Allergy Asthma Immunol Res.
2014 Jan;6(1):75-82. 10.4168/aair.2014.6.1.75.
A Quantitative Study of Airway Changes on Micro-CT in a Mouse Asthma Model: Comparison With Histopathological Findings
- Affiliations
-
- 1Department of Radiology, Soonchunhyang University Bucheon Hospital, Bucheon, Korea. radpsh@schmc.ac.kr.
- 2Division of Allergy and Respiratory Medicine, Soonchunhyang University Bucheon Hospital, Bucheon, Korea.
- 3Department of Radiology, Chonbuk National University Medical School, Jeonju, Korea.
- KMID: 2166943
- DOI: http://doi.org/10.4168/aair.2014.6.1.75
Abstract
- PURPOSE
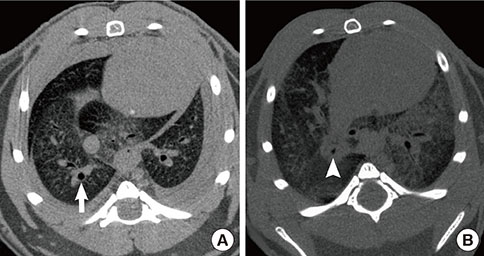
To evaluate airway changes in ovalbumin-induced asthmatic mice in terms of postmortem micro-CT images and pathological findings.
METHODS
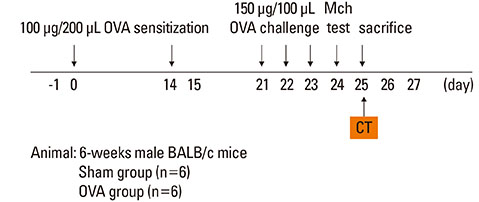
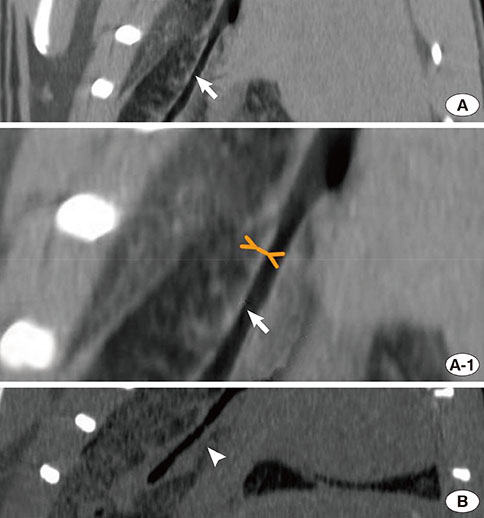
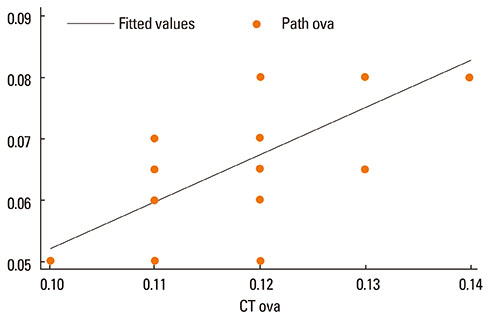
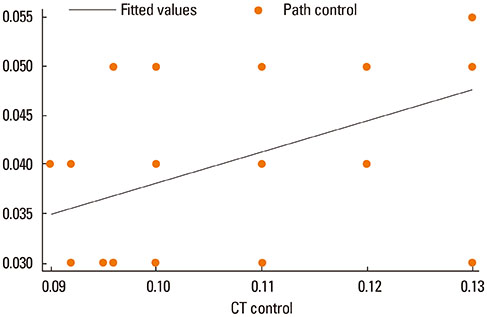
Asthma was induced in mice by intraperitoneal injection and nasal instillation of ovalbumin aluminium hydroxide into mice (experimental group, n=6), and another group of mice received intraperitoneal injection and nasal instillation of distilled phosphate-buffered saline (control group, n=6). Bronchial lumen area was measured in the main bronchial lumen of the distal third bronchial branch level (6 parts per each mouse) on axial scans of Micro-CT, using a Lucion's smart pen (semi-automated) and a curve pen (manual). Bronchial wall thickness was obtained in 4 sections (2 levels on either side) after the third bronchial branch by measuring the diameter which was perpendicular to the longitudinal axis of the main bronchus on curved Multi-planar reconstruction (MPR) images. Histologic slides were obtained from the lesion that was matched with its CT images, and bronchial wall thicknesses were determined.
RESULTS
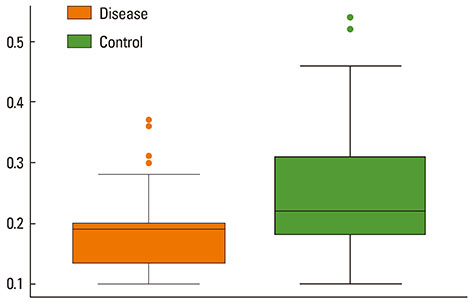
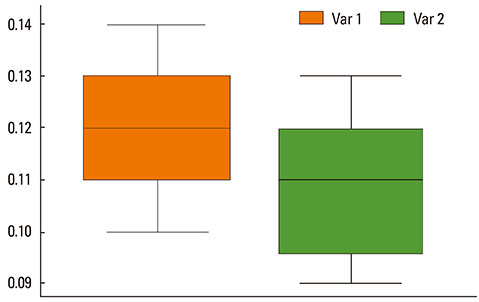
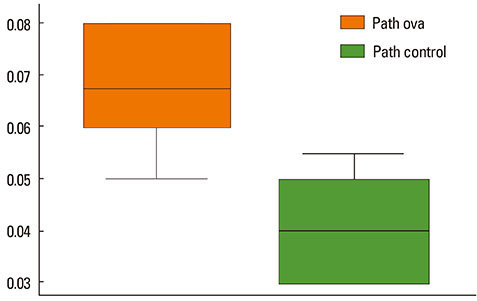
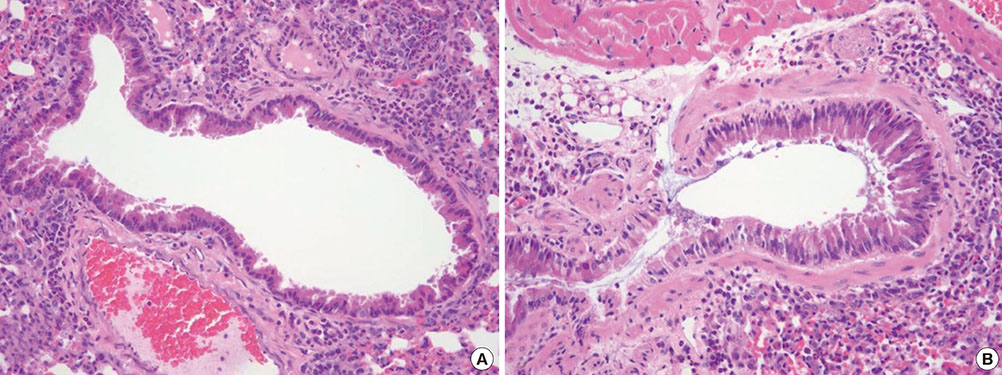
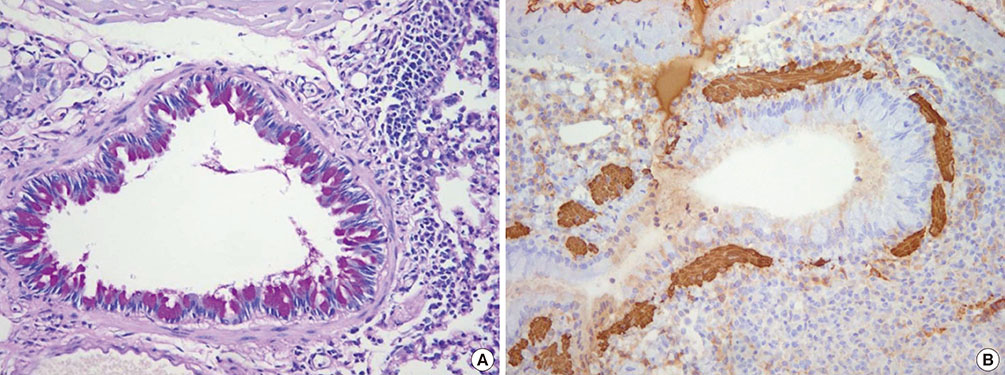
The mean bronchial lumen area was 0.196+/-0.072 mm2 in the experimental group and 0.243+/-0.116 mm2 in the control group; the difference was significant. Bronchial wall thickness on micro-CT images (mean, 0.119+/-0.01 vs. 0.108+/-0.013 mm) and in pathological specimens (mean, 0.066+/-0.011 vs. 0.041+/-0.009 mm) were thicker in the experimental group than in the control group; bronchial wall thickness on micro-CT images correlated well with pathological thickness (for the experimental group, r=0.712; for the control group, r=0.46). The thick bronchial wall in the experimental group demonstrated submucosal hypertrophy along with goblet cell hyperplasia and smooth muscle hyperplasia.
CONCLUSIONS
The results of this study suggest that asthma may induce thickening of bronchial wall and narrowing of the lumen area on micro-CT images and that these results may significantly correlate with pathological findings.
Keyword
MeSH Terms
Figure
Reference
-
1. Standards for the diagnosis and care of patients with chronic obstructive pulmonary disease (COPD) and asthma. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, November 1986. Am Rev Respir Dis. 1987; 136:225–244.2. Bousquet J, Chanez P, Lacoste JY, Barnéon G, Ghavanian N, Enander I, Venge P, Ahlstedt S, Simony-Lafontaine J, Godard P, et al. Eosinophilic inflammation in asthma. N Engl J Med. 1990; 323:1033–1039.3. Bousquet J, Jeffery PK, Busse WW, Johnson M, Vignola AM. Asthma. From bronchoconstriction to airways inflammation and remodeling. Am J Respir Crit Care Med. 2000; 161:1720–1745.4. Rau WS, Hauenstein K, Mittermayer C, Volk P. A simple and rapid method for postmortem radiographic investigation of lung fine structure. Pathol Res Pract. 1980; 170:426–432.5. Feldkamp LA, Goldstein SA, Parfitt AM, Jesion G, Kleerekoper M. The direct examination of three-dimensional bone architecture in vitro by computed tomography. J Bone Miner Res. 1989; 4:3–11.6. Langheinrich AC, Leithäuser B, Greschus S, Von Gerlach S, Breithecker A, Matthias FR, Rau WS, Bohle RM. Acute rat lung injury: feasibility of assessment with micro-CT. Radiology. 2004; 233:165–171.7. Lim KY, Lee HJ, Lee CH, Son KR, Seo JB, Goo JM, Im JG. Detection of the anatomic structure and pathology in animal lung specimens: comparison of micro CT and multi-detector row CT. J Korean Radiol Soc. 2006; 54:385–391.8. Cody DD, Nelson CL, Bradley WM, Wislez M, Juroske D, Price RE, Zhou X, Bekele BN, Kurie JM. Murine lung tumor measurement using respiratory-gated micro-computed tomography. Invest Radiol. 2005; 40:263–269.9. Cavanaugh D, Johnson E, Price RE, Kurie J, Travis EL, Cody DD. In vivo respiratory-gated micro-CT imaging in small-animal oncology models. Mol Imaging. 2004; 3:55–62.10. Froese AR, Ask K, Labiris R, Farncombe T, Warburton D, Inman MD, Gauldie J, Kolb M. Three-dimensional computed tomography imaging in an animal model of emphysema. Eur Respir J. 2007; 30:1082–1089.11. Lee HJ, Goo JM, Kim NR, Kim MA, Chung DH, Son KR, Kim HC, Lee CH, Park CM, Chun EJ, Im JG. Semiquantitative measurement of murine bleomycin-induced lung fibrosis in in vivo and postmortem conditions using microcomputed tomography: correlation with pathologic scores--initial results. Invest Radiol. 2008; 43:453–460.12. Lundblad LK, Thompson-Figueroa J, Allen GB, Rinaldi L, Norton RJ, Irvin CG, Bates JH. Airway hyperresponsiveness in allergically inflamed mice: the role of airway closure. Am J Respir Crit Care Med. 2007; 175:768–774.13. Paganin F, Trussard V, Seneterre E, Chanez P, Giron J, Godard P, Sénac JP, Michel FB, Bousquet J. Chest radiography and high resolution computed tomography of the lungs in asthma. Am Rev Respir Dis. 1992; 146:1084–1087.14. Newman KB, Lynch DA, Newman LS, Ellegood D, Newell JD Jr. Quantitative computed tomography detects air trapping due to asthma. Chest. 1994; 106:105–109.15. Okazawa M, Müller N, McNamara AE, Child S, Verburgt L, Paré PD. Human airway narrowing measured using high resolution computed tomography. Am J Respir Crit Care Med. 1996; 154:1557–1562.16. Park CS, Müller NL, Worthy SA, Kim JS, Awadh N, Fitzgerald M. Airway obstruction in asthmatic and healthy individuals: inspiratory and expiratory thin-section CT findings. Radiology. 1997; 203:361–367.17. Silva CI, Colby TV, Müller NL. Asthma and associated conditions: high-resolution CT and pathologic findings. AJR Am J Roentgenol. 2004; 183:817–824.18. Kasahara K, Shiba K, Ozawa T, Okuda K, Adachi M. Correlation between the bronchial subepithelial layer and whole airway wall thickness in patients with asthma. Thorax. 2002; 57:242–246.19. Brown PJ, Greville HW, Finucane KE. Asthma and irreversible airflow obstruction. Thorax. 1984; 39:131–136.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Understanding asthma using animal models
- Understanding the Mouse Model of Respiratory Allergic Diseases
- Noninvasive monitoring of mouse renal allograft rejection using micro-CT
- Fluorescence bioimaging analysis of collagen antibody-induced arthritis in male mice
- Recent advances in airway imaging using micro-computed tomography and computed tomography for chronic obstructive pulmonary disease