein tyrosine phosphatase receptor type B